The Adrenal Medulla: Catecholamine Secretion and the Acute Stress Response
The adrenal medulla is the inner region of the adrenal gland and functions as a specialised neuroendocrine organ. Derived from neural crest cells, it acts as an extension of the sympathetic nervous system, releasing catecholamines—adrenaline (epinephrine) and noradrenaline (norepinephrine)—directly into the bloodstream. These hormones mediate the body’s rapid “fight-or-flight” response, preparing multiple organ systems for immediate action. The adrenal medulla allows the sympathetic nervous system to exert widespread and coordinated effects within seconds, representing one of the most dynamic and powerful endocrine pathways in the body.
What You Need to Know
The adrenal medulla is a specialised neuroendocrine organ that functions as part of the sympathetic nervous system rather than as a classical endocrine gland. It secretes the catecholamines adrenaline (epinephrine) and noradrenaline (norepinephrine) directly into the bloodstream in response to acute stress. These hormones produce rapid, widespread effects that prepare the body for immediate physical or psychological challenge, commonly described as the fight-or-flight response.
Catecholamine release is triggered by preganglionic sympathetic fibres that synapse directly onto chromaffin cells within the adrenal medulla. This means secretion is fast, precisely timed, and closely linked to neural activity. Adrenaline is the dominant hormone released and accounts for most of the systemic metabolic effects, while noradrenaline contributes more strongly to vascular tone and blood pressure regulation. Together, they act within seconds, far more rapidly than steroid hormones.
The major physiological effects of adrenal medullary catecholamines include:
increased heart rate and myocardial contractility, raising cardiac output
vasoconstriction in skin and viscera with vasodilation in skeletal muscle
bronchodilation and increased respiratory rate
mobilisation of glucose and fatty acids to provide immediate energy
heightened alertness and redistribution of blood flow to vital organs
These effects are short-lived and tightly regulated, allowing the body to respond quickly to danger and return to baseline once the stressor resolves. By acting as a bridge between the nervous and endocrine systems, the adrenal medulla enables rapid amplification of sympathetic signals, ensuring that acute stress responses are both coordinated and effective.
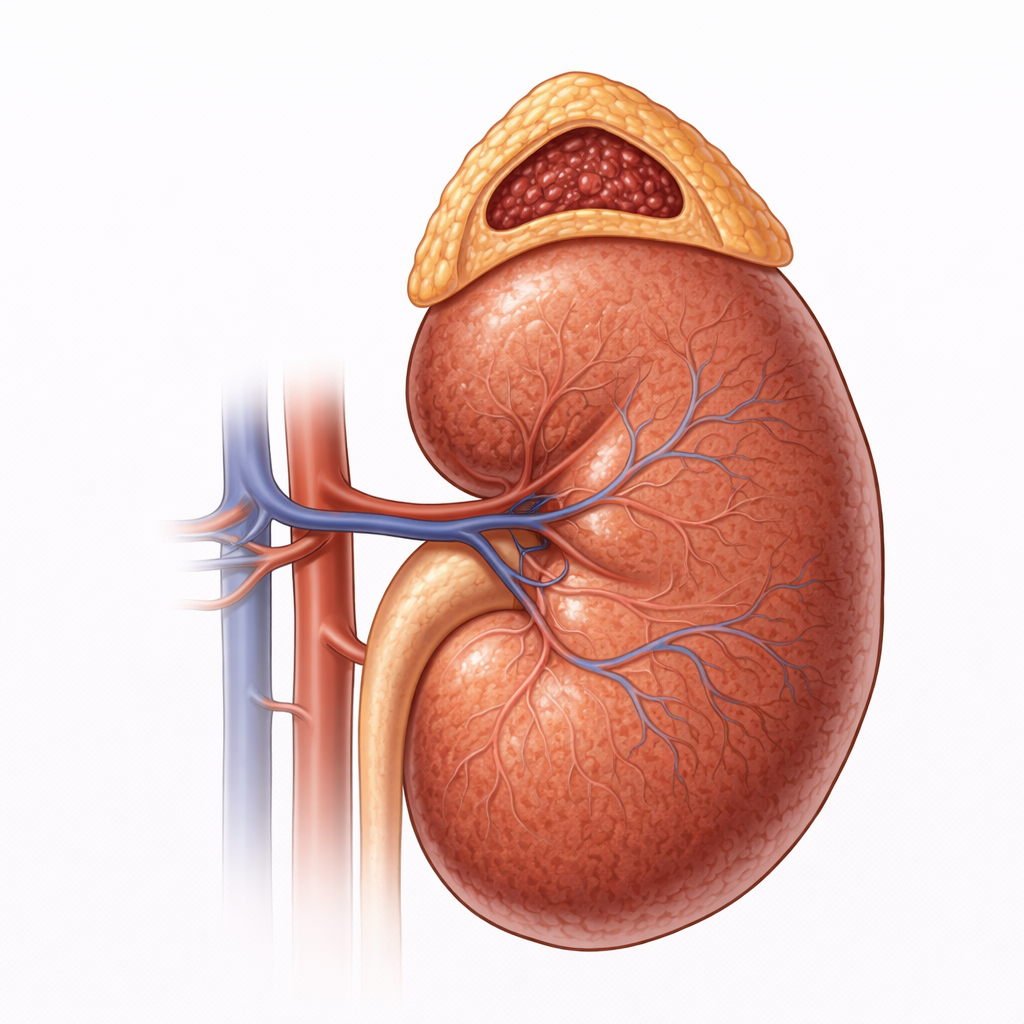
Image: The adrenal gland sits on the superior pole of the kidney and consists of an outer cortex surrounding a central medulla. The adrenal medulla forms a compact inner core and secretes catecholamines, primarily adrenaline and noradrenaline, in response to sympathetic stimulation, driving the body’s acute stress response.
Beyond the Basics
Structure and neural origins
The adrenal medulla is composed of chromaffin cells, which are derived from neural crest tissue and are functionally analogous to postganglionic sympathetic neurons. Unlike typical sympathetic neurons, chromaffin cells do not form synapses with target organs. Instead, they release catecholamines directly into the bloodstream, allowing a single neural signal to produce a widespread systemic response.
Chromaffin cells are innervated by preganglionic sympathetic fibres that release acetylcholine. This direct neural input explains the speed and precision of catecholamine secretion and highlights why the adrenal medulla is considered a neuroendocrine interface rather than a classical endocrine gland.
Synthesis of catecholamines
Catecholamines are synthesised from the amino acid tyrosine through a tightly regulated enzymatic pathway that progresses from tyrosine to L-DOPA, dopamine, noradrenaline, and finally adrenaline. The conversion of noradrenaline to adrenaline is catalysed by phenylethanolamine N-methyltransferase (PNMT), an enzyme expressed almost exclusively in the adrenal medulla.
PNMT activity is strongly stimulated by cortisol delivered from the surrounding adrenal cortex. This anatomical arrangement ensures that high local cortisol concentrations drive efficient adrenaline synthesis, linking glucocorticoid output to catecholamine production and integrating long-term stress signalling with rapid sympathetic responses.
Mechanisms of catecholamine release
During physical or psychological stress, preganglionic sympathetic neurons release acetylcholine onto chromaffin cells. This causes membrane depolarisation, calcium influx, and rapid exocytosis of catecholamine-containing vesicles. Because catecholamines are released into the circulation rather than across synapses, their effects are more diffuse and longer lasting than those of direct sympathetic nerve stimulation.
This mechanism allows a brief neural signal to generate a sustained hormonal response, ensuring that cardiovascular, metabolic, and respiratory systems are activated simultaneously during acute stress.
Physiological actions
Once released, catecholamines bind to adrenergic receptors distributed throughout the body, producing coordinated organ-specific responses that support the acute stress response. These effects include:
increased heart rate, myocardial contractility, and arterial pressure
bronchodilation and increased respiratory rate
mobilisation of energy through glycogenolysis, gluconeogenesis, and lipolysis
redistribution of blood flow toward skeletal muscle and the heart
suppression of gastrointestinal motility and secretory activity
The balance between adrenaline and noradrenaline action, and the distribution of adrenergic receptor subtypes, determines the precise physiological pattern seen in different stress states.
Integration with the sympathetic nervous system
The adrenal medulla functions as an amplifier of sympathetic activity, extending the reach of the nervous system to tissues that lack direct sympathetic innervation. By releasing catecholamines into the bloodstream, it ensures a coordinated whole-body response rather than a series of isolated local effects.
Beyond cardiovascular and metabolic regulation, catecholamines influence immune cell trafficking, platelet activation, thermoregulation, and alertness. This broad reach explains why adrenal medullary activation plays a central role in acute illness, trauma, hypoglycaemia, and emotional stress, and why dysregulation produces systemic consequences rather than isolated organ effects.
Clinical Connections
Disorders of adrenal medullary function are characterised by excessive or inappropriate catecholamine release, producing dramatic cardiovascular and metabolic effects because adrenaline and noradrenaline act rapidly and systemically. The most important pathological example is pheochromocytoma, a catecholamine-secreting tumour of chromaffin cells, which produces episodic surges of sympathetic activity rather than sustained elevation.
Clinically, excessive catecholamine release produces a recognisable constellation of features:
episodic or paroxysmal hypertension with marked blood pressure variability
palpitations, tachycardia, and arrhythmias due to heightened cardiac stimulation
sweating, pallor, tremor, and anxiety reflecting sympathetic overactivation
headache and hyperglycaemia caused by vasoconstriction and metabolic mobilisation
Because catecholamine secretion may be intermittent, symptoms often occur in sudden attacks rather than continuously, which can delay diagnosis. Persistent or untreated catecholamine excess increases the risk of stroke, myocardial infarction, heart failure, and arrhythmias due to sustained cardiovascular strain.
Beyond endocrine tumours, chronic stress states can produce prolonged elevations in sympathetic tone and circulating catecholamines. Over time, this contributes to hypertension, insulin resistance, dyslipidaemia, endothelial dysfunction, and accelerated cardiovascular disease, highlighting the pathological consequences of sustained activation of what is normally an adaptive acute stress response.
Adrenaline also has critical therapeutic applications. It is used clinically during cardiac arrest to support coronary and cerebral perfusion, in anaphylaxis to reverse bronchoconstriction and vasodilation, and in severe asthma to promote bronchodilation and reduce airway oedema. These uses directly exploit its potent β- and α-adrenergic effects.
Concept Check
Why is the adrenal medulla considered a neuroendocrine organ rather than a typical endocrine gland?
How does cortisol regulate adrenaline synthesis?
What triggers chromaffin cells to release catecholamines?
How do catecholamines prepare the cardiovascular and metabolic systems for acute stress?
Why do pheochromocytomas produce episodic rather than constant symptoms?