White Blood Cells: Types and Functions
White blood cells (WBC), or leukocytes, are the body’s primary defenders against infection, injury, and foreign material. Unlike red blood cells, which remain within vessels, WBCs move between blood and tissues, patrolling for pathogens and helping coordinate immune responses. Their diversity is an example of the complexity of the immune system: each type of WBC performs specialised tasks ranging from rapid destruction of bacteria to long-term immune memory. Changes in WBC number and function provide essential clues about infection, inflammation, allergic responses, and haematological diseases.
What You Need to Know
White blood cells are the mobile defence cells of the immune system. They circulate in the blood but also migrate into tissues, lymphatic organs, and sites of injury or infection. Their primary role is to identify, neutralise, and remove pathogens, abnormal cells, and damaged tissue. Unlike red blood cells, white blood cells retain nuclei and complex internal machinery, allowing them to move actively, communicate chemically, and adapt their responses to different threats.
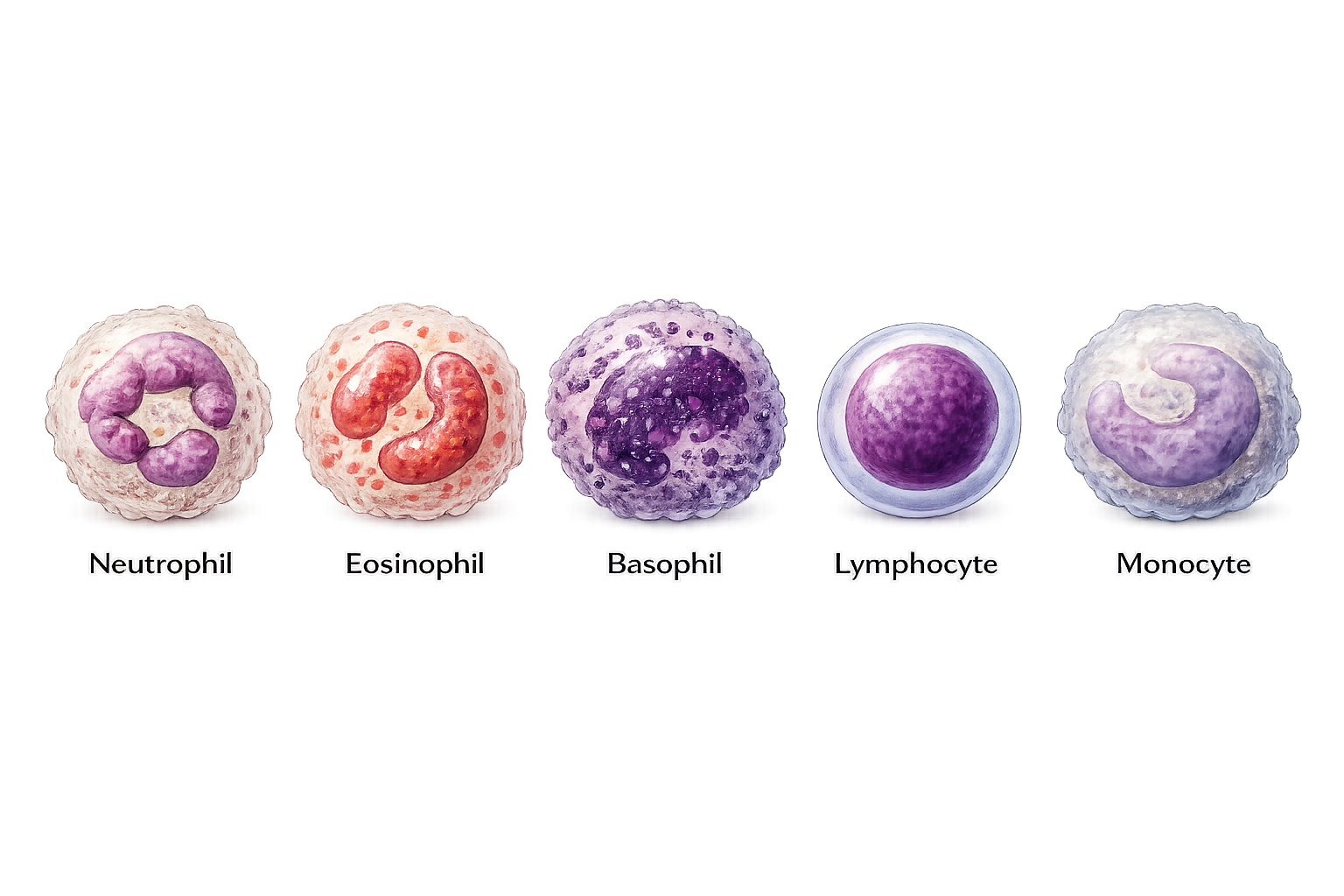
WBCs are divided into two broad groups based on the appearance of their cytoplasm (the gelatinous liquid that fills the inside of a cell): granulocytes and agranulocytes. Granulocytes include neutrophils, eosinophils, and basophils, which contain enzyme-filled granules that are released during immune responses. Agranulocytes include lymphocytes and monocytes, which have more specialised roles in immune regulation and long-term defence.
The major white blood cell types perform distinct but coordinated functions:
Neutrophils are the most abundant WBCs and act as rapid first responders to bacterial infection. They migrate quickly to inflamed tissue, engulf pathogens (phagocytosis), and release antimicrobial enzymes and reactive oxygen species.
Eosinophils are particularly involved in defence against parasites and play an important role in allergic and asthmatic inflammation by releasing toxic granules and inflammatory mediators.
Basophils release histamine, heparin, and other mediators that increase blood vessel permeability and support inflammatory and allergic responses.
Lymphocytes include B cells, T cells, and natural killer (NK) cells. B cells produce antibodies, T cells coordinate immune responses and destroy infected cells, and NK cells provide rapid surveillance against virus-infected and malignant cells without requiring prior exposure.
Monocytes circulate in the blood before migrating into tissues, where they differentiate into macrophages or dendritic cells. These long-lived cells engulf debris, destroy pathogens, present antigens to lymphocytes, and coordinate tissue repair and immune activation.
Together, white blood cells provide both immediate defence and long-term immune memory, allowing the body to respond rapidly to new infections while mounting faster, more effective responses to previously encountered pathogens.
Image: White blood cells (leukocytes), each with distinct roles in immune defence.
Beyond the Basics
Bone Marrow Production and Lineage Commitment
All white blood cells originate from haematopoietic stem cells in the bone marrow. These stem cells give rise to multiple immune lineages under the influence of growth factors and cytokines (chemical messengers that guide immune development). Unlike red blood cells, which follow a single, highly standardised maturation pathway, white blood cell development diverges early. This divergence produces cells with very different sizes, structures, and functions, allowing the immune system to deal with a wide range of biological threats.
Some progenitor cells (a partially specialised cell that can develop into a specific group of related cells, but with limited ability to keep dividing) are directed toward innate immune pathways, producing neutrophils, monocytes, and natural killer cells that provide immediate defence. Others differentiate into lymphoid lineages, giving rise to B cells and T cells that support adaptive immunity. This branching architecture of development is what gives the immune system both speed and specificity.
Circulation, Margination, and Tissue Migration
Although white blood cells are measured in the bloodstream, their primary site of action is in tissues. Many spend only short periods circulating before exiting into organs, connective tissue, or sites of injury. In healthy vessels, WBCs flow freely, but during inflammation they begin to slow and adhere loosely to the inner vessel wall, a process known as margination.
Inflamed tissues release chemical attractants called chemokines that guide white blood cells toward the site of damage. WBCs respond by rolling along the endothelium, attaching firmly, and then squeezing between endothelial cells to leave the bloodstream. This process, known as transmigration, allows immune cells to move directly to where they are needed rather than acting blindly from within the circulation.
Innate Immune Cells: Rapid, Non-Specific Defence
Innate immune cells provide immediate protection and do not require prior exposure to a pathogen. They recognise broad molecular patterns that indicate danger, such as bacterial cell wall components or molecules released by injured cells.
Neutrophils are the most abundant of these cells and are designed for rapid response. They migrate quickly to sites of infection, engulf microbes, and destroy them using enzymes and reactive oxygen species (ROS). Monocytes circulate briefly in the blood before entering tissues, where they become macrophages or dendritic cells. These cells clear debris, kill pathogens, and produce cytokines that shape the inflammatory response.
Natural killer (NK) cells provide immune surveillance by identifying cells that have lost normal surface markers, a common feature of virus-infected or cancerous cells. Rather than recognising specific antigens, NK cells detect abnormal patterns of self, allowing them to eliminate threats that evade other immune mechanisms.
Adaptive Immune Cells: Specificity and Memory
Adaptive immunity is mediated primarily by lymphocytes. B cells and T cells each express highly specific receptors that recognise particular antigens. When these receptors bind their target, the cell becomes activated and undergoes clonal expansion, producing large numbers of identical cells directed against the same pathogen.
B cells differentiate into plasma cells that secrete antibodies, which bind to antigens and tag them for destruction. T cells perform multiple roles, including coordinating immune responses, killing infected cells, and regulating immune activity. Some activated lymphocytes become memory cells that persist long-term, allowing the immune system to respond more rapidly and effectively if the same pathogen is encountered again. This memory is the basis of vaccination and long-lasting immunity.
Activation, Proliferation, and Immune Control
White blood cells remain largely inactive until stimulated by appropriate signals. Activation may occur through direct recognition of a pathogen, interaction with antigen-presenting cells, or exposure to cytokines released during inflammation. Once activated, WBCs rapidly alter gene expression (instructions stored within DNA), proliferate, and secrete signalling molecules that amplify or suppress immune responses. This regulation is essential. An immune response that is too weak leads to infection, while one that is too strong causes tissue damage, autoimmunity, or systemic inflammation. The immune system therefore operates through tightly controlled feedback loops that balance defence with self-protection.
Lifespan Variability and Turnover
White blood cells vary greatly in lifespan. Neutrophils may survive only hours to days, due to their role as expendable first responders. Macrophages and dendritic cells can persist for weeks or months in tissues. Memory lymphocytes may survive for years, maintaining long-term immune readiness. This turnover allows the immune system to remain flexible and responsive while preventing accumulation of damaged or autoreactive cells that could trigger disease.
Integration with Other Body Systems
Immune function is closely linked to other physiological systems. Blood vessels provide the delivery routes for immune cells, the lymphatic system enables antigen sampling and immune coordination, and endocrine hormones modulate immune activity. Stress hormones such as cortisol suppress inflammation, while nutritional status and oxygen availability influence immune cell metabolism and survival. This integration explains why chronic illness, malnutrition, or physiological stress can weaken immunity even when the immune system itself is not primarily diseased.
Structural Features Supporting Immune Function
White blood cells are larger and more complex than red blood cells. Their nuclei allow dynamic gene regulation, and their organelles support protein synthesis, energy production, and intracellular killing. A flexible cytoskeleton enables movement, shape change, and engulfment of pathogens. These structural features allow white blood cells to prioritise adaptability and responsiveness over efficient circulation, reflecting their role as active defenders rather than passive transport cells.
Clinical Connections
Changes in white blood cell count and distribution provide some of the most important clues to underlying disease. Leukocytosis (elevated WBC count) most commonly occurs with infection, inflammation, tissue injury, or physiological stress, but the type of white cell that rises is often more informative than the total number. A predominance of neutrophils typically indicates bacterial infection, tissue necrosis, or corticosteroid exposure, whereas a lymphocyte-predominant pattern is more consistent with viral infection or certain chronic inflammatory states. Eosinophilia points toward allergic disease, asthma, drug reactions, or parasitic infection, while basophilia is more often associated with myeloproliferative disorders.
A reduced white blood cell count (leukopenia) carries significant clinical risk because it compromises innate and adaptive immune defence. Neutropenia in particular places patients at high risk of overwhelming infection, often with minimal early symptoms. Causes include:
bone marrow suppression from chemotherapy, radiation, or viral infection
autoimmune destruction of white cells
severe sepsis, in which immune cells are consumed faster than they can be produced
Because fever or localised inflammation may be blunted, neutropenic patients require urgent evaluation for even minor infective symptoms. In clinical settings, neutropenic patients are often isolated from other patients as a preventative strategy.
Abnormal white blood cell morphology on blood film can be as important as the count itself. The presence of immature cells (blasts), extreme leukocytosis, or distorted cell forms suggests bone marrow disease such as leukaemia or myelodysplasia. In these conditions, white blood cells may be numerous but functionally ineffective, leaving patients simultaneously inflamed and immunocompromised.
Interpreting WBC patterns is essential for early recognition of deterioration. Immunosuppressed patients may not mount a normal inflammatory response, so subtle changes in vital signs, confusion, or malaise may be the first signs of serious infection. Prompt escalation, protective isolation when indicated, and strict infection-control practices are critical to preventing rapid clinical decline.
Concept Check
What is the difference between granulocytes and agranulocytes?
How do neutrophils respond to bacterial infection?
What roles do B-cells and T-cells play in adaptive immunity?
Why are immunocompromised patients at higher risk of severe infection?
What conditions commonly cause eosinophilia?