The Oxygen–Haemoglobin Dissociation Curve
Haemoglobin is not simply a passive carrier of oxygen; it is an adaptive, finely tuned molecule that adjusts its oxygen affinity based on the body’s metabolic needs. The oxygen–haemoglobin dissociation curve illustrates the relationship between the partial pressure of oxygen in the blood and the percentage of haemoglobin that is saturated. This curve explains why oxygen saturations remain relatively stable across a wide range of oxygen levels and why haemoglobin readily releases oxygen in tissues that need it most.
What You Need to Know
The oxygen–haemoglobin dissociation curve describes how tightly haemoglobin binds oxygen at different partial pressures of oxygen (PaO₂). Its characteristic S-shape (sigmoid curve) arises from cooperative binding, meaning that each oxygen molecule that attaches to haemoglobin makes it easier for the next one to bind. This property allows haemoglobin to load oxygen very efficiently in the lungs while still being able to release it readily in tissues where oxygen is needed.
In the lungs, where PaO₂ is high, haemoglobin becomes almost fully saturated. Even if alveolar oxygen fluctuates slightly, saturation remains high because the upper flat portion of the curve provides a safety buffer. This ensures reliable oxygen loading during normal breathing, mild illness, or changes in altitude.
In peripheral tissues, where oxygen is being consumed, PaO₂ is lower. Under these conditions haemoglobin’s affinity for oxygen falls, allowing oxygen to be released into metabolically active cells. This means that the same haemoglobin molecule behaves differently depending on the local oxygen environment, loading in the lungs and unloading in tissues.
The curve is shaped and shifted by several important physiological factors that influence oxygen delivery:
• pH (acidity) – lower pH (more acid) promotes oxygen release
• Carbon dioxide levels – higher CO₂ encourages oxygen unloading
• Temperature – warmer tissues release oxygen more easily
• 2,3-BPG (a red cell metabolite) – increases oxygen release during chronic hypoxia
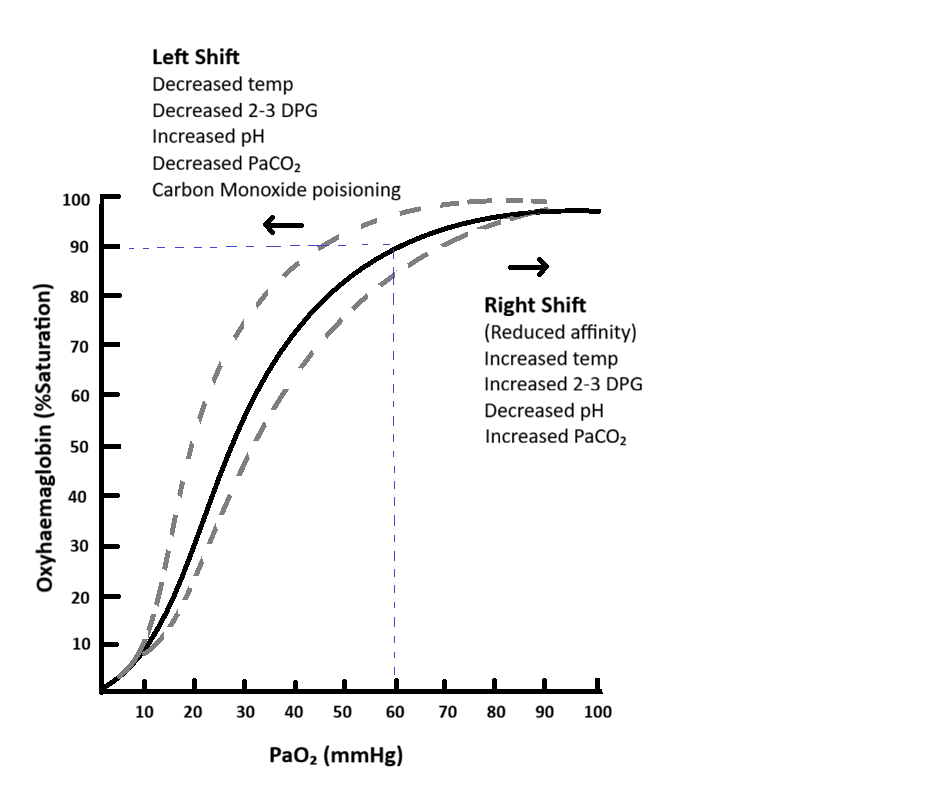
Image: The point where PaO₂ is ~60 mmHg and oxygen saturation is ~90% (blue dotted lines) marks the lower end of the curve’s plateau. Beyond this point, small decreases in PaO₂ lead to disproportionately large drops in saturation, making it a critical threshold in clinical practice.
These factors ensure that oxygen is preferentially delivered to tissues that are most metabolically active, such as exercising muscles or infected, inflamed organs.
A particularly important feature of the curve is its steep lower portion. When PaO₂ falls below about 60 mmHg, haemoglobin saturation drops rapidly. This means that once oxygen levels begin to decline into this range, even small further decreases produce large losses of oxygen bound to haemoglobin. Clinically, this explains why patients can appear stable at moderate hypoxia and then deteriorate very quickly once this threshold is crossed.
This steep section is why oxygen saturation is such a sensitive indicator of respiratory failure and why timely oxygen therapy is critical when PaO₂ begins to fall.
Beyond the Basics
Shifts of the Oxygen–Haemoglobin Dissociation Curve
The oxygen–haemoglobin dissociation curve is not fixed; it shifts left or right in response to changes in the internal environment. These shifts reflect alterations in haemoglobin’s affinity for oxygen and play a critical role in matching oxygen delivery to tissue metabolic demand.
A rightward shift of the curve indicates reduced affinity between haemoglobin and oxygen. In this state, oxygen is released more readily into the tissues. This shift occurs when carbon dioxide levels rise, pH falls, temperature increases, or concentrations of 2,3-bisphosphoglycerate (2,3-BPG) increase. Collectively, these factors characterise metabolically active tissues, where oxygen demand is greatest. This phenomenon, known as the Bohr effect, ensures that oxygen unloading is enhanced precisely where it is most needed.
In systemic illness such as sepsis, fever and metabolic acidosis promote a rightward shift. Although arterial oxygen levels may be reduced, tissue oxygen delivery can be preserved through improved unloading, highlighting the adaptive nature of this physiological response.
Leftward Shifts and Impaired Oxygen Delivery
A leftward shift of the dissociation curve results from increased haemoglobin affinity for oxygen, making oxygen binding stronger and release to tissues more difficult. This occurs in conditions associated with reduced metabolic demand or altered haemoglobin structure.
Common causes of a leftward shift include:
Decreased temperature
Reduced carbon dioxide levels
Alkalosis
Foetal haemoglobin
Carbon monoxide binding to haemoglobin
In these situations, oxygen saturation may appear normal or even elevated, yet tissue hypoxia can develop because haemoglobin holds onto oxygen too tightly. Hypothermic patients and those with carbon monoxide poisoning are examples of this disconnect between oxygen saturation and effective tissue oxygen delivery.
The Protective Role of the Plateau Region
The upper, relatively flat portion of the oxygen–haemoglobin dissociation curve provides an important physiological safety margin. Within this range, large changes in alveolar partial pressure of oxygen result in only small changes in haemoglobin saturation.
This plateau protects oxygen loading in the lungs during periods of moderate hypoxia, such as during sleep, at altitude, or with mild airway obstruction. As long as alveolar oxygen pressure remains within this range, haemoglobin remains highly saturated, preserving arterial oxygen content.
The Steep Portion and Tissue Oxygen Extraction
In contrast, the steep lower portion of the curve allows small decreases in oxygen partial pressure to produce large reductions in haemoglobin saturation. This design facilitates efficient oxygen unloading at the tissue level, particularly during increased metabolic activity.
As tissues extract more oxygen and local partial pressure falls, haemoglobin rapidly releases oxygen, ensuring a responsive and flexible oxygen delivery system. Together, the plateau and steep regions of the curve balance stability in oxygen loading with adaptability in oxygen unloading.
Clinical Connections
Shifts in the oxygen–haemoglobin dissociation curve explain why oxygen saturation numbers do not always reflect how well tissues are actually being oxygenated. Pulse oximetry only measures how much oxygen is bound to haemoglobin, not how effectively that oxygen is being released into organs and cells.
In hypothermia, haemoglobin holds onto oxygen more tightly. A trauma patient who is cold may show a normal or even high oxygen saturation, yet their tissues are starved of oxygen because haemoglobin is not releasing it. This is one reason why active rewarming is essential in trauma and post-operative care, warming the patient shifts the curve back toward normal and improves oxygen delivery at the tissue level.
Carbon monoxide poisoning creates a particularly dangerous clinical illusion. Carbon monoxide binds to haemoglobin far more strongly than oxygen, occupying binding sites and preventing oxygen transport. Carbon monoxide has an affinity for haemoglobin roughly 200-250 times higher than oxyygen. Pulse oximeters cannot distinguish between oxyhaemoglobin and carboxyhaemoglobin, so readings may appear normal even while the patient is profoundly hypoxic. This is why patients with smoke inhalation, headache, dizziness, or confusion require arterial blood gas analysis or co-oximetry rather than reliance on SpO₂ alone.
Several common clinical states alter the curve and change how oxygen is delivered:
• Fever, acidosis, and high CO₂ shift the curve rightward, improving oxygen unloading to tissues
• Hypothermia, alkalosis, and low CO₂ shift it leftward, impairing oxygen release
• Chronic hypoxia (e.g., COPD or high altitude) increases 2,3-BPG, helping oxygen be released
These shifts explain why a febrile or acidotic patient may have slightly lower oxygen saturations but still be delivering oxygen effectively to tissues, while a cold or alkalotic patient may look well-oxygenated on the monitor but be physiologically hypoxic.
In chronic lung disease, a right-shifted curve is adaptive. Elevated CO₂ and lower pH help compensate for low arterial oxygen levels by making haemoglobin unload oxygen more easily. This is why some patients with COPD tolerate lower oxygen saturations than healthy individuals without showing tissue hypoxia.
Concept Check
Why does the dissociation curve become steep at low oxygen levels?
How does fever influence haemoglobin’s affinity for oxygen?
Why might a hypothermic patient have normal saturations yet poor tissue oxygenation?
Why is carbon monoxide poisoning difficult to detect with pulse oximetry?
How does a rightward shift support oxygen delivery in acutely ill patients?