Haematopoiesis and the Bone Marrow
Haematopoiesis is the continuous process by which the body produces all types of blood cells: red cells, white cells, and platelets. This essential function occurs primarily in the bone marrow, a soft, highly vascular tissue located within the medullary cavities of certain bones. Because blood cells have limited lifespans, haematopoiesis must operate efficiently and dynamically, responding to illness, injury, oxygen demands, and hormonal signals.
What You Need to Know
Haematopoiesis is the continuous process by which all blood cells are produced, replaced, and maintained throughout life. It begins with haematopoietic stem cells (HSCs), rare multipotent cells located in the bone marrow that can both self-renew and differentiate into every blood cell type. These stem cells give rise to two major developmental pathways that form the foundation of blood and immune function.
From HSCs, blood cells diverge into two broad lineages:
Myeloid lineage, which produces red blood cells, platelets, neutrophils, eosinophils, basophils, and monocytes
Lymphoid lineage, which produces B-cells, T-cells, and natural killer (NK) cells
This branching architecture allows the marrow to generate both oxygen-carrying cells, clotting cells, and multiple layers of immune defence.
Within the bone marrow, developing cells are guided by a complex network of growth factors and cytokines that regulate survival, proliferation, and maturation. Key regulatory signals include:
Erythropoietin (EPO), produced by the kidneys to stimulate red blood cell production in response to hypoxia
Thrombopoietin (TPO), which controls platelet production from megakaryocytes
Colony-stimulating factors (CSFs), which drive white blood cell development and immune readiness
These signals allow the marrow to rapidly increase or decrease production in response to bleeding, infection, inflammation, or oxygen demand.
In adults, active (red) bone marrow is found mainly in the vertebrae, pelvis, sternum, ribs, and the proximal ends of the femur and humerus, where blood cell production is most efficient. With age, some marrow spaces are replaced by fatty (yellow) marrow, reducing baseline production capacity. Importantly, this process is reversible. During periods of high demand, such as severe anaemia, haemorrhage, or chronic disease, yellow marrow can revert to red marrow, allowing the body to expand blood cell production when it is most needed.
Beyond the Basics
Dynamic Regulation of Blood Cell Production
Haematopoiesis is a continuous, highly regulated process that maintains circulating blood cell populations within narrow physiological limits. At any given moment, the bone marrow must balance routine cellular turnover with the capacity to rapidly increase production in response to stress, illness, or blood loss. This balance is essential, as underproduction compromises oxygen delivery and immune defence, while overproduction increases blood viscosity and inflammatory risk. Rather than producing cells at a fixed rate, haematopoiesis adapts dynamically to systemic signals, ensuring that supply matches physiological demand.
The Bone Marrow Microenvironment (Haematopoietic Niche)
Blood cell production does not occur in isolation. Haematopoietic stem cells reside within a specialised bone marrow microenvironment, often referred to as the haematopoietic niche. This niche consists of stromal cells, macrophages, fibroblasts, endothelial cells, and extracellular matrix components that together provide both physical scaffolding and biochemical signalling.
These supporting cells regulate stem cell quiescence, proliferation, differentiation, and survival. Growth factors, cytokines, and cell-to-cell contact signals ensure that developing blood cells mature appropriately and in correct proportions. Without this microenvironmental regulation, effective haematopoiesis cannot occur, even if stem cells are present.
Red Blood Cell Production and Maturation
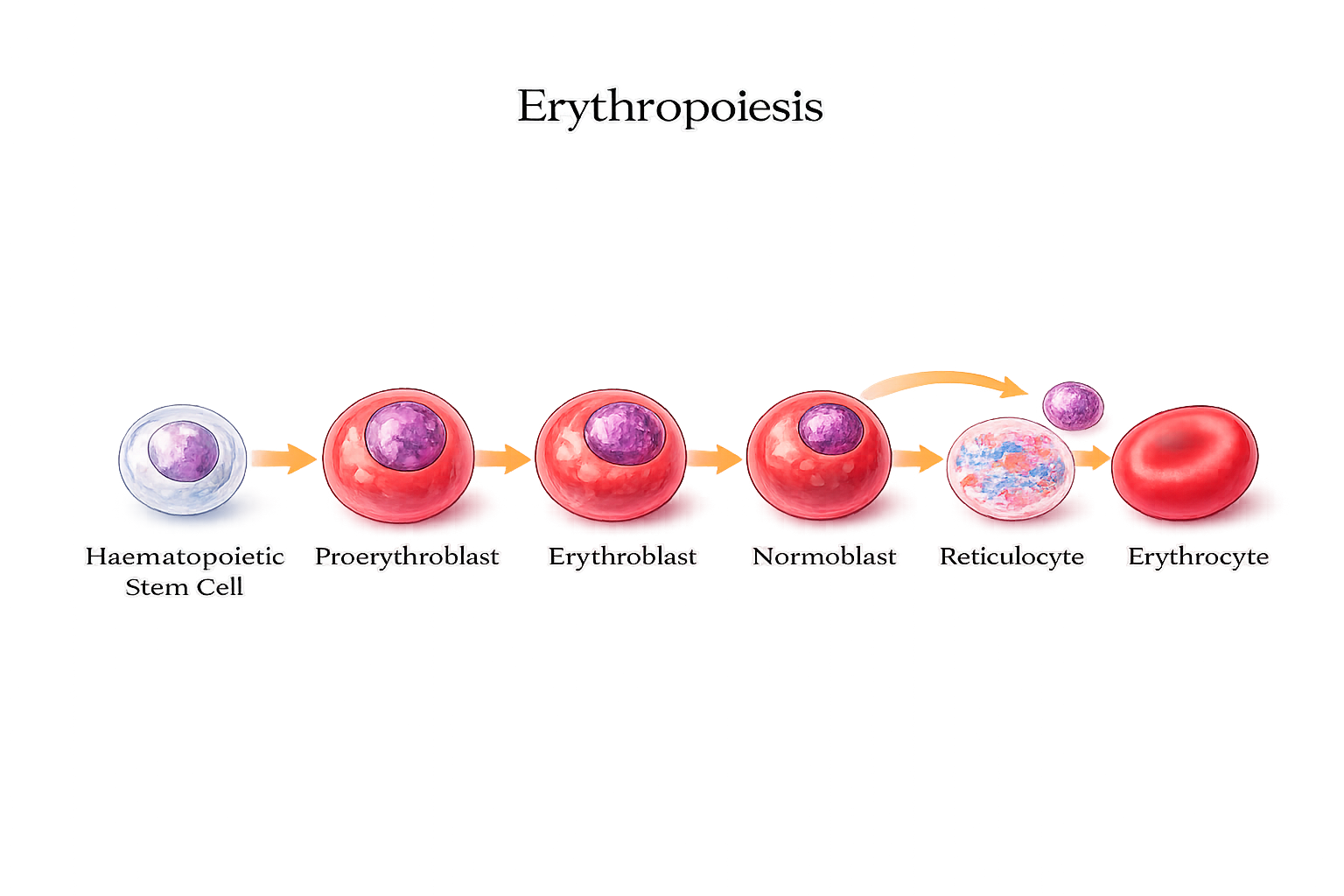
Red blood cell production follows a highly ordered maturation sequence designed to optimise oxygen transport. As erythroid precursors mature (immature red blood cells), they progressively condense and ultimately expel their nucleus and organelles. This process maximises internal space for haemoglobin while eliminating structures that would otherwise consume oxygen.
Once nuclear extrusion occurs, immature red blood cells enter circulation as reticulocytes. These cells retain residual ribosomal material, allowing limited protein synthesis as final membrane and cytoskeletal refinement takes place. Reticulocytes typically complete maturation within 24 hours of entering the bloodstream, becoming fully functional erythrocytes.
Image: Red blood cells develop in the bone marrow from haematopoietic stem cells, progressing through precursor stages before the nucleus is expelled to form a reticulocyte, which matures into a fully functional erythrocyte.
Reticulocyte Count as a Marker of Marrow Responsiveness
Reticulocyte numbers provide a real-time reflection of bone marrow activity. In states of blood loss or anaemia, increased erythropoietin stimulation accelerates red cell production, leading to a rise in circulating reticulocytes. This response indicates that the marrow is capable of mounting an appropriate compensatory response.
Conversely, a low reticulocyte count in the presence of anaemia suggests impaired marrow function or insufficient regulatory signalling. Reticulocyte measurement therefore offers insight into how anaemia is being handled physiologically, rather than simply describing its severity.
White Blood Cell Production and Functional Diversity
White blood cell development is far more heterogeneous (comprised of a mixture of elements) than red blood cell production. Different leukocyte lineages mature at different rates and in response to distinct regulatory signals. Neutrophils are produced rapidly and in large quantities, reflecting their role as short-lived, frontline immune responders. During infection or inflammation, neutrophil release can increase dramatically within hours.
Lymphocyte development follows a slower, more complex pathway. Some lymphocytes mature within the bone marrow, while others migrate to secondary lymphoid organs such as the thymus, lymph nodes, and spleen, where further differentiation and selection occur. This extended maturation process reflects the need for specificity, immune tolerance, and memory formation.
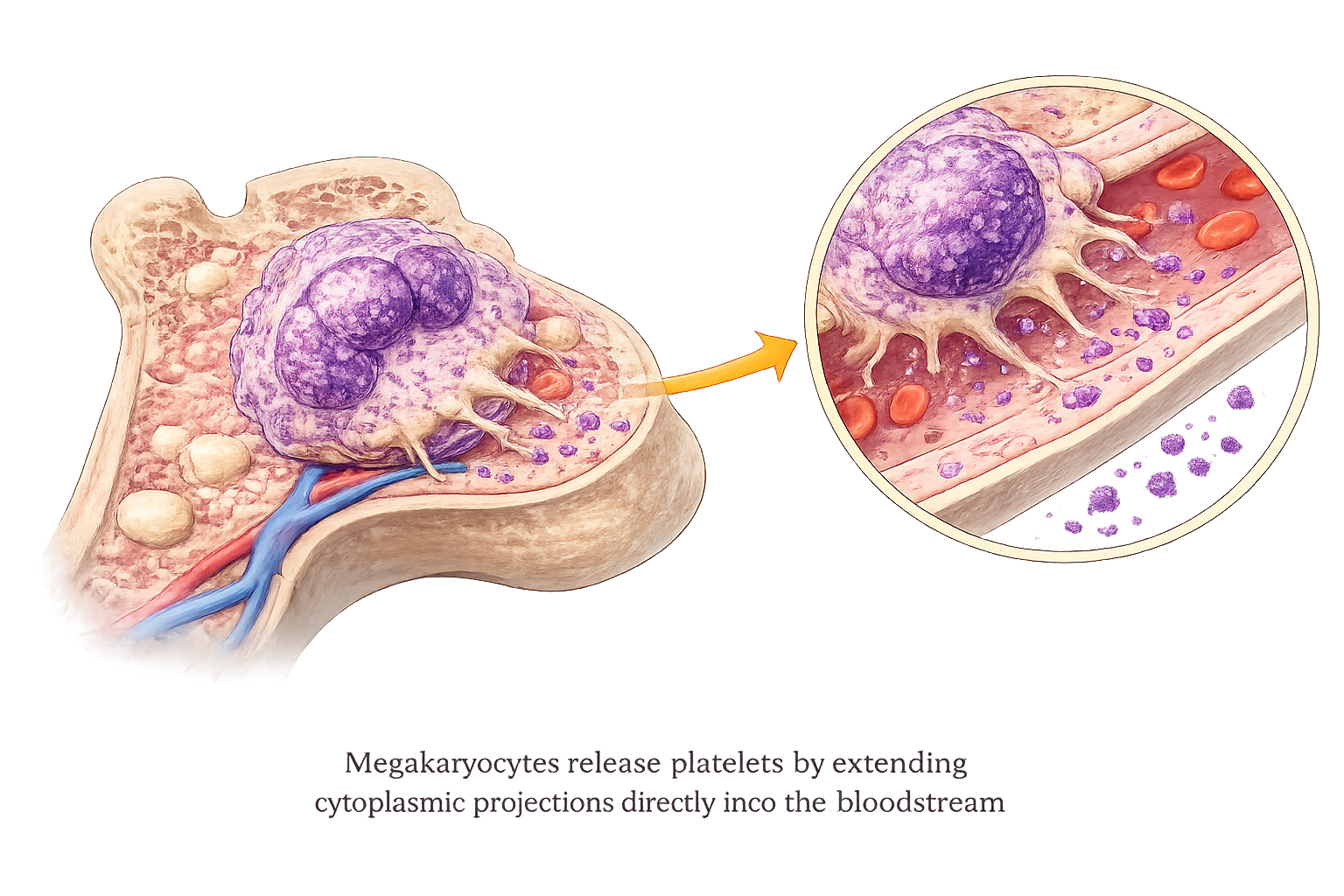
Platelet Formation and Megakaryocyte Specialisation
Platelet production differs fundamentally from red and white cell formation. Platelets are not complete cells, but cytoplasmic fragments derived from megakaryocytes (a giant bone marrow cell responsible for producing platelets). These very large precursor cells sit next to bone marrow blood vessels and release platelets by extending thin cytoplasmic projections directly into the bloodstream.
Mechanical shear forces within the bloodstream help fragment these projections into thousands of platelets, which then enter circulation. This unique mechanism allows rapid adjustment of platelet numbers in response to haemostatic demand without requiring full cellular division.
Regulatory Signals and Systemic Integration
Haematopoiesis is governed by a complex network of hormonal and cytokine signals. Erythropoietin stimulates red cell production in response to tissue hypoxia, while colony-stimulating factors regulate white cell proliferation and differentiation. Thrombopoietin controls platelet production, adjusting output according to circulating platelet mass. These signals integrate information from oxygen levels, immune activity, inflammatory status, and metabolic demand, allowing the bone marrow to respond appropriately to changing physiological conditions.
Adaptive Response to Stress and Illness
During acute stress, haemorrhage, or infection, bone marrow activity increases rapidly. Stem cell proliferation accelerates, maturation time shortens, and stored precursor pools are mobilised. This adaptive capacity is essential for survival during physiological challenge.
In contrast, chronic illness, malnutrition, systemic inflammation, or marrow infiltration can suppress haematopoietic activity. Reduced responsiveness in these settings is due to the prioritisation of energy conservation or disruption of the marrow microenvironment rather than failure of the stem cell population itself.
Haematopoiesis as a Homeostatic System
The primary role of haematopoiesis is not maximal production, but stability. By continuously adjusting output to meet physiological demand, the bone marrow maintains oxygen delivery, immune defence, and haemostatic integrity across a wide range of conditions.
Clinical Connections
Disorders of haematopoiesis produce characteristic patterns of illness because the bone marrow is responsible for generating red cells, white cells, and platelets simultaneously. When marrow function is impaired, patients often present with combinations of fatigue, infection, and bleeding rather than a single isolated abnormality. Aplastic anaemia represents near-complete failure of marrow activity, leading to pancytopenia with profound anaemia, recurrent infections, and spontaneous bleeding. Myelodysplastic syndromes involve disordered maturation, so blood cell numbers may appear normal or low but function poorly, increasing the risk of infection and haemorrhage.
Malignant transformation of marrow precursors leads to leukaemia, in which immature white cells proliferate uncontrollably and crowd out normal blood-forming cells. This results in a paradoxical state where white cell counts may be high, yet immune defence is weak, while red cell and platelet production falls. Patients may present with:
persistent fatigue and pallor from anaemia
fever or recurrent infections due to neutropenia
bruising, petechiae, or bleeding from thrombocytopenia
These combinations should prompt urgent investigation of marrow function.
Many non-malignant conditions also suppress haematopoiesis. Chemotherapy and radiation directly damage rapidly dividing marrow cells. Severe infections and autoimmune disease can disrupt growth factor signalling. Nutritional deficiencies, particularly of iron, B12, and folate, limit the raw materials needed for effective blood cell production. The resulting cytopenias may be subtle at first but become clinically significant if not recognised.
Bone marrow biopsy remains a central diagnostic tool when haematological disease is suspected, allowing direct assessment of cellularity, lineage development, and the presence of malignant or dysplastic cells. For nurses, recognising early signs of marrow suppression, fatigue, fever, mucosal bleeding, or easy bruising, is critical, particularly in patients receiving cytotoxic or immunosuppressive therapies. Prompt escalation can prevent life-threatening infection or haemorrhage and allows timely intervention.
Concept Check
What are the two major stem cell lineages in haematopoiesis?
How do erythropoietin and thrombopoietin regulate blood cell production?
Why are reticulocyte counts clinically important?
What conditions can suppress bone marrow function?
How does marrow infiltration by cancer affect blood cell production?