OVARIAN STRUCTURE & FOLLICULOGENESIS
The ovaries are paired reproductive organs responsible for housing and maturing oocytes, producing sex hormones and coordinating the cyclical changes that define the female reproductive lifespan. Their structure is uniquely designed to support both long-term storage of primordial follicles and the carefully regulated process of follicular development that culminates in ovulation. Folliculogenesis— the maturation of ovarian follicles from their resting state to ovulatory readiness— is a complex, multi-stage process that integrates endocrine signalling, local paracrine pathways and dynamic structural changes within the ovarian cortex.
What You Need to Know
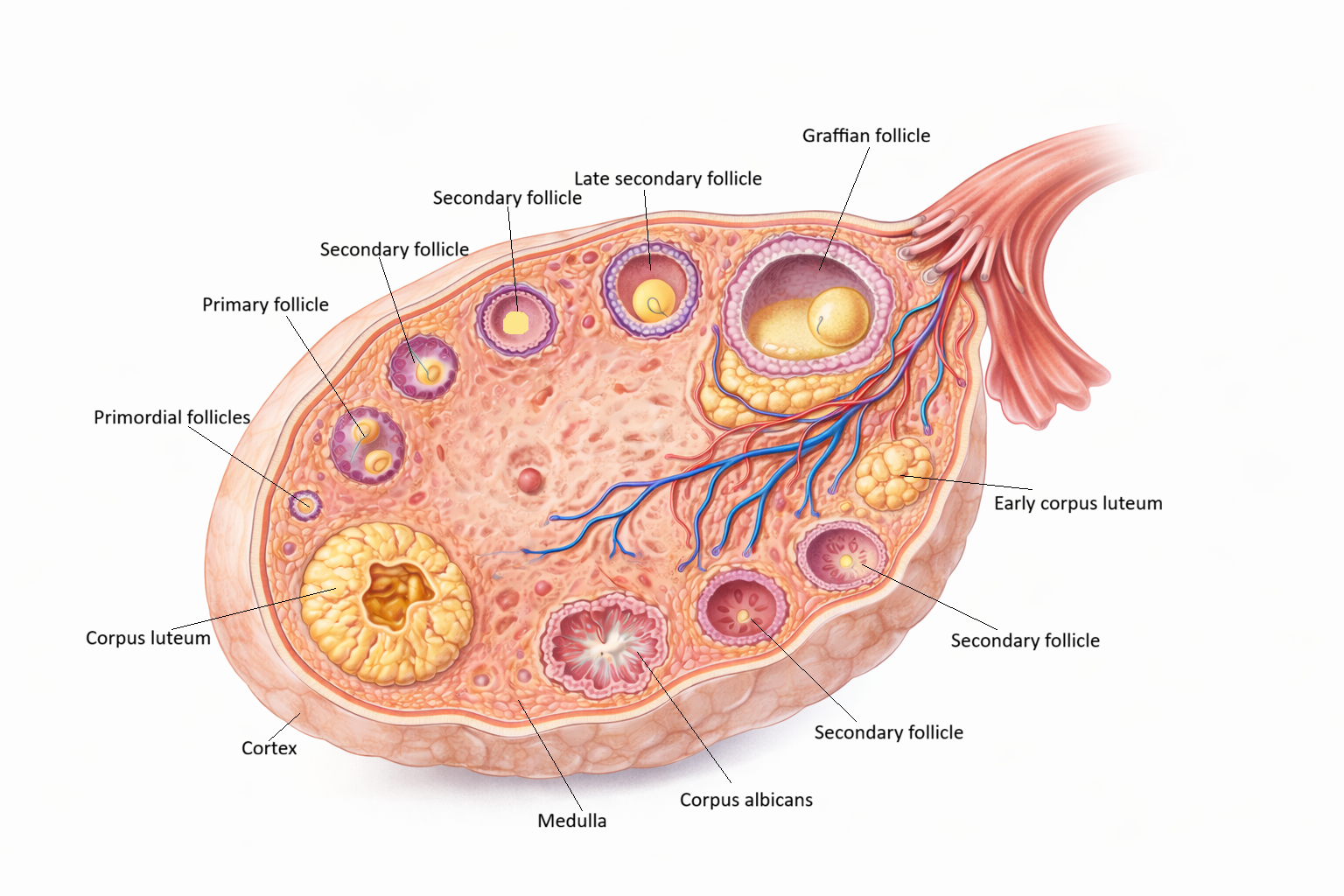
The ovaries are paired endocrine and reproductive organs with a highly organised internal structure that supports both gamete development and hormone production. Each ovary is divided into an outer cortex, where ovarian follicles are located, and an inner medulla, which contains blood vessels, lymphatics and nerves that supply and regulate ovarian function. The cortex is the functional site of folliculogenesis, while the medulla provides the vascular and neural support required for cyclical ovarian activity.
Females are born with a finite number of primordial follicles, established during fetal life, each containing an immature oocyte arrested in early meiosis. From puberty onwards, small groups of these follicles are recruited during each menstrual cycle under the influence of follicle-stimulating hormone (FSH). Although multiple follicles begin to develop, typically only one achieves dominance and progresses to ovulation, while the others undergo programmed degeneration known as atresia. This selective process ensures the release of a single, developmentally competent oocyte.
Follicular development is a stepwise and tightly regulated process that involves structural changes within the follicle and increasing hormonal activity. As follicles mature, they transition through several recognised stages, each associated with specific cellular features and endocrine function. This progression allows the ovary to synchronise oocyte maturation with cyclical hormone secretion, which in turn regulates the menstrual cycle and prepares the reproductive tract for potential fertilisation. Key stages of folliculogenesis include:
Primordial follicles, which contain a small, immature oocyte surrounded by a single layer of flattened support cells
Primary and secondary follicles, characterised by growth of the oocyte, proliferation of granulosa cells and formation of the theca cell layers
Antral and pre-ovulatory follicles, where fluid accumulation, increased oestrogen production and responsiveness to luteinising hormone support final oocyte maturation and ovulation
Together, ovarian structure and folliculogenesis underpin both reproductive capacity and endocrine balance, linking microscopic cellular events within the ovary to whole-body hormonal rhythms.
Image: The ovary is organised into an outer cortex and inner medulla, with follicles at various stages of development located in the cortex. Primordial follicles mature through primary and secondary stages to form a Graafian follicle, which contains a fluid-filled antrum and releases the oocyte at ovulation. Following ovulation, the follicle transforms into the corpus luteum, which later regresses into the fibrous corpus albicans.
Beyond the Basics
Ovarian Architecture and the Follicular Microenvironment
The ovarian cortex forms a dense, collagen-rich region that houses the finite reserve of primordial follicles within a supportive stromal framework. This stromal matrix is not passive scaffolding. It provides mechanical support, local signalling molecules, and a biochemical environment that influences whether follicles remain dormant or enter growth. Across the lifespan, the primordial follicle pool steadily declines through atresia, which underpins age-related reductions in fertility and culminates in menopause when the reserve is exhausted.
In contrast, the medulla is highly vascular and metabolically active. Its dense network of blood vessels, lymphatics and nerves supports follicular growth by ensuring delivery of oxygen, nutrients, and endocrine signals. Although follicles themselves reside in the cortex, the close anatomical relationship between cortex and medulla allows circulating gonadotrophins and locally produced hormones to act efficiently on developing follicles. This spatial organisation is essential for synchronising follicular maturation with systemic hormonal rhythms.
The ovarian stroma also has endocrine functions that directly shape folliculogenesis. Stromal and granulosa cells produce anti-Müllerian hormone, a regulator of early follicle recruitment that reflects the size of the remaining follicular pool. By limiting excessive activation of primordial follicles, anti-Müllerian hormone helps preserve reproductive lifespan. The ovarian surface epithelium, historically termed the germinal epithelium, plays no role in oocyte formation but is clinically significant as the origin of most epithelial ovarian malignancies.
Stages of Folliculogenesis and Hormonal Control
Primordial follicles contain a primary oocyte arrested in prophase I of meiosis, a pause that can last for decades. Each oocyte is surrounded by a single layer of flattened granulosa cells that maintain meiotic arrest and cellular viability. When a follicle is recruited into growth, granulosa cells become cuboidal and proliferate, marking the transition to a primary follicle and initiating increased metabolic and hormonal activity.
As development continues, granulosa cells form multiple layers and surrounding stromal cells differentiate into the theca interna and theca externa. This secondary follicle stage marks the beginning of meaningful steroid hormone production. The appearance of a fluid-filled antrum signals the antral stage, representing a major shift in follicular physiology. At this point, follicles become increasingly dependent on follicle-stimulating hormone, and subtle differences in hormone sensitivity determine which follicle will continue to grow.
One follicle usually becomes dominant due to greater responsiveness to follicle-stimulating hormone and more efficient estrogen production. This dominant follicle suppresses further development of its competitors through endocrine feedback, leading to atresia of non-dominant follicles. Rapid growth of the dominant follicle culminates in the pre-ovulatory or Graafian stage. A surge in luteinising hormone then triggers ovulation and resumption of meiosis, preparing the oocyte for potential fertilisation.
The Two-Cell Two-Gonadotrophin Model
Steroid hormone production within the ovarian follicle depends on coordinated interactions between theca and granulosa cells, each responding to different pituitary signals. Theca interna cells express receptors for luteinising hormone and convert cholesterol into androgen precursors. These androgens cannot be converted into estrogen within the theca cells themselves and must diffuse into adjacent granulosa cells.
Granulosa cells express receptors for follicle-stimulating hormone and contain aromatase, the enzyme responsible for converting androgens into estrogens. As the follicle matures, aromatase activity increases, leading to rising estrogen levels. Estrogen exerts feedback effects on the hypothalamic–pituitary axis, initially suppressing follicle-stimulating hormone to limit further follicle recruitment and later contributing to the luteinising hormone surge that triggers ovulation. This division of labour between cell types allows precise regulation of hormone synthesis across the menstrual cycle.
The Oocyte and Its Supporting Structures
The oocyte is surrounded by the zona pellucida, a specialised glycoprotein layer that provides structural protection and mediates sperm binding during fertilisation. Outside the zona pellucida, cumulus cells form a tightly organised cluster that maintains direct communication with the oocyte through gap junctions. These connections allow transfer of nutrients, ions, and signalling molecules that support meiotic competence and cytoplasmic maturation.
The integrity of the zona pellucida, cumulus cell function, and granulosa cell support collectively determines the developmental potential of the oocyte. Disruption at any level can impair fertilisation or early embryonic development, highlighting how successful reproduction depends not only on the oocyte itself but on the specialised microenvironment created by the surrounding follicle.
Clinical Connections
Disruption of normal folliculogenesis alters ovulatory function and menstrual regularity, with downstream effects on fertility and endocrine balance. When follicles fail to mature appropriately or ovulation does not occur, cycles may become irregular or absent, and estrogen and progesterone secretion becomes dysregulated. These hormonal changes often present clinically with altered bleeding patterns, subfertility, or features of hyperandrogenism, depending on the underlying mechanism.
Several common clinical conditions are directly linked to disordered follicular development and hormone production. Polycystic ovary syndrome is characterised by arrested follicular maturation, excess androgen synthesis, and chronic anovulation, resulting in multiple small antral follicles that do not progress to ovulation. In contrast, ovarian reserve declines gradually with age as the primordial follicle pool is depleted, but this process may be accelerated in premature ovarian insufficiency, where follicular loss occurs well before the expected age of menopause. Assessment of ovarian reserve relies in part on anti-Müllerian hormone, produced by granulosa cells of growing follicles, which reflects the size of the remaining follicular pool and helps guide diagnostic and management decisions. Clinically relevant consequences of impaired folliculogenesis include:
Anovulation or irregular ovulation, leading to unpredictable menstrual cycles and reduced fertility
Altered androgen and estrogen balance, contributing to symptoms such as acne, hirsutism, or abnormal uterine bleeding
Reduced ovarian reserve, associated with diminished response to ovarian stimulation and earlier reproductive ageing
Follicular physiology is also central to assisted reproductive technologies, which deliberately manipulate the normal selection of a single dominant follicle. Controlled ovarian stimulation uses exogenous gonadotrophins to promote the growth of multiple follicles within one cycle, increasing the number of mature oocytes available for retrieval. Detailed understanding of follicular dynamics is essential for timing ovulation triggers, interpreting hormone levels, and minimising the risk of ovarian hyperstimulation syndrome, a potentially serious complication of treatment.
External factors that damage ovarian tissue can have lasting effects on follicular number and function. Chemotherapy, radiation therapy, and some autoimmune disorders accelerate follicular loss by directly injuring oocytes or their supporting cells. Recognition of these risks underpins fertility preservation strategies such as oocyte or ovarian tissue cryopreservation prior to treatment. More broadly, knowledge of follicular structure and endocrine regulation informs clinical assessment of ovarian masses, menstrual disturbances, and endocrine abnormalities, linking cellular-level ovarian changes to real-world reproductive and hormonal presentations.
Concept Check
How does the structure of the ovarian cortex support long-term storage of primordial follicles?
What hormonal and cellular changes allow one follicle to become dominant during each menstrual cycle?
How do granulosa and theca cells cooperate to produce estrogen?
What features distinguish antral follicles from earlier developmental stages?
Why is AMH considered an important indicator of ovarian reserve?