Lymphocytes: B-cells, T-cells & Immune Memory
Lymphocytes (T-cells, B-cells and natural killer cells) are central to the adaptive immune system, the specialised defence system that targets specific pathogens and develops long-lasting immunity. While neutrophils and macrophages provide rapid, non-specific protection, lymphocytes provide precision, recognition, and memory. Their functions help to explain a variety of immune conepts, such as vaccination, long-term immunity, autoimmune disorders, allergic responses, and immunosuppression. Understanding their development and roles provides critical insight into infection risk, immunodeficiency, clinical interpretation of lymphocyte counts, and the mechanisms behind vaccinations.
What You Need to Know
Lymphocytes are a central component of the adaptive immune system and are responsible for targeted, long-lasting immune protection. All lymphocytes originate from hematopoietic stem cells in the bone marrow, but they differentiate into distinct cell types with specialised functions. Their defining feature is specificity: each lymphocyte is programmed to recognise particular antigens, allowing the immune system to respond precisely rather than indiscriminately.
B-cells mature in the bone marrow and are responsible for humoral immunity, which refers to immune defence mediated by antibodies circulating in body fluids. When a B-cell encounters its specific antigen, usually with help from helper T-cells, it becomes activated and differentiates. Some activated B-cells become plasma cells, which secrete large quantities of antibodies that bind to antigens on pathogens or toxins, marking them for neutralisation or destruction. Other activated B-cells become memory B-cells, which persist long-term and allow for a faster, stronger response if the same antigen is encountered again.
T-cells mature in the thymus and are central to cell-mediated immunity, which targets infected, abnormal, or foreign cells directly rather than free-floating pathogens. T-cells recognise antigens only when they are presented on the surface of other cells via major histocompatibility complex (MHC) molecules. This requirement ensures immune responses are tightly regulated and directed toward genuine threats within tissues.
Key lymphocyte types and their core roles include:
B-cells, which produce antibodies and generate immune memory
Helper T-cells (CD4⁺), which coordinate immune responses by activating B-cells, cytotoxic T-cells, and macrophages
Cytotoxic T-cells (CD8⁺), which directly kill virus-infected or malignant cells
Natural killer (NK) cells, which rapidly eliminate abnormal cells without prior sensitisation
Natural killer cells differ from B- and T-cells in that they do not require prior antigen exposure or specific antigen recognition. Instead, they detect cells with abnormal or reduced MHC expression, a common feature of virus-infected and cancerous cells. This allows NK cells to respond rapidly in the early stages of infection, bridging innate and adaptive immunity.
Lymphocytes provide immune defence that is highly specific, adaptable, and capable of long-term memory. This system allows the body not only to eliminate current threats but also to respond more efficiently to future exposures, forming the biological basis of lasting immunity following infection or vaccination.
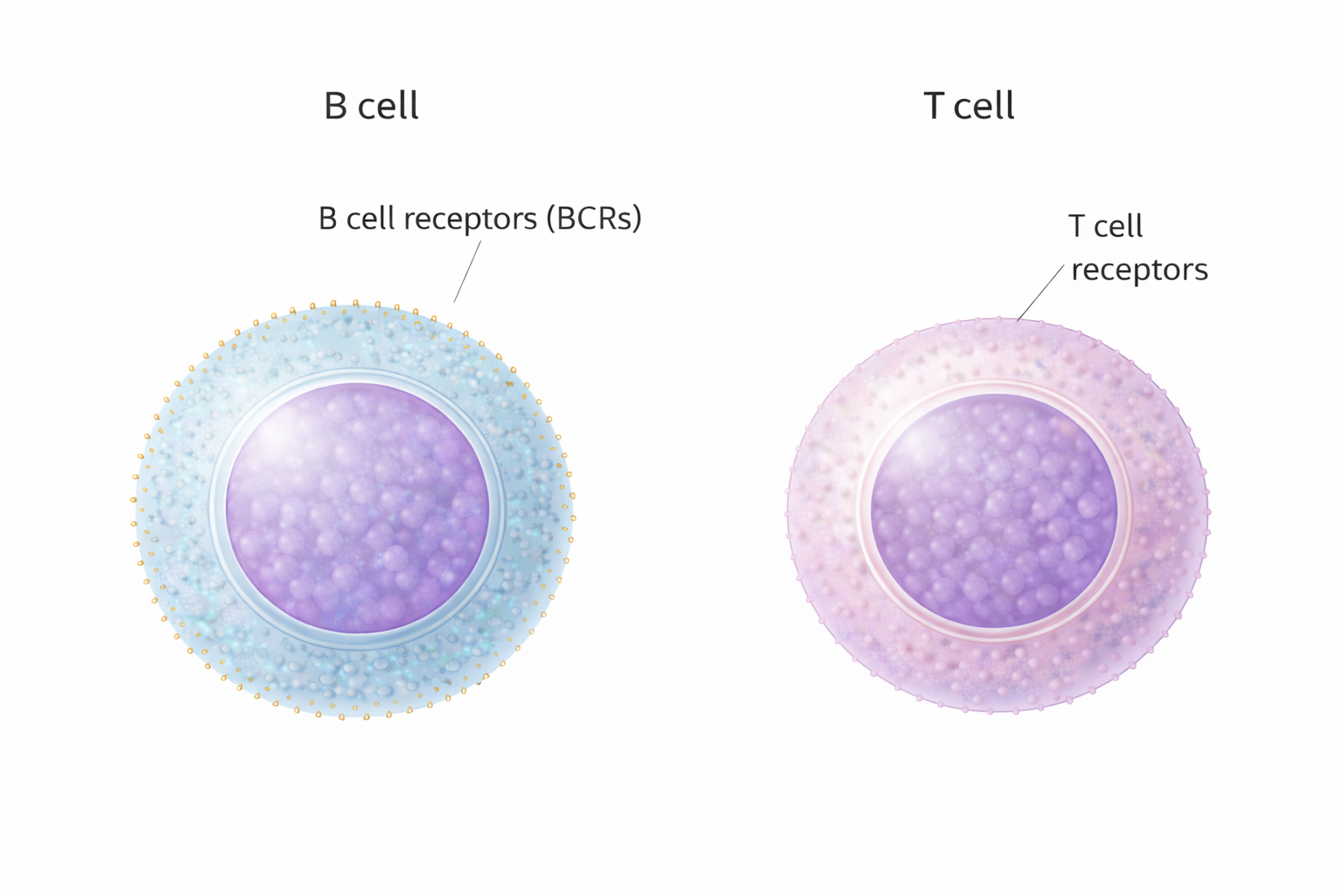
Image: B cells and T cells are lymphocytes characterised by a large central nucleus and a thin rim of cytoplasm. B cells have membrane-bound B cell receptors distributed across their surface, while T cells have T cell receptors, which are less visually prominent but serve a similar antigen-recognition role.
Beyond the Basics
Origin and Lineage Commitment
All lymphocytes arise from haematopoietic stem cells in the bone marrow, but their developmental pathways diverge early to generate functionally distinct immune populations. This shared origin explains why B-cells, T-cells, and natural killer cells are grouped as lymphocytes, even though they participate in different arms of the immune response. Collectively, lymphocytes are responsible for adaptive immunity, characterised by antigen specificity, immune memory, and tightly regulated activation. Natural killer cells retain lymphoid lineage features but also contribute to innate immune defence, responding rapidly without prior sensitisation.
B-Lymphocytes and Antibody-Mediated Immunity
B-cells mature in the bone marrow and form the foundation of humoral immunity. Each B-cell expresses a unique antigen receptor on its surface, generated through gene rearrangement, which allows it to recognise a single specific antigen. This one-cell–one-specificity design enables the immune system to identify an enormous range of pathogens with precision.
When a B-cell encounters its matching antigen, usually with assistance from helper T-cells, it becomes activated and undergoes clonal expansion. This process produces a large population of identical B-cells, all specific for the same antigen. These cells then differentiate into plasma cells and memory B-cells. Plasma cells function as antibody factories, secreting large quantities of antigen-specific antibodies that neutralise pathogens, tag them for phagocytosis, activate complement, and prevent microbial spread. Memory B-cells persist long-term and allow for a faster and more robust response if the same antigen is encountered again, forming the basis of long-lasting immunity following infection or vaccination.
T-Lymphocytes and Immune Coordination
T-cells mature in the thymus, where they undergo rigorous selection to ensure immune competence without self-reactivity. Developing T-cells must demonstrate the ability to recognise foreign antigens while remaining tolerant of self tissues. Cells that fail these checks are eliminated, leaving a population capable of effective but controlled immune responses.
Unlike B-cells, T-cells do not recognise free antigens. Instead, they respond only to antigen fragments presented on major histocompatibility complex molecules displayed on the surface of other cells. This requirement ensures that T-cells target infected, abnormal, or foreign cells within tissues rather than circulating molecules, reducing the risk of inappropriate activation.
Helper T-cells, identified by CD4 expression, act as central regulators of the immune response. Once activated, they release cytokines that activate B-cells, stimulate macrophages, promote cytotoxic T-cell expansion, and shape the overall character of the immune response. Because of this coordinating role, loss of CD4⁺ T-cells, as occurs in advanced HIV infection, results in widespread immune dysfunction rather than failure of a single immune pathway.
Cytotoxic T-cells, identified by CD8 expression, are specialised for direct killing of abnormal cells. They recognise antigen presented on MHC class I molecules, which are expressed by nearly all nucleated cells. When a cell displays abnormal or foreign antigen, cytotoxic T-cells induce programmed cell death through targeted apoptotic mechanisms. This process is essential for controlling viral infections, eliminating cancerous cells, and preventing persistence of intracellular pathogens.
Functional Integration of Lymphocyte Responses
Although B-cells, helper T-cells, and cytotoxic T-cells perform distinct roles, effective immunity depends on their integration rather than isolated action. Antibody production, cellular killing, immune regulation, and memory formation are tightly linked processes. This coordination allows immune responses to be powerful enough to eliminate threats while remaining controlled to minimise tissue damage. Together, B-cells, T-cells, and NK cells provide:
Specificity – targeted responses against distinct antigens
Adaptability – the ability to tailor responses based on the nature of the threat
Memory – enhanced responses upon re-exposure
Regulation – control mechanisms that limit unnecessary tissue damage
Natural Killer (NK) Cells: Rapid Immune Surveillance
NK cells provide immediate immune responses to stressed, infected, or malignant cells without requiring prior sensitisation (the immune system has been previously exposed to a specific antigen and has learned to recognise it). Unlike T-cells, NK cells do not rely on antigen-specific receptors or antigen presentation.
NK cells detect abnormal cells by recognising reduced or absent MHC class I markers (MHC class I mark cells as '“self”), a common feature of virus-infected and cancerous cells. They induce apoptosis (programmed cell death) rapidly and also secrete cytokines that influence broader immune activity. Because of this role, NK cells form a functional bridge between innate and adaptive immunity, providing early defence while adaptive responses are still developing.
Clinical Connections
Lymphocyte counts are a valuable window into immune system function and are routinely interpreted alongside clinical presentation and other blood indices. Because lymphocytes are central to adaptive immunity, abnormal counts often signal infection, immune suppression, or haematological disease rather than isolated laboratory variation.
An elevated lymphocyte count, or lymphocytosis, most commonly reflects an immune response to infection. Viral illnesses are a frequent cause, but persistent or marked lymphocytosis raises concern for lymphoproliferative disorders. In contrast, a reduced lymphocyte count, or lymphopenia, indicates impaired immune capacity and is associated with increased susceptibility to infection and poorer outcomes in systemic illness.
Common clinical interpretations of abnormal lymphocyte counts include:
Lymphocytosis, often seen in viral infections, some chronic bacterial infections, and haematological malignancies such as chronic lymphocytic leukaemia
Lymphopenia, associated with chemotherapy, HIV infection, immunosuppressive medications, severe physiological stress, or malnutrition
Selective lymphocyte dysfunction, where counts may appear normal but functional impairment exists
Defects in specific lymphocyte populations have predictable clinical consequences. Impaired T-cell function, as seen in advanced HIV infection or after certain immunosuppressive therapies, predisposes patients to opportunistic infections, including fungal, viral, and parasitic diseases that rarely affect immunocompetent individuals. B-cell dysfunction reduces antibody production, weakening defence against encapsulated bacteria and increasing susceptibility to recurrent respiratory and bloodstream infections.
Lymphocyte biology is also central to preventive care. Vaccination depends on effective B-cell and T-cell memory formation to provide long-term protection. In immunosuppressed patients, vaccine responses may be reduced or absent, requiring altered vaccination schedules, booster doses, or additional protective strategies.
Concept Check
What are the key differences between B-cells and T-cells?
How do antigen-presenting cells activate T-cells?
Why are memory cells important in vaccination?
What clinical conditions cause lymphopenia?
How do cytotoxic T-cells target virus-infected cells?