Heart Valves: Structure and Function
The heart valves are delicate yet powerful structures that ensure blood moves in only one direction through the heart. Their design allows them to open and close passively in response to pressure changes during the cardiac cycle, preventing backflow and maintaining efficient forward movement of blood. Although small in size, the valves play a critical role in cardiac function, and even mild abnormalities can significantly impair circulation. Understanding valve anatomy and physiology is essential for interpreting heart sounds, recognising murmurs, and identifying early signs of valve dysfunction.
What You Need to Know
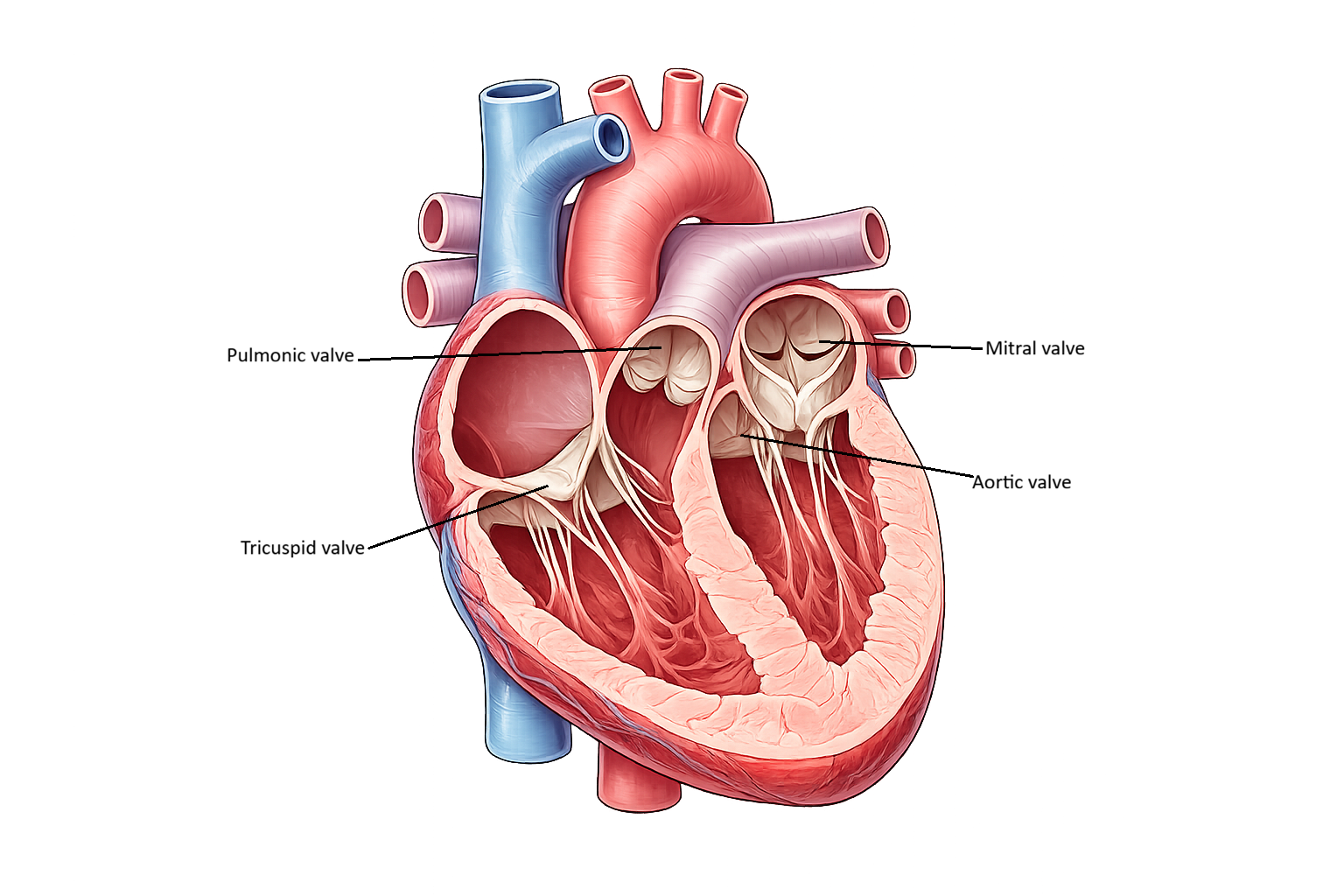
The heart contains four valves that ensure blood flows in one direction through the cardiac chambers: the tricuspid, mitral, pulmonary, and aortic valves. The tricuspid and mitral valves are called atrioventricular (AV) valves because they lie between the atria and ventricles. The tricuspid valve, located on the right side of the heart, has three flexible leaflets, while the mitral valve on the left has two. These valves open when atrial pressure exceeds ventricular pressure, allowing blood to move from the atria into the ventricles during diastole. When the ventricles begin to contract, rising ventricular pressure forces the AV valves closed, preventing blood from flowing backward into the atria.
The AV valves are stabilised by chordae tendineae, which are thin but strong fibrous cords that attach the valve leaflets to papillary muscles in the ventricular walls. When the ventricles contract, the papillary muscles tighten these cords, holding the valves in place and preventing prolapse into the atria.
The AV valve apparatus therefore includes:
the valve leaflets
the chordae tendineae
the papillary muscles
These structures function together to ensure that ventricular contraction produces forward blood flow rather than regurgitation.
The pulmonary and aortic valves are known as semilunar valves because each is formed by three half-moon-shaped cusps. These valves sit at the outflow tracts of the right and left ventricles, where blood enters the pulmonary artery and aorta. They open when ventricular pressure exceeds arterial pressure, allowing blood to be ejected during systole. Unlike the AV valves, they do not require chordae or papillary muscles because arterial pressure naturally holds them closed when the ventricles relax. When blood briefly flows backward at the end of systole, it fills the cusps and snaps the valves shut, creating a tight seal that prevents regurgitation and preserves efficient one-way circulation.
Beyond the Basics
Principles of Valve Function
Cardiac valve function is governed entirely by pressure gradients within the heart. Valves do not contain muscle tissue and do not actively contract; instead, they open and close passively in response to changing pressures between chambers and great vessels. This pressure-dependent mechanism ensures unidirectional blood flow and minimises energy expenditure during the cardiac cycle.
During ventricular filling, pressure within the atria exceeds ventricular pressure, allowing atrioventricular valves to open. As ventricular contraction begins and pressure rises, these valves close promptly, preventing retrograde flow. Semilunar valves open only when ventricular pressure exceeds arterial pressure and close as soon as ventricular pressure falls. The precise timing of these events is essential for efficient cardiac output.
Structural Adaptation of Cardiac Valves
The anatomy of each valve indicates the mechanical demands placed upon it. Atrioventricular and semilunar valves differ in size, flexibility, and supporting structures to suit their roles within the cardiac cycle.
Atrioventricular (AV) valves are larger and more flexible, allowing high-volume, low-pressure flow during ventricular filling. Their leaflets are supported by chordae tendineae and papillary muscles, which stabilise the valve during systole and prevent inversion into the atria.
Semilunar valves are smaller and more rigid, designed to withstand high arterial pressures during ventricular ejection. Their pocket-like cusps allow rapid closure and prevent backflow into the ventricles when pressure reverses.
These structural differences enable valves to maintain competence across a wide range of pressures and flow conditions.
Valve Competence and Ventricular Geometry
Valve competence depends not only on the valve leaflets themselves but also on the surrounding myocardial and connective tissue structures. Normal ventricular geometry ensures appropriate alignment and tension across valve leaflets during closure.
Alterations in ventricular shape can disrupt this balance. Ventricular dilation or hypertrophy may displace papillary muscles or stretch valve annuli, preventing complete leaflet coaptation and leading to regurgitation. In these cases, the valve tissue may be structurally normal, but functional incompetence arises due to altered chamber mechanics.
Degenerative and Structural Valve Changes
Over time, age-related degeneration of connective tissue can impair valve integrity. Mitral valve prolapse commonly results from weakening or redundancy of leaflet tissue, allowing abnormal bulging into the left atrium during systole. Although often benign, prolapse may progress to significant regurgitation in some individuals.
Calcification is particularly common in the aortic valve. Progressive deposition of calcium stiffens the valve cusps and narrows the valve orifice, increasing resistance to ventricular ejection. This gradual reduction in valve area characterises aortic stenosis and places increasing pressure load on the left ventricle.
Anatomical Insight and Clinical Recognition
Because valve dysfunction often occurs due to changes in pressure, geometry, or tissue integrity rather than isolated leaflet abnormalities, anatomical knowledge is essential for accurate interpretation of murmurs, imaging findings, and haemodynamic consequences. This structural–functional relationship forms the foundation for recognising valvular pathology and appreciating its impact on overall cardiovascular performance.
Clinical Connections
Valvular heart disease alters the normal one-way flow of blood and gradually places abnormal pressure or volume loads on the heart. Stenotic valves restrict forward flow, forcing the chamber behind the valve to generate higher pressure to move blood across the narrowed opening. Over time this leads to chamber thickening (hypertrophy) and reduced compliance, eventually impairing filling and cardiac output. Regurgitant valves, by contrast, allow blood to leak backward, causing volume overload and progressive chamber dilation. Both processes ultimately lead to heart failure if untreated.
Clinically, different valves produce distinct patterns of disease:
Aortic stenosis: left ventricular hypertrophy, angina, syncope, heart failure
Mitral regurgitation: left atrial dilation, pulmonary congestion, atrial fibrillation
Tricuspid disease: systemic venous congestion, hepatomegaly, peripheral oedema
Pulmonary valve disease: right ventricular strain and reduced pulmonary flow
Murmurs arise from turbulent blood flow across abnormal valves. Timing is key: systolic murmurs usually result from AV valve regurgitation or semilunar valve stenosis, while diastolic murmurs suggest AV valve stenosis or semilunar valve regurgitation. Careful auscultation, combined with blood pressure, pulse quality, and symptom patterns, helps nurses detect early deterioration.
Echocardiography provides direct visualisation of valve structure, opening area, leaflet movement, and regurgitant flow, allowing disease severity to be graded. Progression may be slow and silent, so regular monitoring is essential.
Valve disease requires ongoing monitoring for:
worsening breathlessness, fatigue, or reduced exercise tolerance
new arrhythmias (especially atrial fibrillation)
pulmonary or peripheral oedema
changes in blood pressure or pulse pressure
Concept Check
Why do the AV valves require chordae tendineae, while the semilunar valves do not?
How do pressure changes during the cardiac cycle control valve opening and closure?
What is the difference between stenosis and regurgitation?
Which clinical signs might indicate a patient is developing aortic stenosis?
Explain why mitral regurgitation increases the risk of pulmonary congestion.