Red Blood Cells and Haemaglobin
Red blood cells (RBCs) are the most abundant cells in the bloodstream and are essential for transporting oxygen from the lungs to tissues and returning carbon dioxide for exhalation. Their structure is elegantly simple, yet highly specialised, allowing them to carry large amounts of haemoglobin while remaining flexible enough to navigate narrow capillaries. Because they are central to oxygen delivery, even small abnormalities in their number, shape, or haemoglobin content can have profound clinical consequences. Understanding the physiology of RBCs allows deeper insight into anaemia, hypoxia, respiratory compensation, and the meaning behind common pathology results.
What You Need to Know
Red blood cells are produced in the bone marrow through a tightly regulated process called erythropoiesis. This process depends on adequate supplies of iron, vitamin B12, folate, amino acids, and the hormone erythropoietin (EPO). EPO is released by the kidneys in response to low oxygen levels in the blood, acting as a key driver of red cell production. When tissues become hypoxic, EPO secretion increases, stimulating the bone marrow to accelerate red blood cell production and restore oxygen delivery.
Newly formed red blood cells are released into the circulation as reticulocytes, which are slightly immature cells that still contain residual cellular material. Over the next one to two days, they complete their maturation in the bloodstream before becoming fully functional erythrocytes. This continuous process allows the body to adapt to changing oxygen demands and maintain adequate tissue oxygenation.
Key features of red blood cells and oxygen transport include:
Red blood cells are produced in the bone marrow in response to erythropoietin (EPO), which increases during hypoxia
Reticulocytes are immature red blood cells that mature in circulation within one to two days
Mature red blood cells have a biconcave shape that maximises surface area and flexibility
They lack a nucleus and organelles, allowing more space for haemoglobin
Haemoglobin contains four haem groups, each capable of binding one oxygen molecule
Red blood cells transport oxygen, assist in carbon dioxide transport, and contribute to acid–base balance
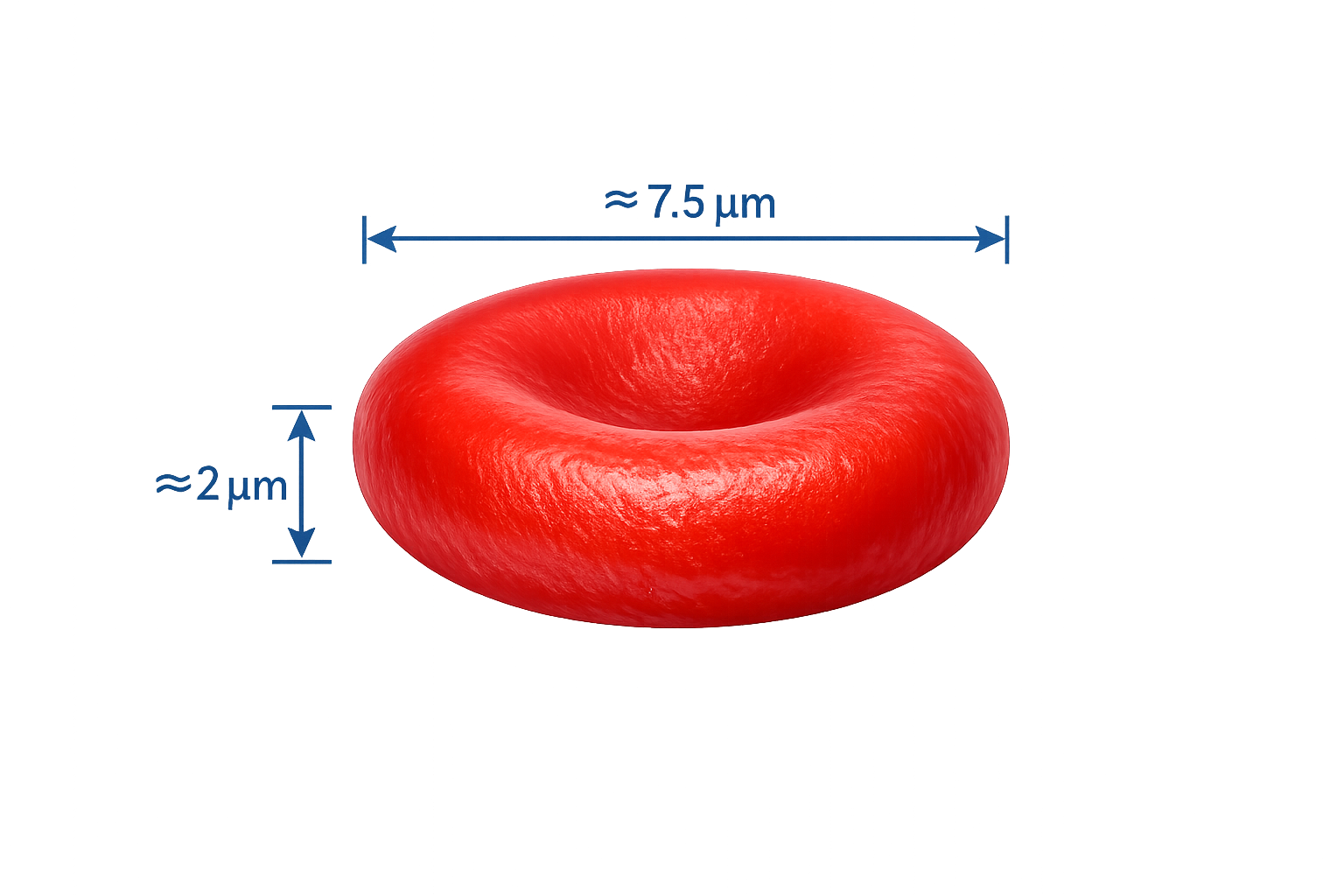
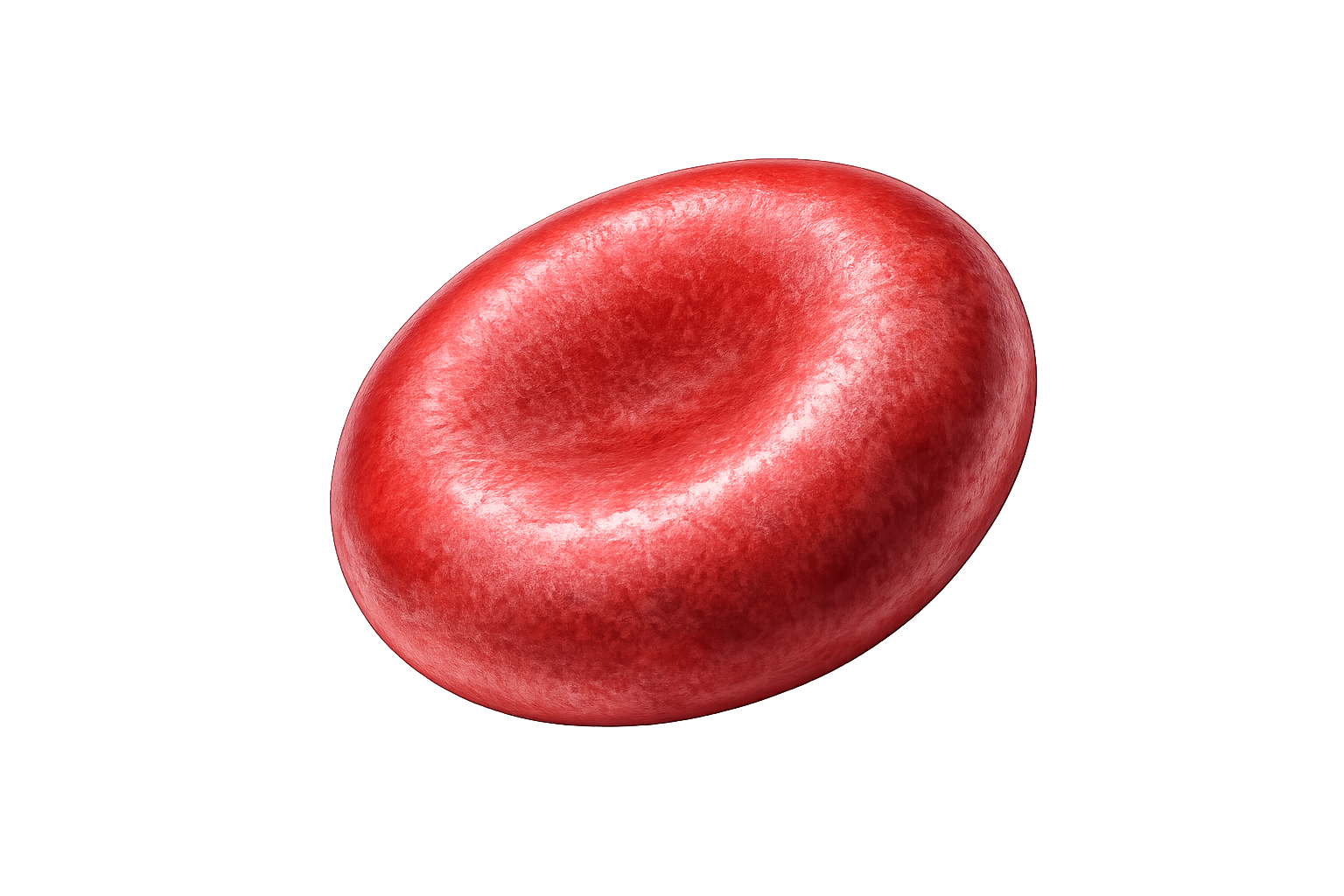
Mature red blood cells have a distinctive biconcave disc shape, thinner in the centre and thicker at the edges, which increases surface area for gas exchange and allows them to deform as they pass through narrow capillaries. This flexibility is essential for efficient oxygen delivery, particularly within the microcirculation where vessel diameter is only slightly larger than the cells themselves.
Haemoglobin, the iron-containing protein that fills most of the red blood cell, is responsible for oxygen transport. Each haemoglobin molecule contains four haem groups, allowing it to bind up to four oxygen molecules at once. In addition to oxygen transport, haemoglobin also carries a portion of carbon dioxide and plays an important role in buffering hydrogen ions, helping to maintain acid–base balance as blood circulates between the lungs and tissues.
Image: A red blood cell is approximately 7–8 µm in diameter, meaning around 10 could fit side by side across the width of a human hair.
Beyond the Basics
Red Blood Cells: Structural Specialisation for Oxygen Transport
Red blood cells are among the most structurally specialised cells in the human body. During maturation they deliberately lose their nucleus and most intracellular organelles, including mitochondria, leaving behind little more than a flexible membrane packed with haemoglobin. This radical simplification creates maximum internal space for oxygen-binding molecules and removes anything that would consume oxygen inside the cell. The result is a circulating carrier whose sole function is to transport gases rather than to maintain its own metabolism.
Because red blood cells lack mitochondria, they generate energy exclusively through anaerobic glycolysis (breaking down glucose into energy (ATP) in the absence of oxygen). This means they do not consume the oxygen they are carrying, preserving it entirely for delivery to tissues. The trade-off is that these cells cannot repair damage, synthesise new proteins, or replace worn membrane components. From the moment they enter circulation, they are slowly accumulating irreversible structural injury.
Red Blood Cell Lifespan and Senescence
A normal red blood cell circulates for around 120 days. Over this time it is repeatedly squeezed through narrow capillaries and exposed to oxidative stress from the very oxygen it transports. These forces gradually damage the lipid membrane and cytoskeletal proteins that give the cell its flexibility. As a result, older red blood cells become stiffer, less deformable, and more prone to mechanical trapping. This loss of flexibility is not accidental. It functions as a biological clock, marking ageing cells for removal. When deformability falls below a critical threshold, red blood cells can no longer navigate the microvasculature efficiently and are flagged for clearance by the spleen.
Splenic Recognition and Controlled Red Cell Removal
The spleen acts as a quality-control organ for circulating blood. Within its red pulp, blood flow slows and red blood cells must squeeze through extremely narrow endothelial slits. Only cells with intact membranes and good deformability can pass through and re-enter circulation. Older or structurally abnormal red blood cells become trapped and are phagocytosed by splenic macrophages.
During this controlled breakdown, haemoglobin is dismantled in a highly organised way. Iron is stripped from the haem groups and transported back to the bone marrow bound to transferrin, where it is reused for new haemoglobin synthesis. The globin proteins are broken down into amino acids, while the haem ring is converted into bilirubin for excretion. This recycling system allows the body to conserve iron and maintain continuous red blood cell production without large dietary losses.
Haemoglobin Structure and Oxygen Binding
Haemoglobin is a complex tetrameric protein made of four globin chains, each containing a haem group with a central ferrous (Fe²⁺) iron atom. Each iron atom can bind one oxygen molecule, allowing a single haemoglobin molecule to carry four oxygen molecules at once. Because each red blood cell contains hundreds of millions of haemoglobin molecules, the oxygen-carrying capacity of blood is enormous.
Oxygen binding is reversible and exquisitely sensitive to the surrounding environment. Haemoglobin is designed to bind oxygen tightly in the lungs, where oxygen pressure is high, and to release it easily in tissues, where oxygen has been consumed. This makes haemoglobin a dynamic transporter rather than a passive storage system.
Image: Haemoglobin is composed of four subunits, each containing a haem group with a central iron (Fe²⁺) ion. Each haem group can bind one oxygen molecule, allowing a single haemoglobin molecule to carry up to four oxygen molecules.
The Oxygen–Haemoglobin Dissociation Relationship
Haemoglobin’s affinity for oxygen shifts depending on local metabolic conditions. In tissues that are metabolically active (tissues with high energy demands, such as the brain, heart, skeletal muscles, kidneys and liver), carbon dioxide levels rise, pH falls (becomes more acidic), and temperature increases. These changes alter haemoglobin’s structure so that it holds oxygen less tightly, allowing more to be released exactly where it is needed. This phenomenon is known as the Bohr effect and explains why oxygen delivery automatically increases in working muscles and inflamed or infected tissues.
In the lungs, the opposite conditions prevail. Carbon dioxide is being removed, pH rises, and temperature is lower, all of which increase haemoglobin’s affinity for oxygen and promote loading. This elegant responsiveness ensures that oxygen transport is matched to physiological demand without any external control.
The Role of 2,3-BPG in Oxygen Release
2,3-bisphosphoglycerate (2,3-BPG) is a metabolite produced inside red blood cells during glycolysis. It binds to haemoglobin and stabilises a low-affinity configuration, encouraging oxygen to be released. When the body experiences sustained hypoxia, such as in anaemia, chronic lung disease, or high-altitude exposure, red blood cells increase their 2,3-BPG production. This adaptive change improves oxygen unloading to tissues even when arterial oxygen content is reduced. This mechanism explains why people can acclimatise to high altitude or chronic anaemia over time. The blood becomes more efficient at releasing oxygen despite limited supply.
Why Haemoglobin Is Essential for Oxygen Transport
Oxygen dissolves very poorly in plasma. Without haemoglobin, the amount of oxygen that could be transported in solution would be far too small to meet metabolic demand. Haemoglobin increases oxygen-carrying capacity by more than seventy-fold, allowing large quantities of oxygen to be transported without dangerously increasing plasma oxygen tension. This design allows blood to remain fluid while still delivering vast amounts of oxygen to every tissue in the body.
Red Blood Cell Shape, Deformability, and Microcirculation
The biconcave shape of red blood cells provides a large surface area for gas exchange and reduces the distance oxygen must diffuse within the cell. Just as importantly, this shape and the flexible cytoskeleton allow red blood cells to deform dramatically as they pass through capillaries that are sometimes narrower than the cells themselves.
When red blood cells lose this flexibility, as occurs in ageing cells or in disorders such as sickle cell disease, microcirculatory flow is impaired. Even if haemoglobin levels appear normal, oxygen delivery falls because red blood cells cannot reach tissues efficiently. This highlights that oxygen transport depends not only on how much haemoglobin is present, but on how effectively red blood cells can move through the microvasculature.
Image: Red Blood Cell- Normal biconcave structure.
Functional Consequences of Abnormal Red Blood Cells
Conditions that alter red blood cell structure, membrane integrity, or haemoglobin composition disrupt oxygen delivery at both the cellular and systemic level. Rigid or misshapen cells increase vascular resistance, promote microvascular obstruction, and reduce tissue perfusion. These effects explain why disorders affecting red blood cell structure or flexibility can produce fatigue, pain, or organ dysfunction disproportionate to measured haemoglobin levels. The problem lies not only in oxygen quantity, but in the ability of red blood cells to deliver it effectively.
Clinical Connections
Disorders of red blood cells are among the most common and clinically significant causes of systemic symptoms. Anaemia occurs when red cell mass, haemoglobin concentration, or both fall below the level needed to maintain adequate oxygen delivery. Patients typically experience fatigue, breathlessness, dizziness, cold extremities, pallor, and reduced exercise tolerance as tissues become relatively hypoxic. In severe or acute anaemia, myocardial oxygen supply may fall enough to cause chest pain, syncope, or heart failure, particularly in those with underlying cardiac disease.
Understanding red blood cell structure and haemoglobin function allows clinicians to interpret laboratory patterns rather than relying on haemoglobin alone. Different forms of anaemia exhibit distinct failures of red cell production, structure, or survival:
Iron deficiency produces small, pale red blood cells (low MCV and low MCH) because haemoglobin synthesis is impaired
Vitamin B12 or folate deficiency causes large, fragile red blood cells (high MCV) due to defective DNA synthesis
Anaemia of chronic disease reflects suppressed erythropoietin signalling and impaired iron mobilisation
Haemolytic anaemias produce elevated bilirubin, reticulocytosis, and shortened red cell lifespan due to premature destruction
Reticulocyte counts are especially valuable because they indicate how actively the bone marrow is responding. A high reticulocyte count suggests ongoing blood loss or haemolysis, while a low count indicates impaired production, such as marrow failure, nutritional deficiency, or reduced erythropoietin.
Polycythaemia results from the opposite physiological problem. Excess red blood cell mass increases blood viscosity, slowing microcirculatory flow and predisposing to thrombosis, stroke, and myocardial infarction. Patients may paradoxically experience symptoms of hypoxia despite elevated haemoglobin because thickened blood cannot deliver oxygen efficiently to tissues.
Red blood cell physiology provides the framework for interpreting pathology results, guiding transfusion decisions, and identifying the underlying cause of abnormal blood counts. Recognising whether a disorder reflects impaired production, abnormal structure, or altered survival is essential for effective diagnosis and long-term management.
Concept Check
How does erythropoietin regulate red blood cell production?
Why does the biconcave shape of RBCs enhance gas exchange?
What factors shift haemoglobin’s ability to bind or release oxygen?
How do iron deficiency and B12 deficiency differ in their effects on RBC morphology?
Why does polycythaemia increase the risk of thrombosis?