Coronary Circulation: The Hearts own Blood Supply
Although the heart pumps blood to every tissue in the body, it cannot extract oxygen from the blood within its own chambers. Instead, it relies on a specialised network of vessels known as the coronary circulation. These vessels deliver oxygen-rich blood to the myocardium and remove waste products produced by its high metabolic activity. Because the heart requires a constant and uninterrupted supply of oxygen to function, even brief disruptions to coronary blood flow can lead to profound clinical consequences. Understanding coronary anatomy and physiology is essential for recognising ischemia, interpreting ECG changes, and understanding the pathophysiology of myocardial infarction.
What You Need to Know
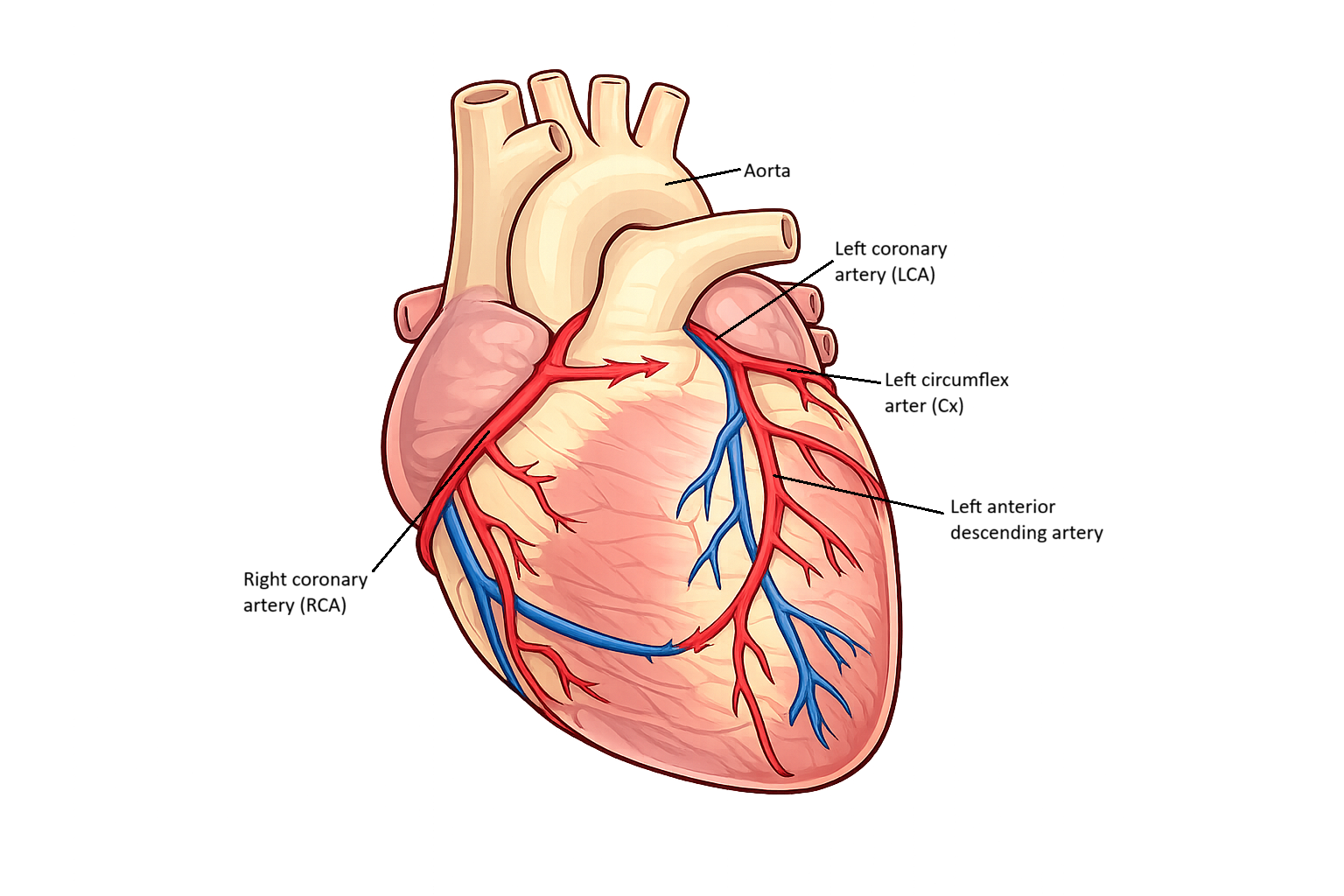
The coronary circulation begins at the base of the aorta, where the left and right coronary arteries originate from the coronary ostia just above the aortic valve. Each coronary artery supplies a specific area of the heart:
The right coronary artery (RCA) supplies the right atrium, right ventricle, and approximately one-third of the interventricular septum.
The left coronary artery (LCA) quickly divides into two major branches: the left anterior descending artery (LAD) and the circumflex artery.
The LAD supplies the anterior wall of the left ventricle and most of the septum, while the circumflex artery supplies the lateral and posterior walls.
Coronary veins collect deoxygenated blood and return it to the right atrium via the coronary sinus. Because the myocardium is highly active tissue with substantial oxygen requirements, the coronary circulation extracts more oxygen from blood than any other organ system. This means that during increased demand, the heart must increase blood flow rather than simply increase oxygen extraction.
Image: Major surface coronary arteries shown (anterior view). Venous and posterior vessels are simplified.
Beyond the Basics
Timing of Coronary Perfusion
Coronary blood flow is uniquely influenced by the mechanical events of the cardiac cycle. In the left ventricle, intramyocardial pressure rises markedly during systole as the ventricular wall contracts. This compresses the coronary vessels running within the myocardium, particularly those supplying the subendocardial layers. As a result, coronary flow through the left coronary circulation is significantly reduced during systole.
During diastole, ventricular relaxation lowers intramyocardial pressure and relieves compression of the coronary vessels. At the same time, aortic pressure remains relatively high due to elastic recoil, creating a strong pressure gradient that drives blood into the coronary arteries. This is why the majority of myocardial perfusion occurs during diastole rather than systole. The dependence on diastolic flow explains why shortened diastole, such as during tachycardia, can markedly reduce coronary perfusion.
Vulnerability of the Subendocardium
The subendocardial region (innermost layer of the heart wall) is exposed to the highest wall stress and intraventricular pressure, making it particularly susceptible to reduced blood flow. Even under normal conditions, perfusion of this region is more precarious than that of the outer myocardial layers. When coronary perfusion pressure falls, or when diastolic filling time is reduced, the subendocardium is the first area to experience inadequate oxygen delivery.
This regional vulnerability helps explain why ischemic changes often begin in the inner myocardial layers. It also highlights the importance of maintaining adequate diastolic pressure and sufficient diastolic duration to support myocardial oxygen supply.
Coronary Autoregulation and Metabolic Control
Coronary vessels possess a strong capacity for autoregulation, allowing them to adjust resistance in response to local metabolic conditions. As myocardial oxygen demand increases, metabolites such as adenosine accumulate within cardiac tissue. These metabolites act directly on vascular smooth muscle, promoting vasodilation and increasing coronary blood flow.
Endothelial-derived factors also play a key role in this process. Nitric oxide released from endothelial cells causes relaxation of vascular smooth muscle, further enhancing flow. Together, these mechanisms ensure that coronary perfusion closely matches myocardial metabolic demand across a wide range of physiological conditions.
Limits of Autoregulation
Although coronary autoregulation is highly effective, it has finite limits. When coronary arteries are partially narrowed, resting blood flow may remain adequate due to compensatory vasodilation downstream of the obstruction. However, this reserve capacity becomes exhausted during increased demand, limiting the ability to further augment flow.
In this setting, myocardial oxygen supply becomes increasingly dependent on perfusion pressure and diastolic duration. Any additional reduction in coronary pressure or increase in heart rate can tip the balance toward ischemia, particularly in vulnerable regions of the myocardium.
Collateral Circulation and Chronic Adaptation
When coronary artery narrowing develops gradually, small interconnecting vessels may enlarge and form collateral pathways. These vessels can partially redirect blood flow around obstructed segments, providing an adaptive response to chronic reductions in perfusion. Collateral circulation may help preserve myocardial viability at rest and during mild exertion.
However, collateral vessels typically cannot match the capacity of a normal coronary artery. During acute occlusion or sudden increases in demand, collateral flow is often insufficient to meet metabolic needs. This limitation explains why chronic adaptation does not reliably protect against acute ischemic events.
Integration of Flow, Pressure, and Demand
Coronary blood flow reflects a delicate balance between perfusion pressure, vascular resistance, and myocardial metabolic demand. The timing of flow within the cardiac cycle, the vulnerability of the subendocardium, and the limits of autoregulatory mechanisms all shape this balance. Appreciating these interactions provides a deeper understanding of why the myocardium is uniquely sensitive to changes in heart rate, blood pressure, and coronary vessel integrity.
Clinical Connections
Coronary circulation is uniquely vulnerable because the myocardium depends almost entirely on continuous oxygen delivery from the coronary arteries. Unlike skeletal muscle, the heart cannot significantly increase oxygen extraction when demand rises, so it must increase coronary blood flow instead. Any narrowing of a coronary artery therefore rapidly limits oxygen delivery during exertion or stress.
Myocardial Ischaemia
Partial obstruction of a coronary artery produces myocardial ischaemia, which manifests clinically as angina — a pressure-like, constricting, or heavy chest discomfort that may radiate to the arm, jaw, neck, or back. This pain reflects anaerobic metabolism, lactate accumulation, and local nerve stimulation within the oxygen-starved myocardium. Rest or vasodilators such as GTN improve symptoms by reducing myocardial oxygen demand and improving coronary perfusion.
Myocardial Infarction (MI)
Complete occlusion produces myocardial infarction, in which prolonged oxygen deprivation causes irreversible myocyte death. The clinical consequences depend on the affected artery:
LAD occlusion damages the anterior wall and interventricular septum, impairing left ventricular pumping
Right coronary artery occlusion may cause inferior infarction and conduction abnormalities
Circumflex occlusion affects lateral ventricular function
ECG Changes
ECG changes arise because ischaemic and infarcted myocardium alters electrical conduction. ST elevation, ST depression, T-wave inversion, and Q-wave formation each reflect different stages and depths of myocardial injury. Troponin rises as damaged cardiac cells leak intracellular proteins into the bloodstream, providing biochemical confirmation of infarction.
Early recognition of myocardial ischaemia or infarction is lifesaving. Ongoing chest pain, dyspnoea, diaphoresis, nausea, hypotension, or arrhythmias may all signal compromised coronary perfusion. Immediate interventions are aimed at restoring oxygen supply and limiting myocardial workload:
Oxygen improves arterial oxygen delivery
GTN reduces preload and dilates coronary vessels
Aspirin limits further thrombus formation
Rapid medical escalation enables reperfusion therapy
Long-term coronary disease reflects chronic atherosclerotic narrowing of the coronary arteries. Management targets both symptom control and prevention of future events through antiplatelet therapy, lipid lowering, blood pressure control, and lifestyle modification. In advanced disease, percutaneous coronary intervention (stenting) or coronary artery bypass grafting restores blood flow to ischaemic myocardium, improving survival and functional capacity.
Concept Check
Why does most coronary perfusion occur during diastole rather than systole?
What regions of the heart are supplied by the LAD, RCA, and circumflex arteries?
How does autoregulation help meet the heart’s metabolic demands?
Why is the subendocardial region more vulnerable to ischemia?
What clinical signs differentiate angina from myocardial infarction?