Venous Thromboembolism (VTE): Deep Vein Thrombosis and Pulmonary Embolism
Venous thromboembolism (VTE) refers to the formation of blood clots within the venous system and includes both deep vein thrombosis (DVT) and pulmonary embolism (PE). These conditions are not separate diseases but different manifestations of the same pathological process. A clot that forms in the deep veins may remain localised or dislodge and travel to the pulmonary circulation. Understanding VTE as a continuum explains why prevention and early recognition are critical. The underlying failure is inappropriate clot formation within low-flow venous circulation.
What You Need to Know
Venous thromboembolism (VTE) refers to the formation of blood clots within the venous circulation and includes both deep vein thrombosis (DVT) and pulmonary embolism (PE). In the venous system, blood flows more slowly and under lower pressure than in arteries, making clot formation more likely when normal regulatory mechanisms are disrupted. When a thrombus forms in a deep vein, it may remain local and cause venous obstruction, or part of the clot may detach and travel to the pulmonary circulation, resulting in a pulmonary embolism.
Although DVT and PE are often discussed as separate conditions, they represent different manifestations of the same underlying process. A DVT primarily affects venous return, leading to local inflammation, increased venous pressure and impaired drainage from the affected limb. A PE occurs when embolised clot material lodges in the pulmonary arteries, abruptly reducing pulmonary blood flow and disrupting gas exchange. In both cases, the problem originates within the venous circulation rather than from primary cardiac or pulmonary pathology.
VTE develops when multiple physiological controls that normally prevent inappropriate clotting fail at the same time. Three broad factors are consistently involved:
Reduced or altered venous blood flow, such as immobility or venous compression
Injury or dysfunction of the vascular endothelium (the inner lining of blood vessels)
A shift toward increased coagulation activity, where clot formation outweighs clot breakdown
These factors rarely act in isolation. Instead, they interact and amplify one another, which explains why VTE is most commonly seen during periods of acute illness, surgery, trauma, pregnancy or prolonged immobility. The cumulative burden of risk factors is more important than any single trigger, showing that VTE usually develops from a combination of factors affecting blood flow and clotting, rather than just one problem.
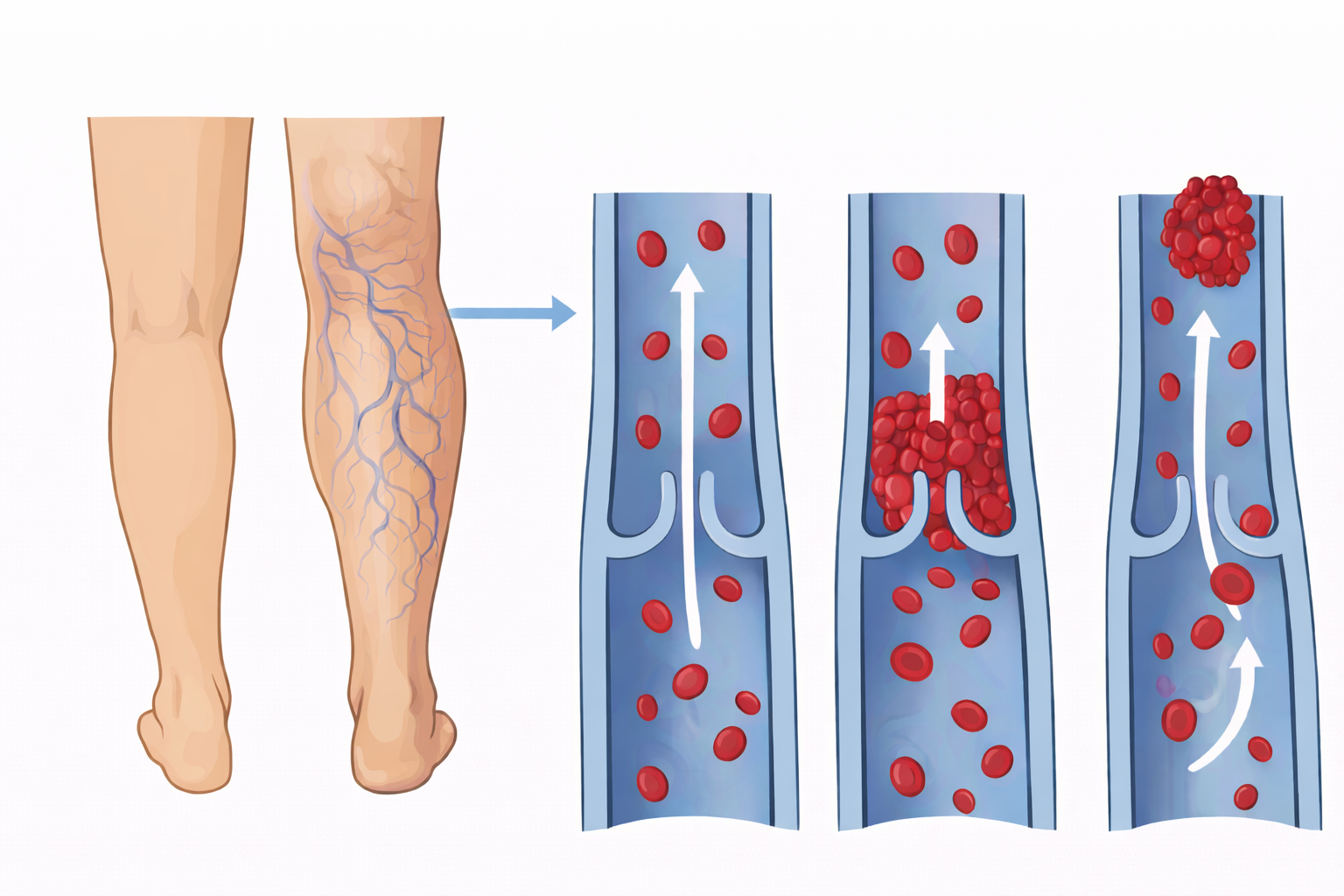
Image: Deep vein thrombosis (DVT) occurs when a clot forms within a deep vein, typically around valve sites where blood flow is slower. This disrupts normal venous return and can lead to swelling in the affected limb. If part of the clot breaks off, it can travel through the circulation as an embolus, potentially causing life-threatening complications such as pulmonary embolism.
Beyond the Basics
Venous Stasis and Reduced Blood Flow
Venous return depends on skeletal muscle contraction and normal pressure gradients to keep blood moving back toward the heart. When movement is limited, such as during prolonged bed rest or immobility, this muscle pump becomes less effective, causing blood flow to slow and pool in the deep veins, particularly in the lower limbs. Slower flow reduces the dilution and clearance of activated clotting factors, allowing them to accumulate locally and create an environment that favours clot formation and supports early thrombus growth.
Endothelial Dysfunction and Localised Clot Formation
Under normal conditions, the endothelium (the inner lining of blood vessels) provides a smooth, antithrombotic surface that prevents clotting. When exposed to inflammation, mechanical pressure, hypoxia , or direct injury, this balance is disrupted and endothelial cells shift toward a procoagulant state. They begin expressing tissue factor, adhesion molecules, and inflammatory mediators, making the vessel wall more adhesive and promoting platelet attachment. This allows fibrin (the protein mesh that stabilises clots) to form and anchor the developing thrombus to the vessel wall.
Hypercoagulable States and Thrombus Propagation
Hypercoagulability refers to a state where the blood is more likely to clot than normal and may result from increased procoagulant factors, reduced anticoagulant activity, or impaired fibrinolysis (the breakdown of clots). Accelerated thrombin generation increases fibrin formation, strengthening and stabilising the clot. Once formed, venous thrombi tend to extend in the direction of blood flow, gradually increasing in size and raising the risk that part of the clot will break away and travel through the circulation.
Thrombus Organisation and Embolisation
Venous thrombi are typically rich in fibrin and red blood cells, making them softer and less firmly attached than arterial clots. Early thrombi are particularly unstable, and changes in venous pressure, limb movement, or blood flow can dislodge fragments. These fragments, known as emboli, travel through the venous circulation, pass through the right side of the heart, and lodge in the pulmonary arteries, marking progression from local venous thrombosis to pulmonary embolism.
Pulmonary Vascular Obstruction and Haemodynamic Consequences
When a thrombus lodges in the pulmonary circulation, it obstructs blood flow through affected vessels, causing an abrupt increase in pulmonary vascular resistance and increasing right ventricular afterload. Reduced pulmonary perfusion leads to ventilation–perfusion mismatch, where areas of the lung are ventilated but not perfused, impairing oxygen exchange. Large or multiple emboli can overwhelm right ventricular function, resulting in haemodynamic instability or cardiovascular collapse, with the clinical impact depending on clot burden, speed of obstruction, and the patient’s underlying cardiopulmonary reserve.
Clinical Connections
Deep Vein Thrombosis (DVT) often develops insidiously and may be asymptomatic. When present, symptoms are usually local and related to venous obstruction and inflammation, including unilateral limb swelling, pain, warmth and erythema. Clinical examination alone is unreliable, so suspected DVT is typically investigated using compression ultrasonography, with D-dimer testing (a blood test that measures protein fragments produced when a blood clot dissolves) used to support exclusion (not used to confirm a clot) in low-risk patients. Many venous thrombi remain undetected until embolisation occurs, meaning the absence of limb symptoms does not exclude clinically significant disease.
Typical features include:
Unilateral limb swelling, usually in the calf or thigh
Localised pain, tenderness or cramping
Warmth and erythema over the affected area
Increased limb circumference or pitting oedema
The absence of pain or swelling does not exclude DVT. Many venous thrombi remain undetected until embolisation occurs.
Pulmonary Embolism (PE) occurs when part of a venous thrombus dislodges and obstructs the pulmonary circulation. Presentation ranges from mild dyspnoea and pleuritic chest pain to haemodynamic collapse, depending on clot burden and cardiopulmonary reserve. Because symptoms are often non-specific, PE is most commonly confirmed with CT pulmonary angiography (CTPA), while ventilation–perfusion (V/Q) scanning may be used when contrast is contraindicated. Small emboli may produce subtle findings, whereas large or multiple emboli can precipitate acute right ventricular failure. Clinical presentation varies depending on clot size and cardiopulmonary reserve, and may include:
Sudden onset dyspnoea or unexplained hypoxia
Pleuritic or central chest pain
Tachycardia and tachypnoea
Syncope or haemodynamic instability in massive PE
Small emboli may cause minimal symptoms, while large or multiple emboli can cause acute right ventricular failure and sudden collapse.
Screening and Risk Assessment are essential because VTE frequently develops without warning signs. Hospitalised patients should undergo routine VTE risk assessment on admission and be reassessed whenever mobility, illness severity or treatment changes. Screening focuses on identifying factors that promote venous stasis or coagulation activation, such as reduced mobility, acute illness, recent surgery, malignancy or previous VTE. Risk stratification tools help guide prophylaxis decisions, but clinical judgement remains critical, particularly when new symptoms such as unexplained hypoxia, tachycardia or limb asymmetry emerge.
Concept Check
Why are DVT and PE considered manifestations of the same disease process?
How does venous stasis promote clot formation?
Why are venous thrombi more likely to embolise than arterial thrombi?
How does a pulmonary embolus impair oxygenation and circulation?
Why can VTE occur without obvious symptoms?