Skin Cancer: Basal Cell Carcinoma, Squamous Cell Carcinoma & Melanoma
Skin cancer develops when cumulative genetic damage overwhelms normal mechanisms of cellular repair, immune surveillance, and growth regulation within the skin. Although often grouped together, skin cancers differ markedly in cell of origin, biological behaviour, capacity for invasion, and metastatic risk. Each type of skin cancer has distinct pathophysiology, which can explain why some lesions remain locally destructive for years, while others metastasise early and carry high mortality despite subtle surface changes.
What You Need to Know
Skin cancer develops when cumulative DNA damage overwhelms normal cellular control mechanisms in the epidermis. Ultraviolet radiation is the dominant driver, causing direct DNA mutations and impairing local immune surveillance that would normally identify and remove abnormal cells. Over time, repeated exposure allows genetically altered cells to survive, proliferate, and acquire additional mutations that increase autonomy from normal growth regulation.
The behaviour of a skin cancer depends on several interacting biological factors rather than malignant transformation alone:
the specific skin cell lineage affected (keratinocyte or melanocyte)
the depth at which malignant cells originate within the epidermis
the capacity of those cells to invade through the basement membrane
access to lymphatic and vascular channels that permit spread beyond the skin
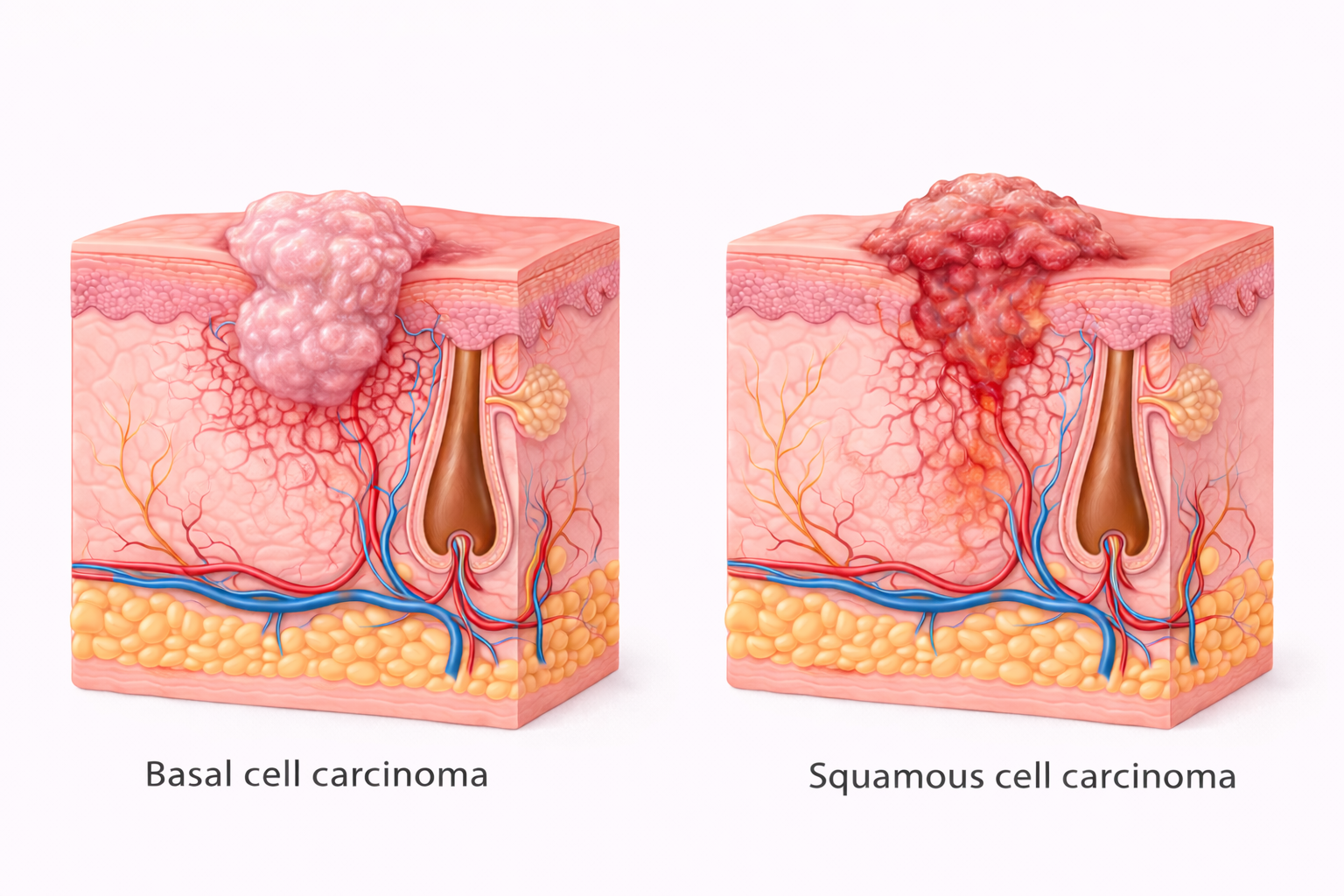
Basal cell carcinoma arises from basal keratinocytes and typically retains dependence on local tissue signals, resulting in slow growth and rare metastasis. Squamous cell carcinoma originates from more differentiated keratinocytes with greater invasive potential, particularly once the basement membrane is breached. Melanoma arises from melanocytes, which are inherently migratory cells derived from the neural crest, explaining their capacity for early invasion and distant spread even when the primary lesion appears small.
Progression from localised skin disease to invasive cancer occurs when malignant cells acquire the ability to degrade the basement membrane, survive outside their original microenvironment, and evade immune destruction. Once these barriers are crossed, tumour behaviour is dictated less by surface appearance and more by depth, biological aggressiveness, and access to systemic pathways. This is why skin cancers vary so widely in risk and outcome, and why they are best understood as a spectrum of biologically distinct diseases rather than a single entity.
Image: Basal cell carcinoma typically presents as a slow-growing, pearly lesion that remains locally invasive, while squamous cell carcinoma is more likely to appear as an irregular, ulcerated lesion with deeper tissue invasion. These differences reflect their underlying behaviour, with BCC rarely metastasising and SCC carrying a greater risk of local destruction and spread.
Beyond the Basics
Basal Cell Carcinoma (BCC)
Pathophysiology of Slow-Growing Keratinocyte Malignancy and Local Tissue Destruction
Basal cell carcinoma originates from basal keratinocytes located in the stratum basale, the proliferative layer responsible for continual epidermal renewal. Because these cells divide throughout life, they accumulate ultraviolet-induced DNA damage more readily than terminally differentiated keratinocytes. Mutations commonly affect tumour suppressor pathways that normally limit cell division and promote apoptosis, allowing persistent but slow expansion of abnormal cells.
Despite malignant transformation, basal keratinocytes retain strong dependence on local tissue signalling and lack the molecular machinery required for effective vascular or lymphatic invasion. Tumour growth therefore occurs through gradual horizontal and downward infiltration rather than dissemination. As malignant cells extend along tissue planes, they progressively destroy surrounding skin, cartilage, and bone, particularly in anatomically confined areas such as the face, nose, and scalp. Morbidity arises from cumulative local destruction rather than systemic spread.
Squamous Cell Carcinoma (SCC)
Pathophysiology of Invasive Keratinocyte Malignancy and Lymphatic Dissemination
Squamous cell carcinoma arises from squamous keratinocytes that form the bulk of the epidermal barrier and are directly exposed to environmental injury. These cells are more differentiated than basal keratinocytes and possess greater intrinsic capacity for invasion once malignant change occurs. Repeated ultraviolet exposure, chronic inflammation, and impaired immune surveillance promote mutations that enable cells to detach, degrade the basement membrane, and infiltrate the dermis.
Once SCC breaches the epidermal–dermal junction, tumour cells encounter a vascularised environment rich in lymphatic channels. This access fundamentally alters disease behaviour by enabling regional nodal spread. Ongoing inflammatory signalling further enhances tumour aggressiveness by promoting angiogenesis and suppressing local immune containment, particularly in chronically damaged skin or immunosuppressed individuals.
Key Biological Differences Driving Behaviour
depth of origin within the epidermis
intrinsic migratory capacity of the cell type
ability to breach the basement membrane
access to lymphatic and vascular pathways
effectiveness of local immune surveillance
Melanoma
Pathophysiology of Melanocyte Malignancy, Early Invasion, and Systemic Metastasis
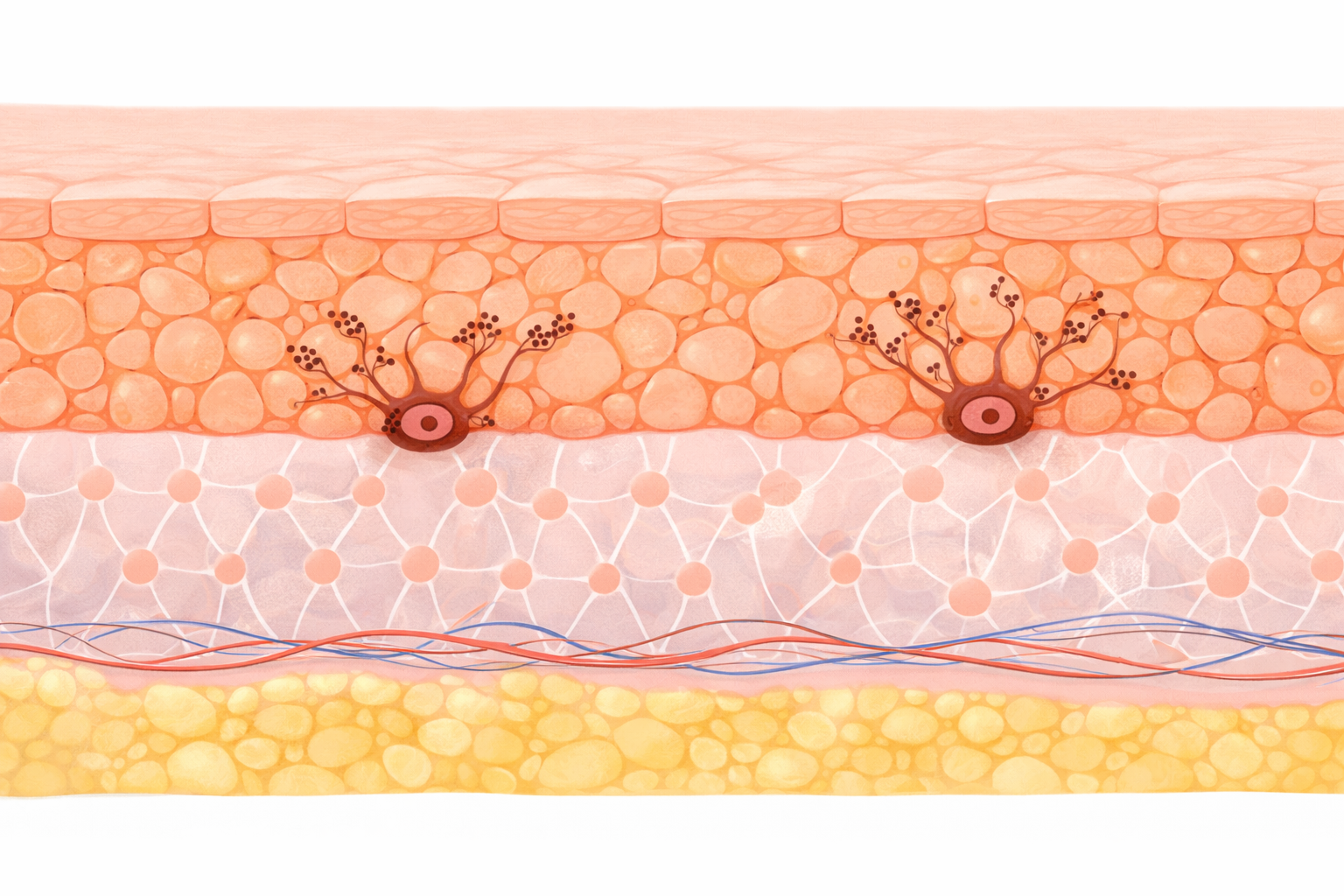
Melanoma develops from melanocytes situated at the dermal–epidermal junction. Unlike keratinocytes, melanocytes are derived from neural crest cells and retain inherent migratory capacity. Malignant transformation amplifies this property, enabling early vertical invasion through the basement membrane even when surface changes appear limited.
As melanoma cells enter the dermis, they rapidly gain access to lymphatic and blood vessels. Progression from radial growth confined to the epidermis to vertical growth into deeper tissue marks a critical biological transition associated with metastatic competence. Genetic instability, immune evasion, and high proliferative capacity allow melanoma to disseminate early, explaining why prognosis correlates more strongly with depth of invasion than surface diameter.
Image: Melanocytes reside in the basal layer of the epidermis and produce melanin, which is transferred to surrounding keratinocytes. This distribution of pigment provides protection against ultraviolet radiation and contributes to overall skin colour.
Basement Membrane Integrity and Disease Escalation
The basement membrane represents a decisive biological barrier separating avascular epidermis from the vascular dermis. While intact, malignant cells remain locally constrained. Once degraded, tumour cells encounter growth factors, immune cells, and transport pathways that enable systemic spread. Differences in how readily cancer cells breach this structure explain the stark contrast between indolent local disease and aggressive metastatic behaviour.
Immune Surveillance and Ultraviolet-Induced Failure
Cutaneous immune surveillance plays a critical role in eliminating dysplastic cells before malignant progression. Ultraviolet radiation suppresses antigen presentation and local immune activation, reducing clearance of mutated cells. When immune containment fails—due to cumulative UV exposure, ageing, or immunosuppression—tumour progression accelerates. This interaction between genetic damage and immune failure underpins both cancer development and variability in disease behaviour.
Why Different Skin Cancers Behave So Differently
The clinical behaviour of skin cancers is determined less by tumour size and more by fundamental biological properties of the cell of origin and its interaction with surrounding tissue. Progression from localised disease to invasive or metastatic cancer depends on several interrelated mechanisms that govern how malignant cells grow, move, and access transport pathways:
Depth and function of the cell of origin within the epidermis, which determines proximity to vascularised tissue
Intrinsic migratory capacity of the malignant cell, retained from normal developmental biology
Ability to degrade and breach the basement membrane, enabling transition from confinement to invasion
Access to lymphatic and vascular channels, which permits regional and distant spread
Effectiveness of local immune surveillance, influencing containment versus progression
These mechanisms explain why basal cell carcinoma remains locally destructive, squamous cell carcinoma acquires regional metastatic risk once invasive, and melanoma can disseminate systemically at an early stage.
Clinical Connections
Skin cancers may present as non-healing lesions, ulceration, induration, bleeding, or evolving pigmentation, but surface appearance alone does not reliably indicate biological behaviour. Lesions with minimal visible change may already have breached the basement membrane or acquired access to lymphatic or vascular pathways, while larger lesions may remain biologically confined. Clinical assessment therefore relies on understanding how different malignant cells grow, invade, and spread rather than on size or symptoms alone.
Management priorities differ because each skin cancer reflects a distinct underlying pathophysiological process. Treatment decisions are guided by how aggressively malignant cells interact with surrounding tissue and their capacity for systemic dissemination:
basal cell carcinoma is managed with complete local excision because disease progression occurs through slow, destructive local infiltration
squamous cell carcinoma requires assessment for dermal invasion and regional nodal involvement due to lymphatic access once the basement membrane is breached
melanoma demands urgent evaluation based on depth of invasion, as early vascular and lymphatic access confers high metastatic risk independent of surface size
Early detection interrupts the transition from confined epidermal malignancy to invasive disease by preventing basement membrane breach and systemic spread. This explains why timely biopsy, accurate depth assessment, and appropriate escalation of management are central to improving outcomes across all skin cancer types, even when lesions appear clinically subtle.
Concept Check
Why does basal cell carcinoma remain locally destructive rather than metastatic?
How does squamous cell carcinoma gain access to lymphatic spread?
Why does melanoma metastasise early compared with other skin cancers?
Why is breaching the basement membrane a critical prognostic event?
How does immune suppression alter skin cancer behaviour?