Acute Respiratory Distress Syndrome (ARDS)
Acute respiratory distress syndrome (ARDS) is a form of acute, non-cardiogenic respiratory failure characterised by widespread inflammation within the lungs and severe impairment of gas exchange. It develops in response to a range of direct or indirect insults and is associated with high morbidity and mortality. ARDS is life-threatening, has a rapid onset, and is caused by illnesses, such as pneumonia, aspiration, trauma, and fat embolism, that trigger an inflammatory cascade, leading to alveolar-capillary damage, fibrotic alveolar ducts and severe hypoxaemia.
What you need to know
ARDS occurs when an initial insult, such as pneumonia, sepsis, aspiration, or major trauma, triggers a widespread inflammatory response within the lungs. This inflammation disrupts the alveolar–capillary membrane, which normally acts as a barrier between air in the alveoli and blood in the pulmonary capillaries. As this barrier becomes more permeable, fluid leaks into the alveolar spaces, leading to pulmonary oedema that is not caused by heart failure.
As fluid accumulates within the alveoli, gas exchange becomes severely impaired. Oxygen is unable to diffuse effectively into the bloodstream, resulting in hypoxaemia that is often resistant to standard oxygen therapy. At the same time, surfactant production is reduced (due to destruction of Type II epethelial cells, the cells within the alveoli that produce surfactant), which increases alveolar surface tension and contributes to alveolar collapse (atelectasis), further worsening oxygenation.
Common features that raise suspicion for ARDS include:
Rapid onset of severe dyspnoea
Marked hypoxaemia, often requiring high levels of oxygen support
Bilateral infiltrates on chest imaging
Reduced lung compliance, making ventilation more difficult
ARDS is a clinical syndrome (a collection of symptoms and clinical findings), rather than a single disease, and severity can range from moderate impairment to life-threatening respiratory failure requiring mechanical ventilation. Due to treatment often requiring high levels of oxygen, lung-protective mechanical ventilation, and in severe cases Extracorporeal Membrane Oxygenation (ECMO), ARDS is typically treated in critical care areas, such as intensive care.
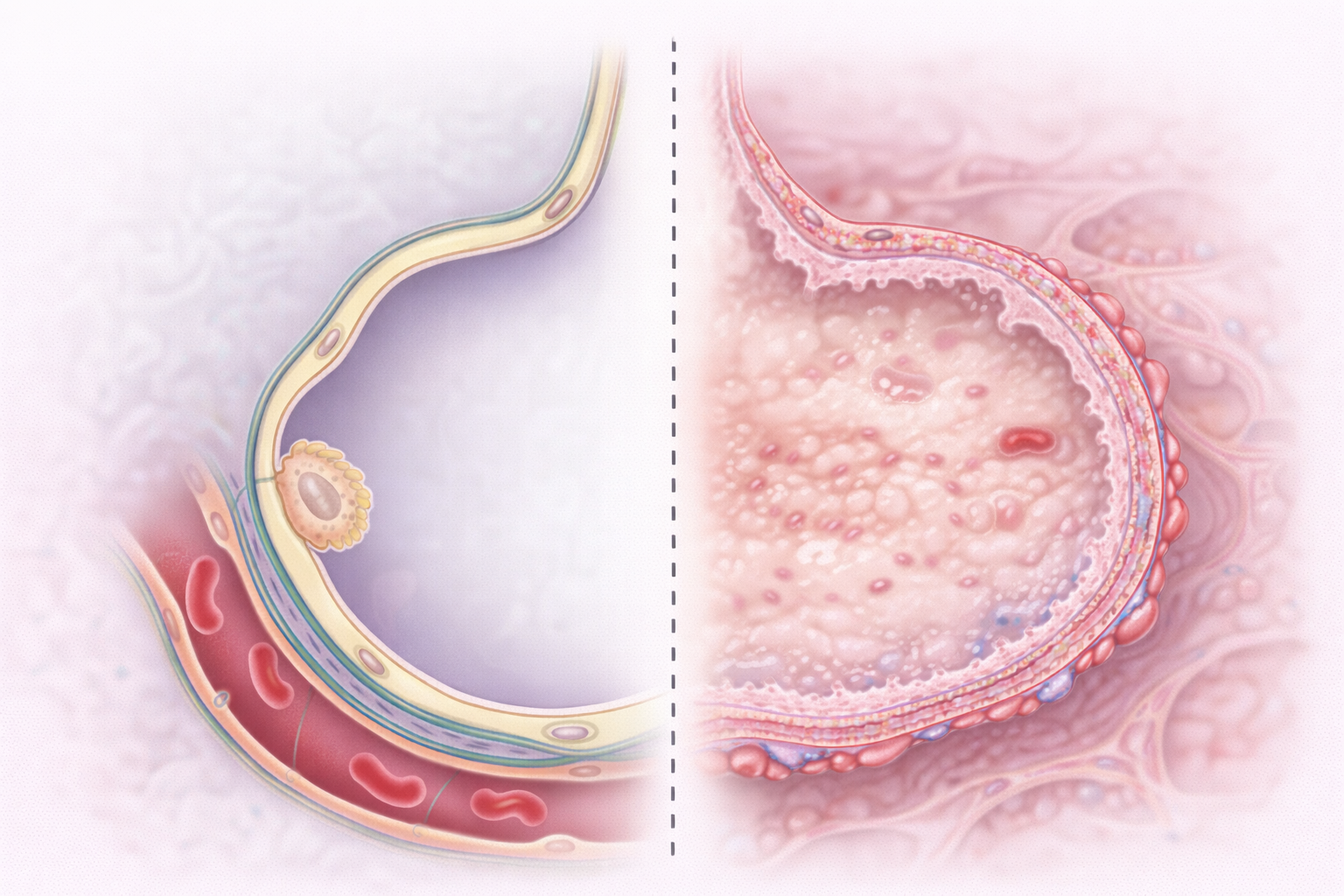
Image: Left: normal alveolus with a thin alveolar–capillary membrane and open air space for gas exchange.
Right: ARDS, where fluid fills the alveolus and the wall is thickened with hyaline membrane formation, impairing oxygen diffusion.
Beyond the basics
Diffuse alveolar damage
Diffuse alveolar damage is the hallmark pathological process in ARDS. Injury to both the alveolar epithelium and the capillary endothelium disrupts the normally tight barrier that separates air from blood. This increases permeability, allowing protein-rich fluid to leak into the alveolar spaces. As fluid accumulates, it dilutes surfactant and physically occupies space needed for gas exchange. In addition, cellular debris and plasma proteins form hyaline membranes, a layer of proteinaceous material lining the alveoli that further impairs oxygen diffusion. The combined effect is a loss of effective gas exchange across the alveolar–capillary interface (the thin barrier where oxygen and carbon dioxide are exchanged), which is central to the profound hypoxaemia seen in ARDS.
Surfactant dysfunction and alveolar collapse
Type II pneumocytes, responsible for producing surfactant, are damaged by inflammation and injury. Surfactant reduces surface tension within the alveoli, preventing them from collapsing, particularly at end-expiration. When surfactant production is reduced or the surfactant becomes dysfunctional, alveoli become unstable and prone to collapse, a process known as atelectasis (collapse of lung units). This leads to fewer open alveoli available for gas exchange and contributes to uneven ventilation. As more alveoli collapse, the work of breathing increases, as greater effort is required to reopen and ventilate stiff, non-compliant lungs.
Ventilation–perfusion mismatch and shunt
ARDS is characterised by severe ventilation–perfusion (V/Q) mismatch, where the balance between airflow (ventilation) and blood flow (perfusion) is disrupted. Many alveoli remain well perfused but are poorly ventilated due to fluid filling or collapse. This results in shunt physiology, where blood passes through the lungs without participating in gas exchange. Because this blood is not exposed to oxygen, it returns to the systemic circulation in a deoxygenated state. This explains why hypoxaemia in ARDS is often refractory to supplemental oxygen, as increasing inspired oxygen cannot correct areas where ventilation is absent.
Phases of ARDS
ARDS progresses through overlapping phases rather than clearly separated stages, and not all patients move through each phase in the same way. The degree of injury, the underlying cause, and the timeliness of treatment all influence how this process unfolds.
Exudative phase The exudative phase occurs in the first few days following the initial insult. It is characterised by intense inflammation and increased permeability of the alveolar–capillary barrier. As this barrier becomes disrupted, protein-rich fluid leaks into the interstitium (the space between cells) and the alveoli, leading to pulmonary oedema. Neutrophils migrate into the lungs and release inflammatory mediators, including cytokines and reactive oxygen species, which further damage the alveolar epithelium and capillary endothelium.
Hyaline membranes begin to form during this phase, consisting of fibrin (a protein involved in clotting) and cellular debris lining the alveolar walls. These membranes interfere with oxygen diffusion and contribute to reduced lung compliance (stiff lungs that are harder to inflate). Clinically, this phase is associated with rapidly worsening hypoxaemia and increased work of breathing.
Proliferative phase The proliferative phase typically develops after the first week and reflects an attempt at lung repair. During this phase, type II pneumocytes proliferate to restore the damaged alveolar lining and resume surfactant production. Fibroblasts (cells involved in tissue repair) begin to deposit collagen and extracellular matrix within the interstitium, which helps to stabilise the damaged lung structure.
There is often partial resolution of oedema as fluid is reabsorbed, and some improvement in gas exchange may occur. However, the ongoing presence of inflammation and early fibrosis can still impair lung function. Lung compliance may remain reduced, and patients often continue to require respiratory support during this phase.
Fibrotic phase Not all patients progress to the fibrotic phase, but when it occurs, it is associated with more severe and prolonged disease. This phase is characterised by extensive deposition of fibrous tissue within the interstitium and alveolar spaces, leading to architectural distortion of the lung. The normal thin, elastic alveolar walls become thickened and rigid, significantly impairing gas exchange.
This results in persistently reduced lung compliance and increased work of breathing. In severe cases, the fibrotic changes can be irreversible, leading to long-term respiratory impairment. Patients in this phase may have prolonged dependence on mechanical ventilation and a higher risk of poor outcomes.
Clinical connections
ARDS presents as rapidly worsening respiratory failure in the context of a known precipitating event. Patients often develop increasing dyspnoea, tachypnoea, and hypoxaemia over hours to days. Oxygen requirements escalate quickly, and many patients require mechanical ventilation to maintain adequate oxygenation.
On examination and investigation, findings are consistent with diffuse lung involvement rather than focal disease. Imaging typically shows bilateral infiltrates, and there is no evidence of left-sided heart failure to explain the pulmonary oedema. Lung compliance is reduced, making ventilation more challenging and increasing the risk of ventilator-associated injury if not managed carefully.
Features commonly seen in clinical practice include:
Severe hypoxaemia despite supplemental oxygen
Increasing respiratory distress and fatigue
Bilateral opacities on chest imaging
Reduced lung compliance requiring ventilatory support
Management focuses on treating the underlying cause while providing supportive care. This includes lung-protective ventilation strategies to minimise further injury, careful fluid management to reduce pulmonary oedema, and appropriate treatment of the precipitating condition such as infection or sepsis. ARDS requires close monitoring and escalation of care, often in an intensive care setting.
Concept check
What structural change in the lung is central to the development of ARDS?
Why is hypoxaemia in ARDS often resistant to supplemental oxygen?
How does surfactant dysfunction contribute to disease severity?
What is meant by shunt physiology in ARDS?
Why is lung-protective ventilation important in managing ARDS?