Benign Prostatic Hyperplasia (BPH)
Benign prostatic hyperplasia is a common, age-related condition characterised by non-malignant enlargement of the prostate gland, leading to progressive bladder outlet obstruction. Although benign, BPH can significantly impair urinary function and quality of life and may contribute to upper urinary tract complications if untreated. BPH symptoms often develop gradually, with obstruction worsening over time. Bladder and renal changes may occur despite the absence of malignancy.
What You Need to Know
Benign prostatic hyperplasia develops as a result of progressive, hormone-dependent enlargement of prostatic tissue surrounding the proximal urethra. The prostate sits immediately below the bladder neck and encircles the urethra, so any increase in tissue volume directly increases resistance to urine flow. As the urethral lumen narrows, the bladder must generate higher pressures to empty, placing increasing functional demand on the detrusor muscle over time.
The cellular growth seen in BPH is driven largely by dihydrotestosterone, a potent androgen formed locally within prostatic tissue. This hormone promotes proliferation of both glandular and stromal cells, leading to nodular enlargement. Growth occurs predominantly in the periurethral transition zone, which is anatomically critical for urinary outflow. Because of this location, even relatively small increases in prostate size can cause clinically significant obstruction, while larger prostates may sometimes produce fewer symptoms depending on growth pattern.
Several core features explain why BPH produces progressive lower urinary tract dysfunction:
Compression of the urethra increases outflow resistance rather than directly impairing bladder contraction
Hormone-driven cellular proliferation continues gradually over years
Transition zone enlargement disproportionately affects urine flow due to its periurethral position
As obstruction develops, bladder emptying becomes less efficient and compensatory changes occur within the bladder wall. Early symptoms arise from increased effort required to void, while later effects reflect structural and functional bladder changes rather than prostate size alone. This explains why symptom severity in BPH does not always correlate directly with measured prostate volume.
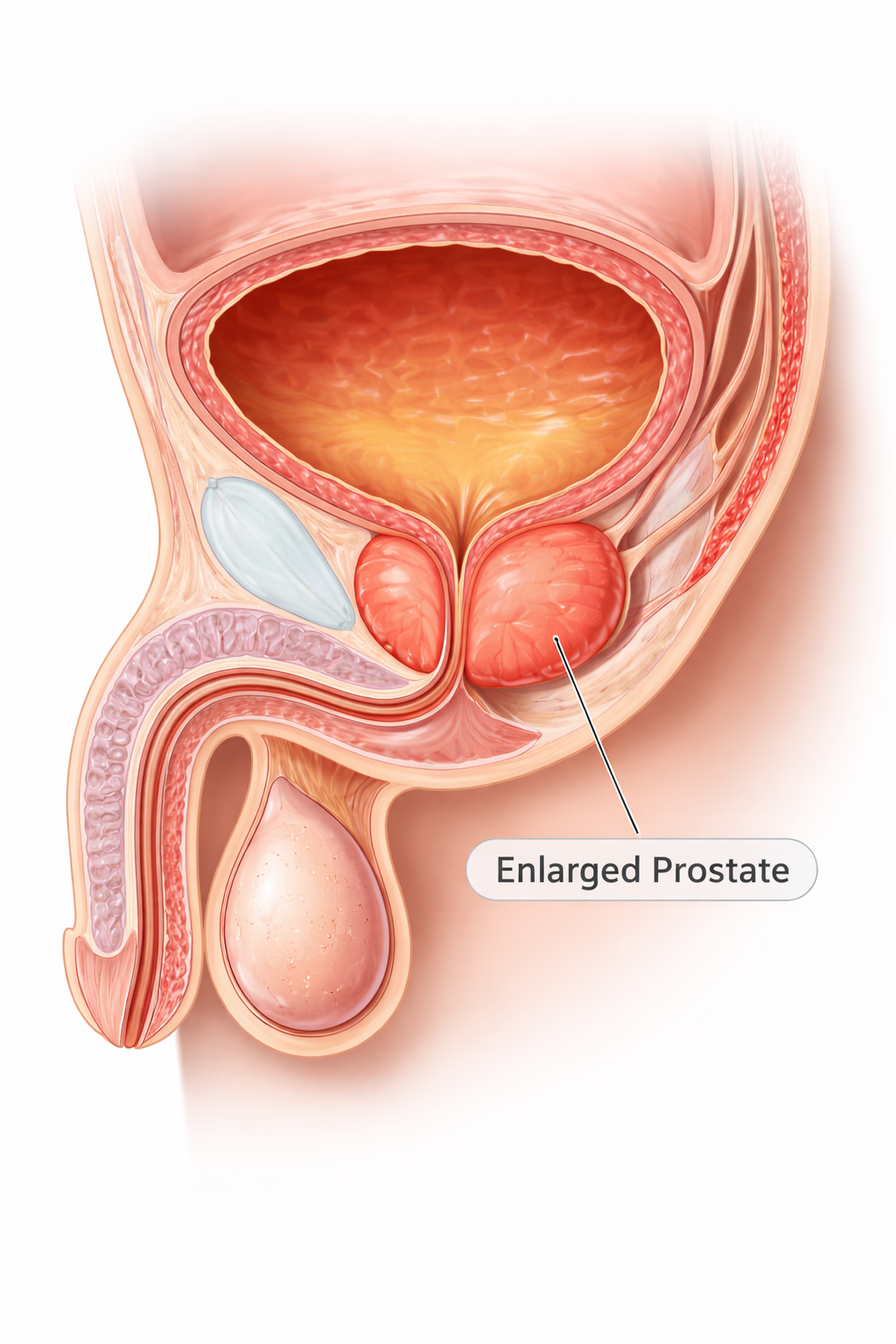
Image: The prostate surrounds the urethra just below the bladder. When enlarged, it compresses the urethral lumen, obstructing urine flow and leading to symptoms such as hesitancy, weak stream, and incomplete bladder emptying.
Beyond the Basics
Androgen regulation and prostatic growth
Prostatic growth in benign prostatic hyperplasia is driven by local androgen signalling rather than circulating testosterone levels alone. Within prostatic cells, testosterone is converted to dihydrotestosterone, a more potent androgen that strongly stimulates cellular proliferation. Although serum testosterone declines with ageing, intraprostatic dihydrotestosterone activity is maintained, allowing growth signals to persist for decades.
This sustained hormonal environment promotes gradual expansion of both glandular and stromal components of the prostate. Importantly, this process represents hyperplasia, an increase in cell number, rather than hypertrophy, an increase in cell size, which explains the slow but progressive nature of prostate enlargement over time.
Static and dynamic components of obstruction
Bladder outlet obstruction in BPH arises through two interacting mechanisms. The static component is caused by physical compression of the urethra as periurethral prostatic tissue enlarges and encroaches on the lumen. This structural narrowing increases baseline resistance to urine flow. Superimposed on this is a dynamic component related to smooth muscle tone within the prostate and bladder neck.
Sympathetic nervous system activity increases contraction of this smooth muscle, further narrowing the urethra. Because smooth muscle tone fluctuates, symptoms may worsen or improve independently of prostate size, particularly in response to stress, medications, or autonomic stimulation.
Bladder compensation and detrusor hypertrophy
As outflow resistance increases, the bladder adapts by generating higher pressures to maintain urine flow. The detrusor muscle undergoes hypertrophy, increasing contractile force to overcome urethral obstruction. In early disease, this compensatory response is effective, allowing relatively normal bladder emptying despite rising resistance. However, the increased workload places sustained mechanical stress on the bladder wall, altering its structure and function over time.
Progression to bladder dysfunction
With prolonged obstruction, compensatory mechanisms begin to fail. The detrusor muscle becomes stiff and less compliant, and contractile efficiency declines. Incomplete bladder emptying develops, leading to increased post-void residual volumes and urinary stasis. This progression explains the shift in symptom pattern over time. Early BPH is dominated by voiding symptoms such as hesitancy, weak stream and straining, whereas later disease is characterised by storage symptoms including frequency, urgency, nocturia and a sensation of incomplete emptying, reflecting bladder dysfunction rather than worsening obstruction alone.
Upper urinary tract consequences
Chronic bladder outlet obstruction can raise intravesical pressure to levels that transmit retrograde stress to the ureters and kidneys. Persistent elevation of pressure impairs ureteric drainage and may lead to hydronephrosis, with dilation of the renal pelvis and calyces. Over time, sustained back-pressure compromises renal function, producing progressive renal impairment. These changes arise from mechanical obstruction and altered pressure dynamics rather than primary renal pathology.
Inflammation and symptom amplification
Urinary stasis and ongoing epithelial stress promote low-grade inflammation within both the prostate and bladder. Inflammatory mediators increase sensory nerve sensitivity and alter smooth muscle behaviour, intensifying urgency, frequency and discomfort.
This inflammatory contribution explains why symptom severity varies widely between individuals and does not correlate reliably with prostate size alone. Symptoms in BPH therefore reflect the combined effects of hormonal growth, mechanical obstruction, bladder adaptation and inflammatory sensitisation rather than enlargement in isolation.
Clinical Connections
Benign prostatic hyperplasia most commonly presents with lower urinary tract symptoms that evolve slowly over time. Early features include hesitancy, weak urinary stream and prolonged voiding, reflecting increasing outflow resistance at the level of the prostate. As obstruction persists, secondary changes within the bladder become more prominent. Nocturia, urgency and frequency develop as detrusor compliance declines and residual urine volume increases, meaning symptoms increasingly reflect bladder dysfunction rather than prostate enlargement alone.
Several clinical features help link symptoms to the underlying mechanisms of obstruction and compensation:
Hesitancy and weak stream arising from urethral compression and increased outlet resistance
Urgency and frequency driven by detrusor overactivity and reduced bladder compliance
Incomplete emptying and rising residual volumes due to detrusor fatigue and impaired contractility
Assessment therefore considers both prostate-related and bladder-related contributors. Digital rectal examination, prostate-specific antigen testing and imaging provide information about prostate size and structure, while symptom patterns and residual urine volumes offer insight into bladder adaptation and failure. Acute urinary retention represents a tipping point where compensatory mechanisms are no longer sufficient, often triggered by infection, medications that increase smooth muscle tone, or further prostate enlargement.
Management targets the dominant physiological drivers of symptoms. Therapies that reduce smooth muscle tone improve dynamic obstruction and relieve symptoms rapidly, while treatments that reduce prostate volume address the static component over time. Failure to address ongoing obstruction allows progressive urinary stasis, increasing the risk of recurrent infection, bladder stone formation and retrograde pressure transmission to the upper urinary tract. Renal impairment in BPH reflects chronic back-pressure and altered bladder dynamics rather than intrinsic kidney disease, reinforcing the importance of timely intervention before irreversible complications develop.
Concept Check
Why does prostatic enlargement disproportionately affect urine flow?
How does dihydrotestosterone drive benign prostatic growth?
What is the difference between static and dynamic obstruction in BPH?
Why does bladder function deteriorate over time in chronic obstruction?
How can BPH contribute to renal dysfunction despite being benign?