MALE REPRODUCTIVE SYSTEM: Accessory Glands
The accessory glands of the male reproductive system play essential roles in producing the fluid components of semen, creating an environment that supports sperm survival, motility and fertilising capacity. Although spermatozoa originate in the testes, they require the biochemical and physiological contributions of these glands to form functional semen. The seminal vesicles, prostate gland and bulbourethral glands each secrete specific substances that optimise sperm transport, neutralise acidity, provide nutrients and protect sperm within the male and female reproductive tracts.
What You Need to Know
The accessory glands of the male reproductive system produce the majority of the fluid component of semen and play a crucial role in supporting sperm survival, motility, and fertilising capacity. These glands include the seminal vesicles, prostate gland, and bulbourethral glands, each contributing distinct secretions that combine with sperm during ejaculation. Together, their coordinated function ensures that sperm are delivered in an environment optimised for transport and function within the female reproductive tract.
The seminal vesicles contribute the largest volume of seminal fluid. Their secretions are rich in fructose, which serves as an energy source for sperm, as well as prostaglandins that influence smooth muscle activity in the female reproductive tract. Clotting proteins produced by the seminal vesicles allow semen to coagulate briefly after ejaculation, helping retain sperm within the vaginal canal. The prostate gland adds a milky, slightly alkaline fluid containing enzymes, zinc, and prostate-specific antigen, which later liquefies the coagulated semen to release sperm and enhance motility. The bulbourethral glands secrete a clear mucus that lubricates the urethra and neutralises residual acidity, protecting sperm as they pass through the urethra.
Accessory gland function is tightly regulated by androgens, particularly testosterone, which maintains glandular structure and secretory activity. Disruption of androgen signalling can alter seminal volume and composition, even when sperm production is intact. Key roles of accessory gland secretions include:
Providing metabolic support, supplying energy substrates and protective factors for sperm
Optimising pH and viscosity, enhancing sperm motility and survival
Facilitating transport and retention, supporting effective sperm delivery after ejaculation
By contributing volume, nutrients, enzymes, and protective compounds, the accessory glands transform sperm into semen capable of surviving the reproductive tract and achieving fertilisation.
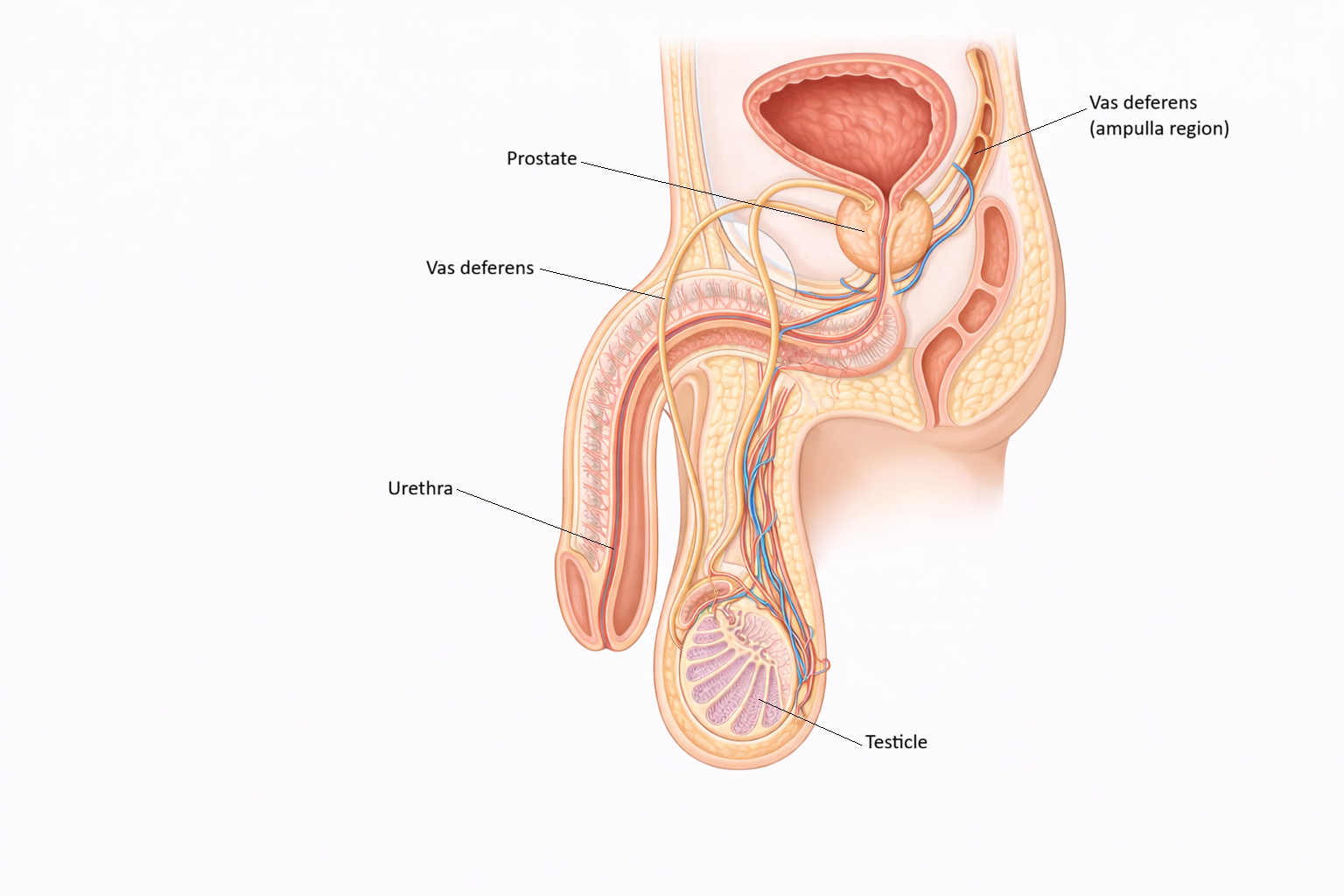
Image: The accessory male reproductive glands include the seminal vesicles (not pictured, located behind the bladder) and prostate, which contribute fluid to semen. The seminal vesicles produce a nutrient-rich, fructose-containing fluid that supports sperm, while the prostate adds an alkaline secretion that helps protect sperm and enhance motility as they pass through the reproductive tract.
Beyond the Basics
Seminal Vesicles: Energy Supply and Semen Structuring
The seminal vesicles are paired, elongated glands positioned posterior to the bladder and superior to the prostate. They contribute the majority of seminal fluid, accounting for roughly 60 to 70 percent of semen volume. Their secretion is thick, viscous, and slightly alkaline, helping buffer acidity encountered in both the male urethra and the female reproductive tract during ejaculation.
Seminal vesicle fluid plays a key energetic and structural role in early post-ejaculatory sperm function. Fructose serves as the primary metabolic substrate for sperm motility, supporting energy production once sperm are released. Prostaglandins influence smooth muscle activity within the female reproductive tract, facilitating sperm transport toward the uterine cavity. Fibrinogen-like proteins promote transient semen coagulation immediately after ejaculation, helping retain sperm within the vagina before later liquefaction allows progressive motility.
Prostate Gland: Enzymatic Activation and pH Regulation
The prostate is a compact, fibromuscular gland located inferior to the bladder and encircling the proximal urethra. It contributes approximately 25 to 30 percent of semen volume and produces a thin, milky, alkaline secretion that plays a critical role in semen activation. During ejaculation, contraction of the prostatic smooth muscle expels this fluid directly into the urethra, where it mixes with sperm and seminal vesicle secretions.
Prostatic fluid contains prostate-specific antigen, a proteolytic enzyme that breaks down coagulated seminal proteins, allowing semen to liquefy several minutes after ejaculation. This process is essential for releasing sperm from the initial coagulum and enabling forward motility. Zinc within prostatic secretions stabilises sperm chromatin and provides antimicrobial protection, while citrate and other enzymes support sperm metabolism and viability. The balance of these components helps optimise pH, viscosity, and enzymatic activity within semen.
Bulbourethral Glands: Lubrication and Urethral Protection
The bulbourethral glands are small, paired glands located in the deep perineal pouch, with ducts opening into the spongy urethra. Although they contribute only a small volume of fluid, their role is functionally important. Their clear, mucus-rich secretion is released prior to ejaculation and acts to lubricate the urethral lining, reducing friction during semen passage.
This pre-ejaculatory fluid also neutralises residual acidity from urine within the urethra, creating a more protective environment for sperm before ejaculation occurs. In addition, bulbourethral secretions may contribute modestly to vaginal lubrication during intercourse, supporting comfort and sperm survival at the time of deposition.
Hormonal Regulation and Changes Across the Lifespan
Development, growth, and maintenance of the accessory glands are regulated primarily by androgens, particularly testosterone and its more potent derivative dihydrotestosterone. Androgen signalling preserves glandular structure, secretory capacity, and smooth muscle function required for ejaculation. Disruption of androgen production or receptor activity alters semen volume and composition, even when spermatogenesis remains intact.
With advancing age, structural and functional changes occur within the accessory glands. Prostatic enlargement due to proliferation of stromal and epithelial tissue can compress the urethra, leading to urinary symptoms characteristic of benign prostatic hyperplasia. Seminal vesicle size and secretory output may also decline, contributing to reduced semen volume. These age-related changes reflect altered hormonal responsiveness rather than abrupt loss of reproductive capacity, highlighting the continued endocrine dependence of accessory gland function across the lifespan.
Clinical Connections
Normal male fertility depends not only on sperm production, but also on intact accessory gland function and unobstructed delivery of glandular secretions into the ejaculate. Disorders affecting the prostate, seminal vesicles, or ejaculatory ducts can therefore impair fertility even when spermatogenesis is normal. Because accessory gland secretions make up the majority of semen volume, dysfunction often presents as changes in semen volume, consistency, or biochemical composition rather than complete absence of sperm.
Several common clinical conditions disrupt accessory gland structure or output, with effects on both reproductive and urinary function:
Prostatitis, which may alter semen composition, cause pelvic pain, and impair ejaculation
Benign prostatic hyperplasia, leading to urinary obstruction, incomplete bladder emptying, and secondary infection risk
Prostate cancer, which can affect urinary flow, sexual function, and semen parameters depending on disease extent and treatment
Seminal vesicle or ejaculatory duct obstruction, resulting in low semen volume and infertility despite normal testicular sperm production
Accessory gland pathology is closely linked to lower urinary tract symptoms. Prostatic enlargement can compress the urethra, impairing urinary flow and increasing residual urine volume, which predisposes to recurrent urinary tract infections and bladder dysfunction. Inflammatory or obstructive processes within the ejaculatory ducts may also cause painful ejaculation or haematospermia, reflecting local glandular involvement.
Understanding the contribution of each accessory gland is essential when interpreting semen analysis. Low semen volume or absent fructose suggests seminal vesicle dysfunction or obstruction, while altered semen liquefaction or abnormal prostate-specific antigen levels point toward prostatic disease. Because semen parameters reflect the combined output of the testes, epididymis, duct system, and accessory glands, abnormalities should be assessed in anatomical and physiological context rather than attributed solely to impaired sperm production.
Concept Check
What components of seminal vesicle fluid support sperm survival and transport?
How does prostatic fluid contribute to the liquefaction of semen after ejaculation?
Why is the alkalinity of seminal and prostatic fluid important for reproductive success?
What roles do the bulbourethral glands play before and during ejaculation?
How might dysfunction of the seminal vesicles or prostate influence male fertility?