The Spleen: Structure, Function and Clinical Importance
The spleen is often overlooked in basic anatomy, yet it is one of the most important organs for immune defence, blood filtration, and red blood cell maintenance. Tucked beneath the left rib cage, the spleen continuously samples circulating blood, removing old or damaged cells, responding to pathogens, and recycling key components such as iron. Its role becomes most evident when it is absent or dysfunctional, patients without a spleen are suddenly vulnerable to severe infections, rapid deterioration from common pathogens, and complications in blood cell regulation. Understanding the spleen’s anatomy and physiology is essential for interpreting haematological abnormalities, recognising splenic enlargement, and safely caring for patients who are asplenic or immunocompromised.
What You Need to Know
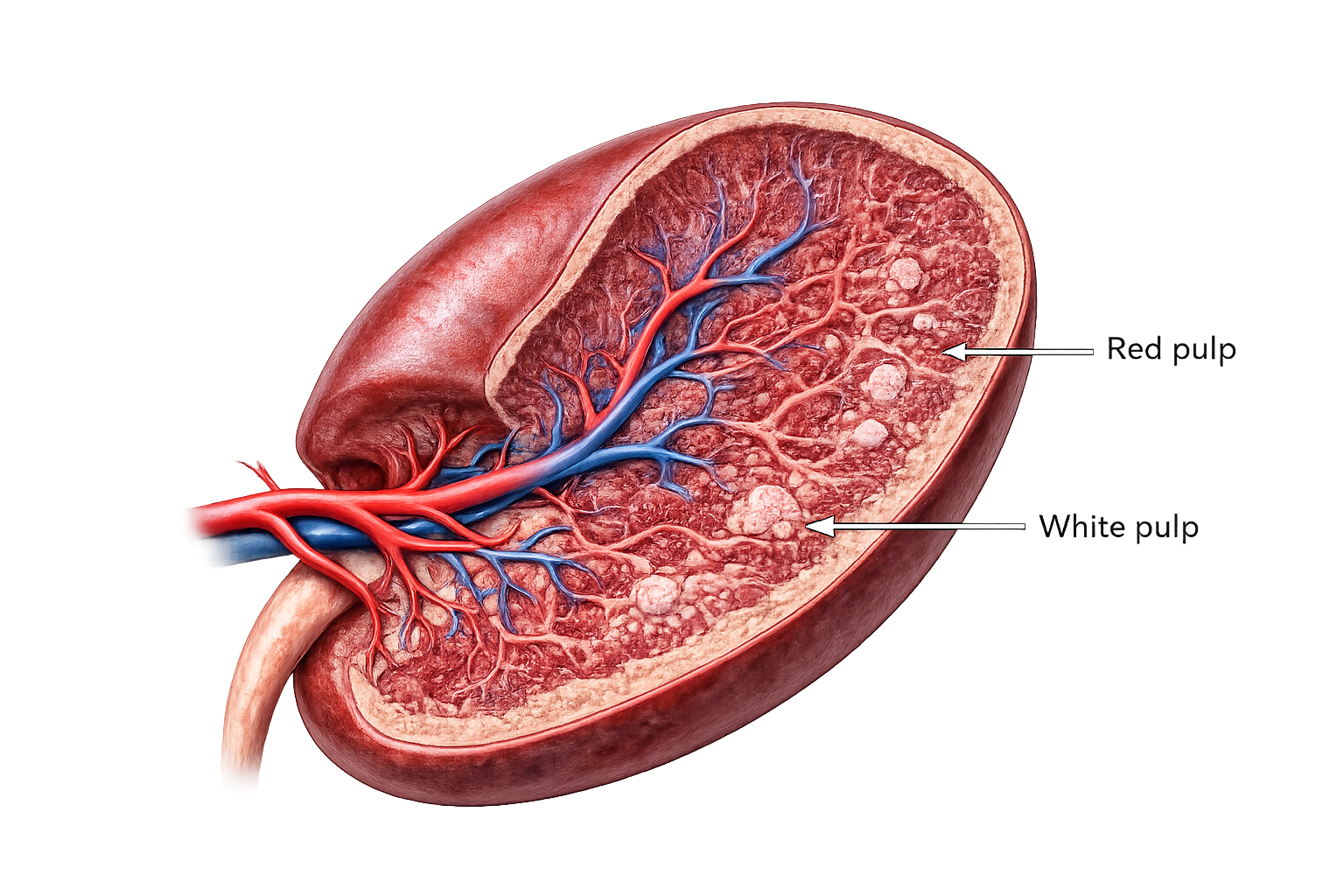
The spleen is composed of two main types of tissue: white pulp and red pulp, each serving distinct but complementary roles.
White pulp surrounds central arteries and functions much like a lymph node, initiating immune responses. It is rich in lymphocytes - B-cells that produce antibodies and T-cells that target infected cells. As blood flows through, the white pulp detects circulating pathogens and activates the adaptive immune system.
Red pulp makes up the majority of the spleen. It filters blood by forcing red blood cells through narrow channels called venous sinuses. Healthy RBCs can deform and pass through easily, but old, rigid, or damaged cells become trapped and are removed by macrophages. This process also clears cellular debris, abnormal platelets, and microorganisms. Through these mechanisms, the spleen maintains blood quality, prevents circulating infections, and regulates RBC lifespan.
Image: Red pulp and white pulp in a transected spleen.
Beyond the Basics
Red Pulp Architecture and Mechanical Filtration
The red pulp accounts for the majority of splenic tissue and is responsible for filtering circulating blood. Blood entering the spleen is diverted from fast-flowing arterial circulation into a low-pressure, slow-moving environment. Red blood cells must pass through narrow endothelial slits to re-enter venous circulation.
Healthy red blood cells are highly deformable and can change shape to pass through these tight spaces. As red blood cells age or become structurally abnormal, their membranes stiffen and lose flexibility. These cells are unable to traverse the splenic cords efficiently and become trapped within the red pulp. This process is mechanical rather than biochemical. The spleen does not rely on molecular tagging alone; instead, it physically challenges red blood cells, ensuring that only cells capable of normal deformation remain in circulation.
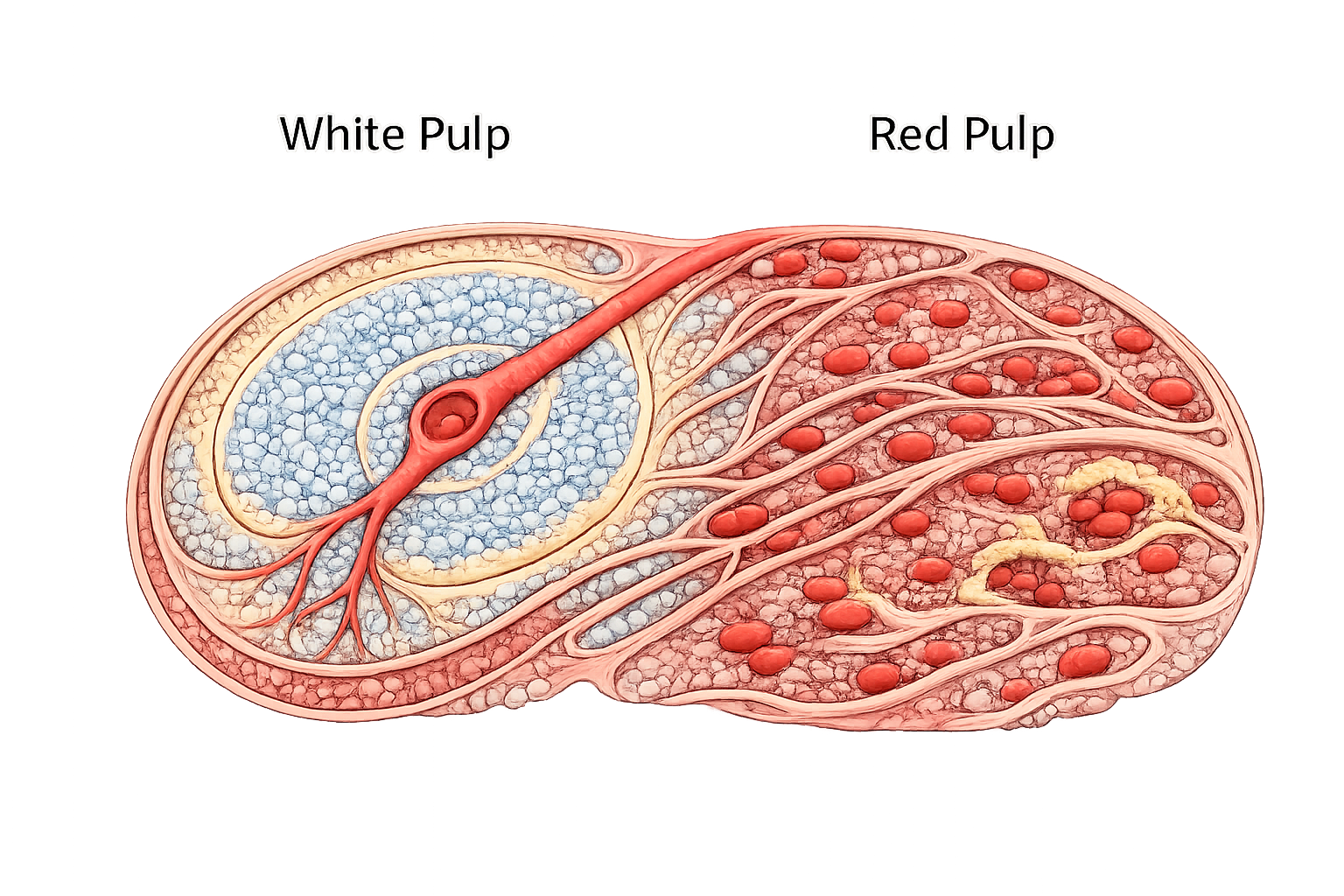
Image: Splenic microstructure. The spleen is divided into white pulp and red pulp. White pulp surrounds central arterioles and functions in immune surveillance and lymphocyte activation, while red pulp filters blood, removes aged red blood cells, and recycles iron.
Macrophage Recognition and Red Cell Clearance
Once red blood cells are sequestered (isolated) within the red pulp, resident macrophages assess them for structural integrity. Stiffened, damaged, or antibody-coated red cells are recognised as non-functional and are phagocytosed. During this process, haemoglobin is broken down into its components. The globin chains are recycled as amino acids, while iron is carefully conserved. Iron is released from haem, bound to transport proteins, and returned to the bone marrow, where it is reused for haemoglobin synthesis in newly formed red blood cells. This recycling function is critical for maintaining iron homeostasis and reducing reliance on dietary iron intake.
Low-Oxygen Environment and Red Cell Stress Testing
The splenic red pulp is deliberately hypoxic. This low-oxygen environment places additional metabolic stress on red blood cells. Cells with compromised membranes, enzyme deficiencies, or abnormal haemoglobin are less able to tolerate this environment and are preferentially removed. In this way, the spleen functions as a physiological quality-control organ, ensuring that circulating red blood cells are both mechanically flexible and metabolically resilient.
White Pulp and Immune Surveillance
Interspersed within the red pulp is the white pulp, which plays a key role in immune defence. The white pulp contains lymphoid tissue organised around central arterioles, allowing immune cells to directly sample blood-borne antigens. This structure enables rapid detection of pathogens circulating in the bloodstream, particularly those not easily cleared by other immune mechanisms. The spleen is therefore especially important for immune responses to organisms that evade phagocytosis unless effectively opsonised.
Clearance of Encapsulated Bacteria
Encapsulated bacteria possess a polysaccharide capsule that inhibits direct phagocytosis. The spleen is uniquely equipped to handle these organisms because it combines efficient antibody production, complement activation, and macrophage-mediated clearance within a single organ. Splenic macrophages and B cells work together to opsonise encapsulated organisms, marking them for destruction. Without a functioning spleen, this coordinated response is impaired, leaving individuals highly vulnerable to overwhelming bloodstream infection.
Blood Reservoir Function and Splenic Contraction
In addition to filtration and immune roles, the spleen acts as a dynamic blood reservoir. It stores a proportion of circulating red blood cells and platelets within its vascular sinusoids. In response to sympathetic stimulation, such as haemorrhage or acute stress, the spleen can contract, expelling stored blood into the circulation. This transiently increases circulating volume and oxygen-carrying capacity. This reservoir function is more prominent in children and diminishes with age as splenic tissue becomes less elastic and vascular responsiveness declines.
Splenomegaly and Functional Consequences
Splenic enlargement occurs when increased demand is placed on its filtering or immune functions. This may result from increased red cell destruction, immune activation, portal hypertension, or infiltration by malignant or infectious processes.
As the spleen enlarges, its filtering capacity increases. While this initially enhances clearance of abnormal cells, it can become maladaptive. Excessive sequestration and destruction of red blood cells, white blood cells, and platelets may occur, a process known as hypersplenism. Hypersplenism reflects overactivity of a normal function, rather than intrinsic splenic failure, and can result in cytopenias despite normal bone marrow production.
Clinical Connections
The spleen plays a central role in immune surveillance, blood filtration, and platelet regulation, which explains why splenic disorders have wide-ranging clinical consequences. Patients who are asplenic or functionally asplenic, such as those with sickle cell disease, are at high risk of overwhelming post-splenectomy infection (OPSI). This syndrome is characterised by rapid progression from mild symptoms to septic shock, most commonly caused by encapsulated organisms such as Streptococcus pneumoniae, Neisseria meningitidis, and Haemophilus influenzae. Prevention relies on:
strict adherence to vaccination schedules
prophylactic antibiotics in selected patients
immediate medical assessment for any fever or flu-like symptoms
Even minor infections can become life-threatening in the absence of splenic immune function.
Splenomegaly (enlargement of the spleen) occurs in a wide range of conditions including haematological malignancies, liver disease, infections, and autoimmune disorders. As the spleen enlarges, it may sequester increasing numbers of red blood cells, white blood cells, and platelets, leading to cytopenias. Patients may develop anaemia, thrombocytopenia, and increased susceptibility to infection as a result of hypersplenism. Clinically, splenomegaly can cause left upper quadrant abdominal discomfort, early satiety due to gastric compression, and a feeling of fullness after eating small amounts.
A ruptured spleen is a surgical emergency, most often following blunt abdominal trauma such as a motor vehicle accident or sporting injury. Because the spleen is highly vascular, rupture can lead to massive internal haemorrhage. Signs may include left-sided abdominal pain, referred pain to the left shoulder (Kehr’s sign), hypotension, tachycardia, and shock. Rapid recognition and escalation of care are critical to survival.
The spleen’s role in filtering blood, mounting immune responses, and regulating circulating blood cells means that structural or functional splenic disease has systemic effects, reinforcing why splenic assessment is an important component of both acute and chronic care.
Concept Check
What roles do the white pulp and red pulp play in splenic function?
Why are patients without a spleen at higher risk of severe bacterial infection?
How does the spleen identify and remove aged or damaged RBCs?
What is hypersplenism, and how does it affect blood cell counts?
Why is splenic rupture considered a medical emergency?