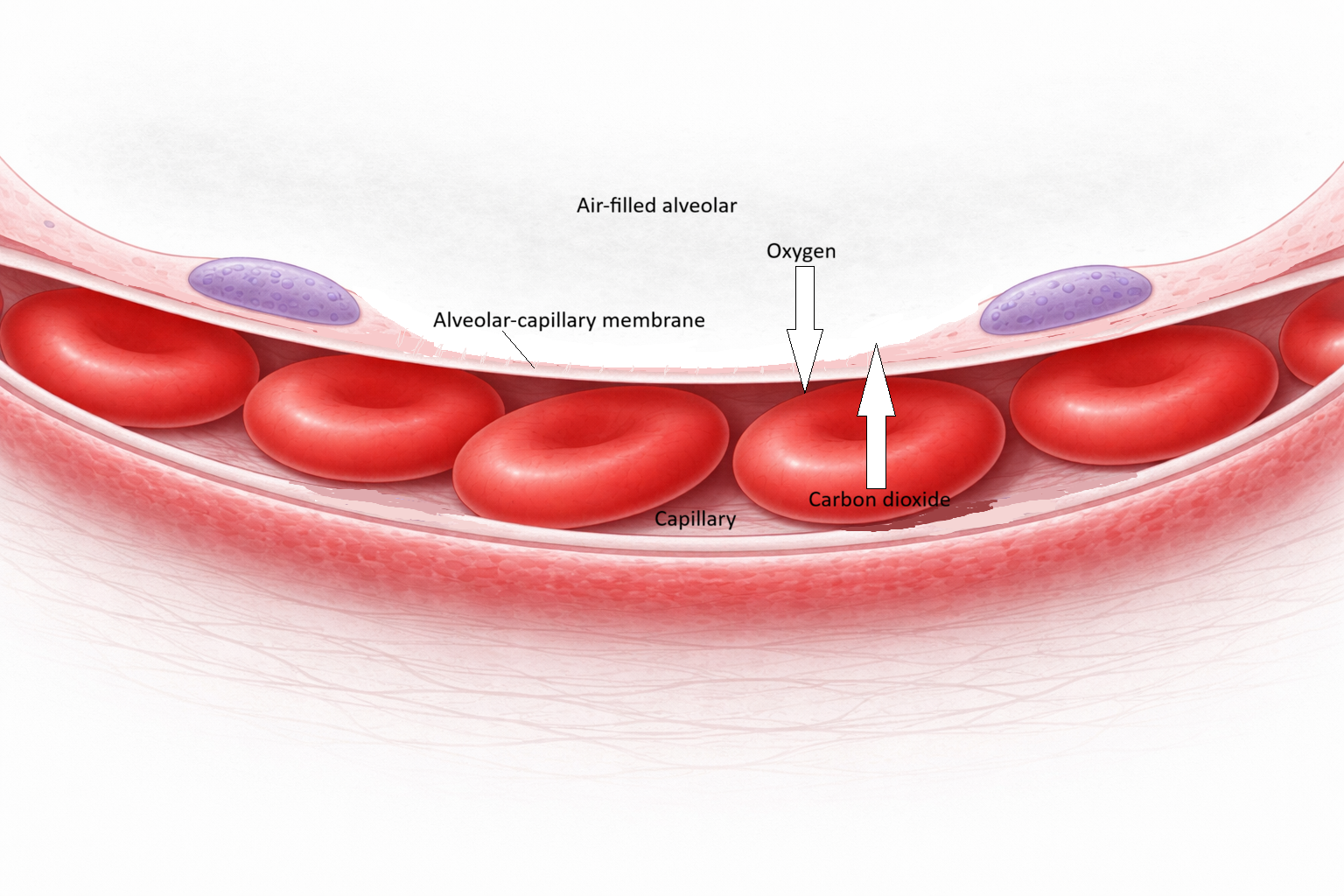
Gas Exchange at the Alveolar–Capillary Membrane
Gas exchange is the core purpose of the respiratory system. Every breath delivers oxygen to the alveoli, and every heartbeat brings deoxygenated blood to the pulmonary capillaries. The interface between these two systems, the alveolar–capillary membrane, is remarkably thin, highly specialised, and perfectly engineered for rapid diffusion. When functioning normally, the entire process is so efficient that blood becomes fully oxygenated in a fraction of a second. When impaired, oxygenation can collapse quickly, making the physiology of gas exchange essential knowledge for nurses in all clinical environments.
What You Need to Know
The alveolar–capillary membrane is the critical interface where oxygen enters the blood and carbon dioxide leaves it. It consists of three ultra-thin layers:
the alveolar epithelium (mainly Type I pneumocytes, specialised for diffusion)
a shared basement membrane
the capillary endothelium
Together these layers form a barrier less than one micrometre thick, allowing gases to move rapidly between air and blood.
Gas movement occurs by diffusion, meaning molecules move from areas of higher partial pressure (concentration) to lower partial pressure. In the lungs, oxygen moves from alveolar air into pulmonary capillary blood, while carbon dioxide moves from blood into the alveoli for exhalation.
Several factors determine how effectively this diffusion occurs:
Partial pressure gradients – the bigger the difference between alveoli and blood, the faster diffusion occurs
Membrane thickness – thicker membranes slow diffusion
Surface area – more alveoli and capillaries allow more gas exchange
Ventilation–perfusion matching – air flow (ventilation) must be matched to blood flow (perfusion) for efficient exchange
Under normal conditions, deoxygenated blood entering the lungs has a PO₂ of about 40 mmHg, while alveolar air has a PO₂ of about 104 mmHg. This steep gradient (the difference between the higher pressure and the lower pressure) causes oxygen to move quickly into the blood until the values nearly equalise. At the same time, carbon dioxide moves from the blood (≈46 mmHg) into the alveoli (≈40 mmHg). Even though this gradient is smaller, carbon dioxide diffuses easily because it is far more soluble than oxygen.
Once oxygen crosses the membrane, it binds to haemoglobin inside red blood cells. This binding keeps blood oxygen levels low, maintaining the gradient and allowing more oxygen to continue diffusing into the bloodstream. In this way, haemoglobin acts as a sink that pulls oxygen across the membrane and supports continuous gas exchange.
Efficient alveolar–capillary exchange is therefore essential for maintaining normal blood oxygen levels and removing metabolic carbon dioxide from the body.
Image: The alveolar–capillary membrane forms an ultra-thin (membrane thickness exaggerated in image) interface between air and blood, composed of flattened Type I pneumocytes, a fused basement membrane, and capillary endothelium.
Beyond the Basics
The Respiratory Membrane: A Highly Specialised Interface
Gas exchange occurs across the respiratory membrane, a structure optimised to minimise diffusion distance while maximising surface area. Type I pneumocytes form approximately 95% of the alveolar surface and are among the thinnest cells in the body. Their flattened morphology creates a thin barrier between alveolar gas and capillary blood, allowing rapid diffusion of oxygen and carbon dioxide.
Type II pneumocytes play a critical stabilising role by secreting surfactant, which reduces surface tension within the alveoli. By preventing alveolar collapse, surfactant preserves the integrity and surface area of the gas-exchange interface. Loss or dysfunction of Type II cells therefore compromises both ventilation and diffusion efficiency.
Pulmonary capillaries are tightly wrapped around alveoli and are so narrow that red blood cells must pass through in single file, often deforming to fit the capillary lumen. This close apposition ensures maximal contact between erythrocytes and the respiratory membrane. Under normal conditions, haemoglobin becomes fully saturated with oxygen within approximately 0.25 seconds, despite total capillary transit time being closer to 0.75 seconds. This physiological reserve allows adequate oxygenation even during increased cardiac output, such as exercise.
Partial Pressure Gradients and Diffusion
Gas exchange across the respiratory membrane depends entirely on partial pressure gradients, not on the absolute amount of gas present. Oxygen diffuses from areas of higher partial pressure in the alveoli to lower partial pressure in pulmonary capillary blood, while carbon dioxide diffuses in the opposite direction.
The magnitude of this gradient directly determines the rate of diffusion. At altitude, reduced atmospheric pressure lowers alveolar partial pressure of oxygen, decreasing the gradient driving diffusion and impairing oxygen uptake. Similarly, conditions that reduce alveolar ventilation or replace air with fluid or collapse reduce alveolar PO₂, limiting diffusion even when pulmonary blood flow remains intact.
This explains why oxygenation may be severely impaired in diseases such as pneumonia, atelectasis, or advanced COPD, despite normal cardiac output and preserved haemoglobin levels.
Ventilation–Perfusion Matching (V/Q Ratios)
Efficient gas exchange requires an appropriate match between ventilation and perfusion at the alveolar level. The ventilation–perfusion (V/Q) ratio describes this relationship and is a key determinant of arterial oxygenation.
In regions with high V/Q ratios, ventilation is preserved but perfusion is reduced or absent. These alveoli contribute to physiological dead space, as oxygen cannot enter the bloodstream despite adequate airflow. Pulmonary embolism is a classic example, where obstructed blood flow prevents gas exchange.
In contrast, low V/Q ratios occur when perfusion exceeds ventilation. Blood passes through poorly ventilated or non-ventilated alveoli, resulting in inadequate oxygen uptake. This shunt-like physiology is seen in pneumonia, airway obstruction, and alveolar collapse. Importantly, true shunt physiology does not respond effectively to supplemental oxygen if alveoli remain unventilated, making it particularly dangerous.
Carbon Dioxide Exchange and Ventilatory Failure
Carbon dioxide diffuses across the respiratory membrane far more readily than oxygen due to its higher solubility. As a result, CO₂ elimination is usually preserved even when oxygen diffusion is significantly impaired.
This explains why early respiratory failure often presents with hypoxaemia without hypercapnia. As disease progresses and ventilation becomes inadequate, due to respiratory muscle fatigue, reduced compliance, or airway obstruction, carbon dioxide clearance eventually fails, leading to hypercapnia and respiratory acidosis.
The dissociation between oxygen and carbon dioxide exchange highlights the importance of assessing both oxygenation and ventilation when evaluating gas-exchange disorders.
Clinical Connections
Changes to the alveolar–capillary membrane rarely occur in isolation. In clinical practice, alterations in thickness, surface area, or perfusion are often accompanied by changes in ventilation and blood flow, compounding their impact on gas exchange.
Key patterns to recognise:
Increased membrane thickness → slower diffusion
Reduced surface area → less gas exchange capacity
Impaired perfusion → wasted ventilation (dead space)
Impaired ventilation → perfused but non-functional alveoli (shunt)
Pneumonia
Inflammation fills the alveoli with fluid and cellular debris, increasing the distance between air and blood while also reducing ventilation. Blood continues to perfuse these poorly ventilated regions, creating a low V/Q state (physiological shunt). As a result, oxygen cannot effectively reach the bloodstream, and hypoxaemia may persist despite supplemental oxygen. In more severe cases, positive pressure ventilation may be required to reopen collapsed or fluid-filled alveoli and restore surface area.
Pulmonary Oedema
Fluid initially accumulates in the interstitial space before spilling into the alveoli, progressively thickening the alveolar–capillary membrane and impairing diffusion. As this barrier expands, oxygen transfer becomes increasingly inefficient. Clinically, this presents with dyspnoea, crackles, and in more advanced cases, pink frothy sputum. The underlying cause is often cardiac, but similar mechanisms are seen in ARDS and fluid overload states.
COPD (Emphysema & Chronic Bronchitis)
In emphysema, destruction of alveolar walls leads to a loss of surface area, significantly reducing the capacity for gas exchange. Chronic bronchitis contributes by increasing mucus production and airway narrowing, limiting ventilation. Together, these changes produce V/Q mismatch and impair both diffusion and oxygen delivery, particularly during exertion.
Pulmonary Embolism
A thrombus obstructs pulmonary blood flow, meaning alveoli may be ventilated but not perfused. This creates high V/Q (dead space), where gas exchange cannot occur despite adequate ventilation. Although the membrane itself may remain structurally intact, the absence of blood flow renders it functionally ineffective, leading to sudden hypoxia and compensatory tachycardia.
Acute Respiratory Distress Syndrome (ARDS)
Diffuse inflammation damages both the alveolar epithelium and capillary endothelium, disrupting the integrity of the membrane. This results in marked thickening, fluid leakage, and surfactant loss, causing alveolar collapse and severely impaired diffusion. The combination of shunt and reduced compliance leads to refractory hypoxaemia, often requiring mechanical ventilation with high PEEP to maintain alveolar recruitment.
Anaemia
In anaemia, the membrane and gas exchange process may be entirely normal, but oxygen transport is reduced due to low haemoglobin levels. This highlights an important distinction: effective diffusion does not guarantee adequate oxygen delivery. Patients may appear well oxygenated on pulse oximetry, yet still experience tissue hypoxia due to reduced oxygen content.
Concept Check
Why is the alveolar–capillary membrane so thin, and how does this support diffusion?
What factors determine the rate of gas exchange?
Why does carbon dioxide diffuse more easily than oxygen?
Explain the difference between high V/Q and low V/Q mismatch with clinical examples.
Why can anaemic patients have normal oxygen saturations despite tissue hypoxia?