Thymic Hormones and the Endocrine Regulation of T-Cell Development
The thymus is a primary lymphoid organ with essential endocrine functions that shape the development and maturation of T-lymphocytes. Sitting in the anterior mediastinum, the thymus plays its most active role in childhood, programming the adaptive immune system to recognise pathogens while avoiding harmful responses to self-antigens. This immunological education is driven by thymic epithelial cells, which secrete hormones and signalling molecules, most notably thymosin, thymopoietin and thymulin. These hormones promote T-cell differentiation, survival and functional competence. Although the thymus gradually involutes with age, its early endocrine activity has lifelong consequences for immune function.
What You Need to Know
The thymus is a central immune organ with an important endocrine function, secreting peptide hormones that regulate the development, maturation, and functional competence of T lymphocytes. These thymic hormones act locally within the thymus and systemically to guide T-cell differentiation, ensuring that emerging T cells are both immunocompetent and self-tolerant. Through this role, the thymus links endocrine signalling directly to adaptive immune function.
Key thymic hormones include thymosin, which promotes T-cell differentiation and survival, thymopoietin, which regulates T-cell development and membrane marker expression, and thymulin, which enhances T-cell function and responsiveness. Together, these hormones orchestrate the complex processes of positive and negative selection that occur within the thymic cortex and medulla, shaping a functional T-cell repertoire.
The endocrine contribution of the thymus can be summarised as supporting:
differentiation of immature lymphoid precursors into functional T cells
expression of appropriate surface receptors and membrane markers
selection of T cells that recognise foreign antigens while tolerating self
maturation of immune competence before T cells enter the circulation
The thymus is largest and most hormonally active during childhood and early adolescence, a period when immune education is most intense. With increasing age, the thymus undergoes physiological involution and is gradually replaced by adipose tissue, leading to reduced thymic hormone output and diminished generation of naïve T cells. Despite this decline, the early endocrine programming provided by the thymus establishes long-lasting adaptive immunity, allowing peripheral T-cell populations to maintain immune defence throughout adult life.
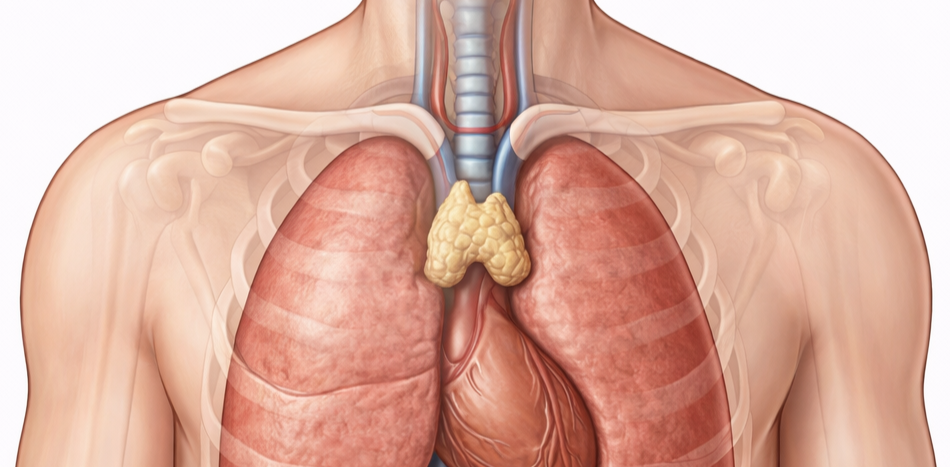
Image: The thymus is located in the anterior superior mediastinum, positioned behind the sternum and anterior to the heart and great vessels. In adults, it appears as a small, lobulated structure that gradually involutes and becomes less prominent over time.
Beyond the Basics
Thymic structure and hormone-secreting cells
The thymus is organised into lobules, each containing a densely cellular cortex and a less cellular medulla. The cortex is packed with immature thymocytes undergoing rapid proliferation and early selection processes, while the medulla contains more differentiated T cells approaching functional maturity. This compartmental organisation allows sequential stages of T-cell development to occur in a tightly controlled microenvironment.
Thymic epithelial cells (TECs) form both the structural scaffold and the functional signalling network of the thymus. These specialised cells secrete thymic hormones, growth factors, and cytokines that guide T-cell maturation. Cortical TECs drive positive selection, ensuring that developing T cells can recognise self–major histocompatibility complex molecules, a prerequisite for effective antigen recognition. Medullary TECs support negative selection by presenting a wide array of self-antigens, eliminating T cells that react too strongly to self and thereby enforcing central immune tolerance.
Thymosin, thymopoietin, and thymulin
Thymosin refers to a family of peptide hormones rather than a single molecule. These peptides promote T-cell differentiation, survival, and expansion, increasing the pool of circulating, immunocompetent T lymphocytes. Through these actions, thymosins enhance peripheral immune responsiveness and help establish a functional adaptive immune system.
Thymopoietin plays a key role in regulating T-cell maturation by influencing the expression of surface markers such as CD3, CD4, and CD8, which define T-cell lineage and function. Thymulin acts in concert with other thymic hormones to enhance T-cell activation and coordination of immune responses. Its biological activity is dependent on adequate zinc availability, and zinc deficiency reduces thymulin function, contributing to impaired cellular immunity.
Age-related involution and endocrine decline
The thymus reaches its maximal size and endocrine activity in early life and begins to involute around puberty. During this process, functional thymic tissue is progressively replaced by adipose tissue, and thymic hormone secretion declines markedly. Although this involution reduces the output of new naïve T cells, peripheral T-cell populations remain functional for many years due to the persistence of long-lived memory T cells generated earlier in life.
Despite this compensation, age-related thymic decline contributes to immunosenescence. Reduced generation of naïve T cells limits the ability to respond to novel antigens, increasing susceptibility to infections, malignancy, and diminished vaccine efficacy in older adults. This decline reflects reduced immune adaptability rather than complete immune failure.
Thymus–endocrine interactions
Thymic function is tightly regulated by interactions with other endocrine systems. Glucocorticoids suppress thymic activity and promote thymocyte apoptosis, particularly during physiological stress or chronic illness. In contrast, growth hormone and insulin-like growth factor 1 support thymic maintenance and T-cell development, linking somatic growth pathways with immune competence.
Sex hormones exert a powerful influence on thymic structure. Rising androgen and oestrogen levels at puberty accelerate thymic involution, explaining the abrupt decline in thymic size and activity during adolescence. These interactions illustrate how immune development is integrated with broader endocrine maturation and life-stage transitions.
Clinical Connections
Abnormalities of thymic structure or function have profound effects on immune competence because the thymus is responsible for endocrine programming of T-cell development and self-tolerance. Disruption at this level alters the quantity, quality, or regulation of T-cells, leading to predictable patterns of immunodeficiency or autoimmunity rather than isolated immune defects.
Clinically, thymic disorders present with characteristic immune consequences:
thymomas, arising from thymic epithelial cells, which are strongly associated with autoimmune diseases such as myasthenia gravis due to impaired negative selection
thymic hypoplasia or aplasia, as seen in DiGeorge syndrome, resulting in severe T-cell deficiency and recurrent, often opportunistic infections
thymic rebound hyperplasia, occurring after chemotherapy or withdrawal of corticosteroids, reflecting renewed thymic activity and recovery of immune function
age- or disease-related thymic failure, contributing to immunosenescence, poor vaccine responses, and increased infection risk
In autoimmune conditions associated with thymomas, defective central tolerance allows autoreactive T-cells to escape deletion, illustrating the thymus’s critical role in preventing immune attack against self. In contrast, congenital or acquired thymic insufficiency produces vulnerability to infection due to inadequate T-cell maturation rather than immune dysregulation.
Therapeutic manipulation of thymic hormones has been explored in selected immune deficiencies and ageing-related immune decline. While thymic peptides can enhance certain aspects of T-cell function experimentally, their routine clinical use remains limited due to variable efficacy and incomplete understanding of long-term effects. Understanding thymic endocrine physiology is therefore essential for interpreting immune disorders rooted in developmental failure, tolerance breakdown, or immune recovery after systemic stress.
Test Yourself
How do thymic epithelial cells contribute to T-cell maturation?
Why does thymic involution not completely eliminate immune function in adulthood?
How do thymic hormones such as thymosin and thymopoietin influence T-cell development?
Why is thymulin activity dependent on zinc?
How do sex hormones influence thymic size and endocrine function?