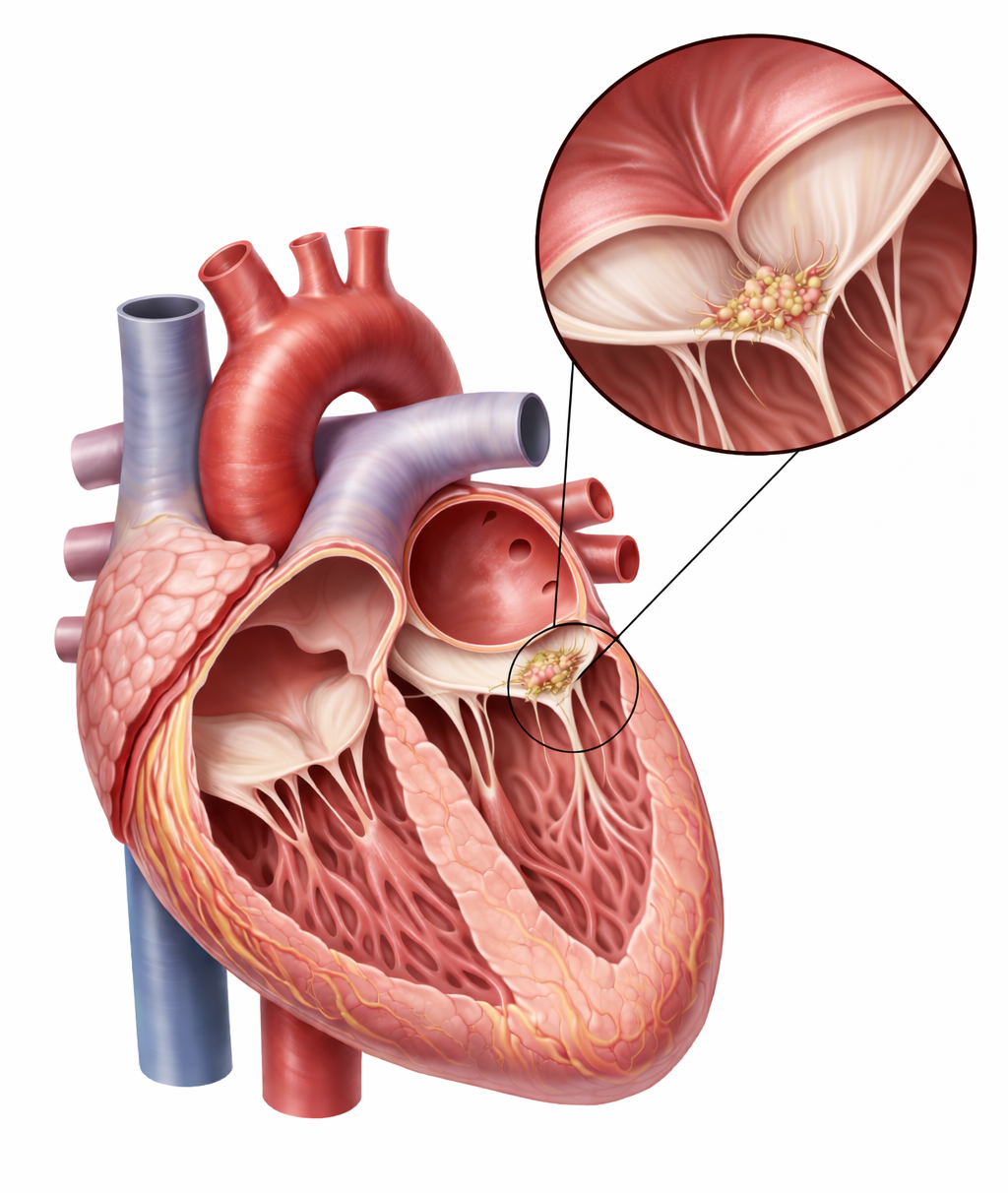
Infective Endocarditis: Infection of the Endocardium
Infective endocarditis is a microbial infection of the endocardium, most commonly affecting the cardiac valves. It develops when circulating microorganisms adhere to areas of endothelial disruption (damage to the thin membrane lining blood vessels) and become embedded within thrombotic material, where they multiply and form vegetations. Although relatively uncommon, it is a serious condition associated with significant morbidity and mortality, particularly when diagnosis or treatment is delayed.
What you need to know
Infective endocarditis develops when microorganisms within the bloodstream adhere to the endocardium, typically at sites of endothelial injury such as abnormal native valves, prosthetic valves, or regions exposed to turbulent flow. This leads to the formation of vegetations, which consist of microorganisms embedded within a matrix of platelets and fibrin (thrombotic material formed during clotting). These vegetations allow ongoing microbial proliferation and act as a source of persistent bacteraemia, driving both local cardiac damage and systemic illness.
As vegetations enlarge, they interfere with normal valvular function, most commonly causing regurgitation due to impaired leaflet coaptation (the leaflets don’t close properly). This creates a volume load on the affected chamber and can progress to haemodynamic compromise. In more severe cases, the infection can extend into surrounding cardiac structures, leading to complications such as perivalvular abscess formation or involvement of the cardiac conduction system, which may present as new conduction abnormalities.
Common features that raise suspicion for infective endocarditis include:
Persistent fever
New or changing cardiac murmur
Evidence of embolic phenomena, such as stroke or peripheral ischaemia
Constitutional symptoms including fatigue, weight loss, and night sweats
If untreated, infective endocarditis can lead to progressive valvular dysfunction, heart failure, systemic embolisation, and sepsis. Therefore, endocarditis is typically treated urgently and aggressively, with high-dose intravenous antibiotics for several weeks. Surgery may be required to remove or repair damaged valves and infarcted tissue.
Image: Mitral valve with a small, irregular vegetation attached along the line of closure, consistent with infective endocarditis.
Beyond the basics
Endothelial disruption and thrombus formation
Endothelial disruption is the key initiating step in infective endocarditis and is most often driven by abnormal flow patterns across diseased or prosthetic valves. Turbulent flow creates shear stress on the endocardial surface, leading to microscopic injury and exposure of underlying tissue. This promotes the deposition of platelets and fibrin, forming a sterile thrombus known as non-bacterial thrombotic endocarditis. This thrombus provides an ideal surface for microbial adhesion, particularly during transient episodes of bacteraemia that can occur with everyday activities such as dental procedures or minor mucosal trauma.
Microbial adherence and vegetation development
Once microorganisms reach the circulation, they can adhere to this thrombotic surface through specific adhesion molecules that bind to host matrix proteins. Following attachment, they proliferate within the fibrin-platelet matrix, forming vegetations that grow over time. The structure of these vegetations is clinically important, as it limits penetration of immune cells and reduces the effectiveness of antibiotics. This allows infection to persist even in the presence of an active immune response and contributes to the need for prolonged intravenous antimicrobial therapy.
Valvular destruction and haemodynamic consequences
As infection progresses, local tissue destruction becomes more significant. Valve leaflets may become thickened, perforated, or partially destroyed, leading to worsening regurgitation. In acute cases, particularly with more virulent organisms, this can result in sudden and severe haemodynamic compromise because the ventricle has not had time to adapt to the increased volume load. In subacute disease, these changes occur more gradually, allowing some degree of compensatory adaptation before decompensation occurs.
Embolisation and systemic spread
Vegetations are friable (fragile and easily damaged) and prone to fragmentation, and embolisation (fragements of the vegetation become mobile in the bloodstream) is a common complication. Fragments can dislodge and travel through the circulation, with the clinical consequences depending on the site of embolisation. Left-sided endocarditis is associated with systemic emboli, including stroke, renal infarction, and splenic infarction, while right-sided disease is more likely to cause septic pulmonary emboli. In addition to mechanical obstruction, these emboli may carry infection to distant sites, leading to metastatic infection.
Immunological complications
Persistent infection and ongoing antigen exposure can lead to immune complex formation. These circulating immune complexes may deposit in tissues such as the kidneys, contributing to glomerulonephritis, or in small vessels, producing vasculitic features. These processes are more commonly seen in subacute disease, where prolonged infection allows time for immune-mediated complications to develop.
Clinical connections
Infective endocarditis often presents with a combination of systemic symptoms and evolving cardiac findings. Fever is the most consistent feature and is often accompanied by fatigue, malaise, reduced exercise tolerance, and weight loss. A new or changing murmur may be detected as valve function deteriorates, particularly in left-sided disease where regurgitation becomes more pronounced.
Symptoms result from complications of the condition, rather than the primary infection itself. Acute valvular regurgitation may present with dyspnoea, pulmonary oedema, or features of heart failure, particularly if the onset is rapid. Embolic events (disruption of blood flow due to a detached, travelling clot) can present suddenly, with neurological deficits in the case of cerebral emboli, or with organ-specific symptoms such as flank pain or abdominal pain when the kidneys or spleen are involved. In right-sided endocarditis, respiratory symptoms such as pleuritic chest pain, cough, or haemoptysis may occur due to septic pulmonary emboli.
Findings that may be seen in clinical practice include:
Fever with a new or changing murmur
Signs of heart failure, particularly in acute valvular dysfunction
Focal neurological deficits suggestive of embolic stroke
Peripheral signs such as splinter haemorrhages, petechiae, or other microvascular phenomena
Diagnosis relies on a combination of blood cultures and echocardiography. Blood cultures are essential for identifying the causative organism and guiding antimicrobial therapy, while echocardiography allows visualisation of vegetations, assessment of valve integrity, and detection of complications such as abscess formation. Management involves prolonged intravenous antibiotic therapy, and surgical intervention is considered when there is severe valve dysfunction, uncontrolled infection, or recurrent embolisation.
Concept check
How does endothelial injury contribute to the development of infective endocarditis?
Why do vegetations allow infection to persist despite immune and antibiotic activity?
What mechanisms lead to acute haemodynamic deterioration in infective endocarditis?
Why does left-sided endocarditis commonly result in systemic emboli?
What clinical features indicate progression to complicated disease requiring surgical management?