THE THYMUS & LYMPHOID ORGANS
The immune system relies on a network of specialised organs that support the development, activation and coordination of immune cells. These structures, collectively known as lymphoid organs, provide the environments necessary for lymphocyte maturation, antigen detection and immune activation. Among them, the thymus plays a uniquely critical role by shaping and educating T cells, ensuring they can recognise pathogens without attacking the body’s own tissues. The bone marrow, thymus, spleen and lymph nodes create a functional pathway through which immune cells are generated, trained and mobilised, allowing the body to mount targeted and efficient immune responses.
What You Need to Know
Lymphoid organs are specialised tissues that support the development, organisation, and activation of immune cells. They are divided into primary and secondary lymphoid organs based on their role in lymphocyte development and immune responses. Primary lymphoid organs are responsible for generating and educating lymphocytes, ensuring that immune cells are functional but tolerant of self. Secondary lymphoid organs provide structured environments where mature immune cells encounter antigens and coordinate immune responses.
Primary lymphoid organs include the bone marrow and thymus, each with a distinct function:
Bone marrow produces all blood cells and is the site of B cell maturation, where developing B cells undergo tolerance checks before entering circulation
The thymus supports T cell maturation, including positive and negative selection processes that establish MHC recognition and self-tolerance
These organs operate continuously, particularly during early life, to establish a competent and self-tolerant immune repertoire. While bone marrow remains active throughout adulthood, thymic activity declines with age, reducing the output of new naïve T cells over time.
Secondary lymphoid organs include lymph nodes, the spleen, and mucosa-associated lymphoid tissue. These structures are strategically positioned to monitor lymphatic fluid, blood, or mucosal surfaces for foreign antigens. Within these organs, immune cells are arranged into defined zones that promote efficient interaction between antigen-presenting cells, T cells, and B cells. This organisation allows rapid activation, clonal expansion, and differentiation of lymphocytes, as well as formation of memory cells that support long-term immunity.
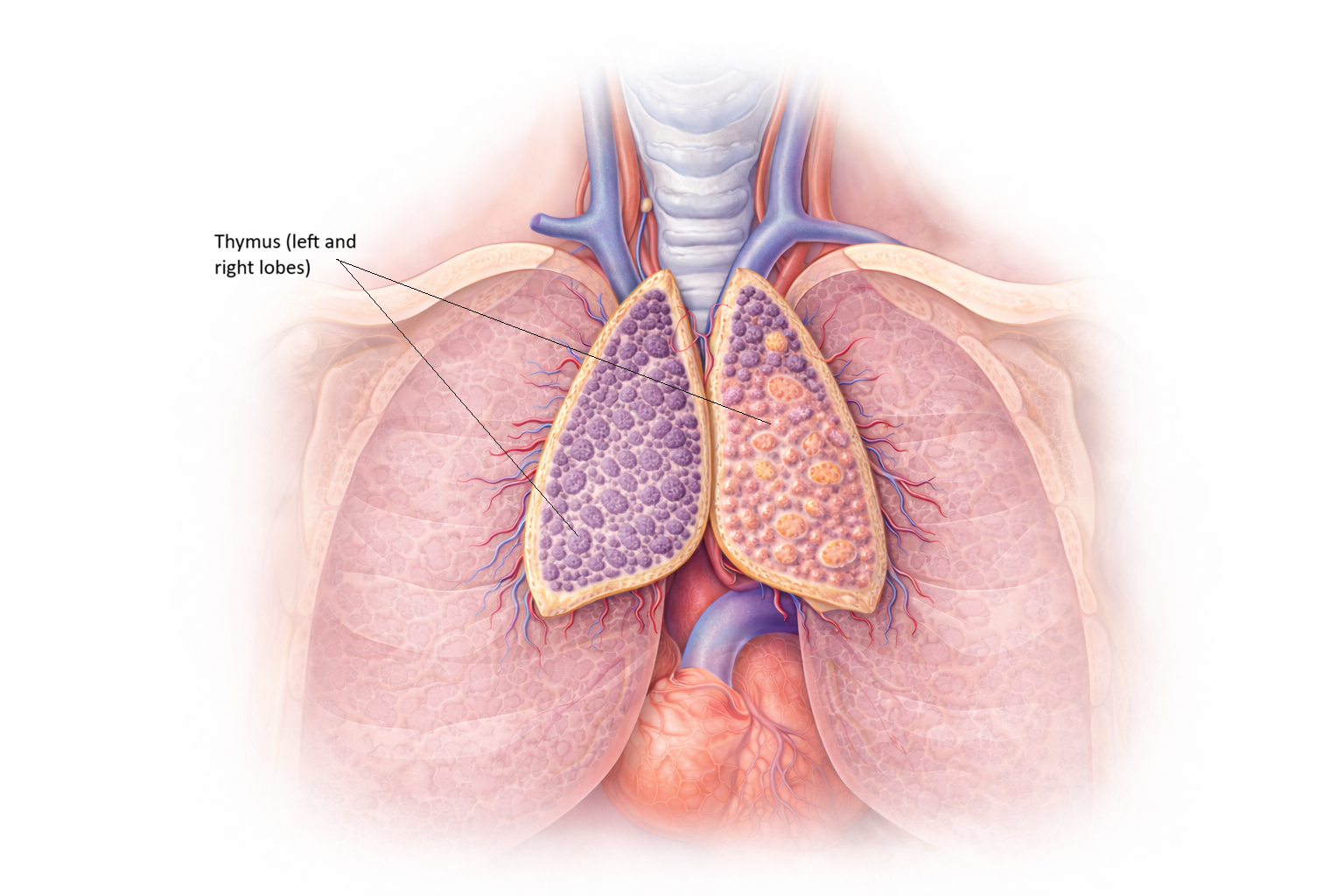
Image: The thymus is a bilobed lymphoid organ located in the anterior superior mediastinum, positioned behind the sternum and in front of the heart. It sits between the lungs and extends superiorly toward the base of the neck, overlying the great vessels.
Beyond the Basics
The Thymus: A Centre for T Cell Education
The thymus is located in the anterior mediastinum, behind the sternum, and plays a central role in establishing a functional and self-tolerant T cell population. It is largest and most active during childhood, with gradual involution after puberty. Although thymic tissue decreases with age, the thymus continues to contribute to immune regulation throughout life.
Within the thymus, immature T cells, known as thymocytes, progress through tightly controlled developmental stages. Positive selection occurs first and ensures that T cells can recognise antigens presented on self-MHC molecules. Thymocytes that fail to recognise MHC undergo apoptosis, as they would be unable to participate in immune responses. Surviving cells then undergo negative selection, where those that bind strongly to self-antigens are eliminated. This step limits the release of autoreactive T cells into circulation. The outcome is a diverse repertoire of T cells capable of recognising foreign antigens while remaining tolerant of self tissues.
Bone Marrow: The Origin of Immune Cells
Bone marrow is the primary site of haematopoiesis, where all blood cells arise from multipotent haematopoietic stem cells. It remains active throughout life and continuously replenishes immune and non-immune cell populations. B lymphocytes complete their maturation within the bone marrow, where they develop functional antigen receptors and undergo tolerance checks to remove or alter strongly self-reactive cells.
In addition to lymphocyte development, bone marrow provides a supportive environment for long-lived plasma cells. These plasma cells occupy specialised niches and can continue producing antibodies for many years after infection or vaccination. This function contributes directly to sustained humoral immunity and long-term immune protection.
Lymph Nodes: Filters of the Lymphatic System
Lymph nodes are distributed along lymphatic vessels and act as filtration points for lymph draining from peripheral tissues. As lymph passes through a node, pathogens, cellular debris, and antigens are trapped and processed. Dendritic cells migrate from tissues to lymph nodes carrying captured antigens, positioning them for interaction with naïve T cells.
Within lymph nodes, immune cells are organised into functional zones that support efficient activation:
T cells localise primarily to paracortical regions where antigen presentation occurs
B cells cluster within follicles, where activation and germinal centre formation take place
Activated lymphocytes proliferate and differentiate before exiting to sites of infection
This organisation allows lymph nodes to coordinate adaptive immune responses in a highly controlled manner.
Spleen: Guardian of the Bloodstream
The spleen plays a specialised role in immune surveillance of the blood. Unlike lymph nodes, it does not filter lymph but instead monitors circulating blood for pathogens and abnormal cells. The white pulp contains organised aggregates of T cells and B cells that respond to blood-borne antigens, while the red pulp removes aged or damaged red blood cells and recycles iron.
Because the spleen is a major site of antibody production against blood-borne organisms, loss of splenic function significantly increases susceptibility to infection. This risk is particularly high for encapsulated bacteria, which rely heavily on antibody and complement-mediated clearance.
Mucosa-Associated Lymphoid Tissue
Mucosa-associated lymphoid tissue includes Peyer’s patches in the intestine, tonsils, adenoids, and bronchial-associated lymphoid tissue. These structures protect mucosal surfaces, which represent the largest and most continuous interface between the body and the external environment. Antigens are sampled directly from the lumen and presented in a way that balances immune defence with tolerance.
MALT plays a key role in generating IgA-producing plasma cells. IgA is secreted across mucosal surfaces and limits pathogen adherence and invasion without provoking excessive inflammation. This localised immune strategy allows effective protection while preserving tissue integrity at sites of constant antigen exposure.
Clinical Connections
Normal function of lymphoid organs is essential for immune competence, and disruption at any level has clear clinical consequences. Impaired thymic function limits T cell development and reduces the diversity of the T cell repertoire. Congenital absence or dysfunction of the thymus, such as in DiGeorge syndrome, leads to profound T cell deficiency and vulnerability to viral, fungal, and opportunistic infections. Bone marrow failure states, including aplastic anaemia, compromise production of all blood cells, resulting in combined defects in innate and adaptive immunity.
Several clinical patterns arise from dysfunction of specific lymphoid organs:
Severe T cell deficiency associated with thymic absence or impaired thymic selection
Broad immunodeficiency due to bone marrow failure affecting lymphocyte production
Increased susceptibility to encapsulated bacterial infections following splenectomy
Lymphadenopathy occurring in infection, inflammation, or malignancy
Loss of splenic function has particularly important clinical implications. The spleen plays a central role in clearance of blood-borne pathogens and antibody-mediated immune responses. Individuals who have undergone splenectomy, or who have functional asplenia, face increased risk of overwhelming infection, especially from encapsulated bacteria. Preventive strategies include targeted vaccination, patient education, and in some cases long-term antibiotic prophylaxis.
Changes in lymph node size and structure often provide important diagnostic clues. Enlarged lymph nodes commonly occur during infection and inflammatory responses as lymphocytes proliferate and immune activity increases. Persistent, non-tender, or unexplained lymphadenopathy raises concern for malignancy. Many cancers of the immune system, including lymphomas and some leukaemias, originate within lymphoid tissues themselves, reflecting the high rate of cell division and immune activation that normally occurs in these organs.
Concept Check
What are the primary differences between primary and secondary lymphoid organs?
How does the thymus ensure that T cells recognise pathogens without attacking self-antigens?
Why are lymph nodes essential for initiating adaptive immune responses?
How does the spleen contribute to immune defence differently from lymph nodes?
What is the role of MALT in protecting mucosal surfaces?