Microcirculation and Starling Forces
Microcirculation refers to the movement of blood through the smallest vessels- arterioles, capillaries, and venules, where exchange of oxygen, nutrients, hormones, and waste products occurs. This level of circulation is critical to maintaining cellular function and supporting tissue health. Understanding microcirculation requires knowledge of Starling forces, the principles governing how fluid shifts across capillary walls. These forces determine whether fluid remains in the bloodstream or moves into the interstitial space, influencing blood volume, oedema formation, and tissue perfusion. Interpreting microcirculatory changes is essential when caring for patients with shock, heart failure, malnutrition, burns, and sepsis.
What You Need to Know
The microcirculation consists of a branching network of arterioles that regulate flow into capillary beds. Capillaries are thin-walled, consisting of a single layer of endothelial cells designed for efficient diffusion. Blood flows slowly through capillaries, allowing time for exchange. On the venous side, venules collect deoxygenated blood and return it to larger veins for transport back to the heart.
Starling forces describe the movement of fluid between the blood and interstitial space. Two opposing pressures drive this exchange: hydrostatic pressure (the force pushing fluid out of the capillaries) and oncotic pressure (the “pulling” force exerted by large plasma proteins), largely generated by plasma proteins such as albumin, which draws fluid back in.
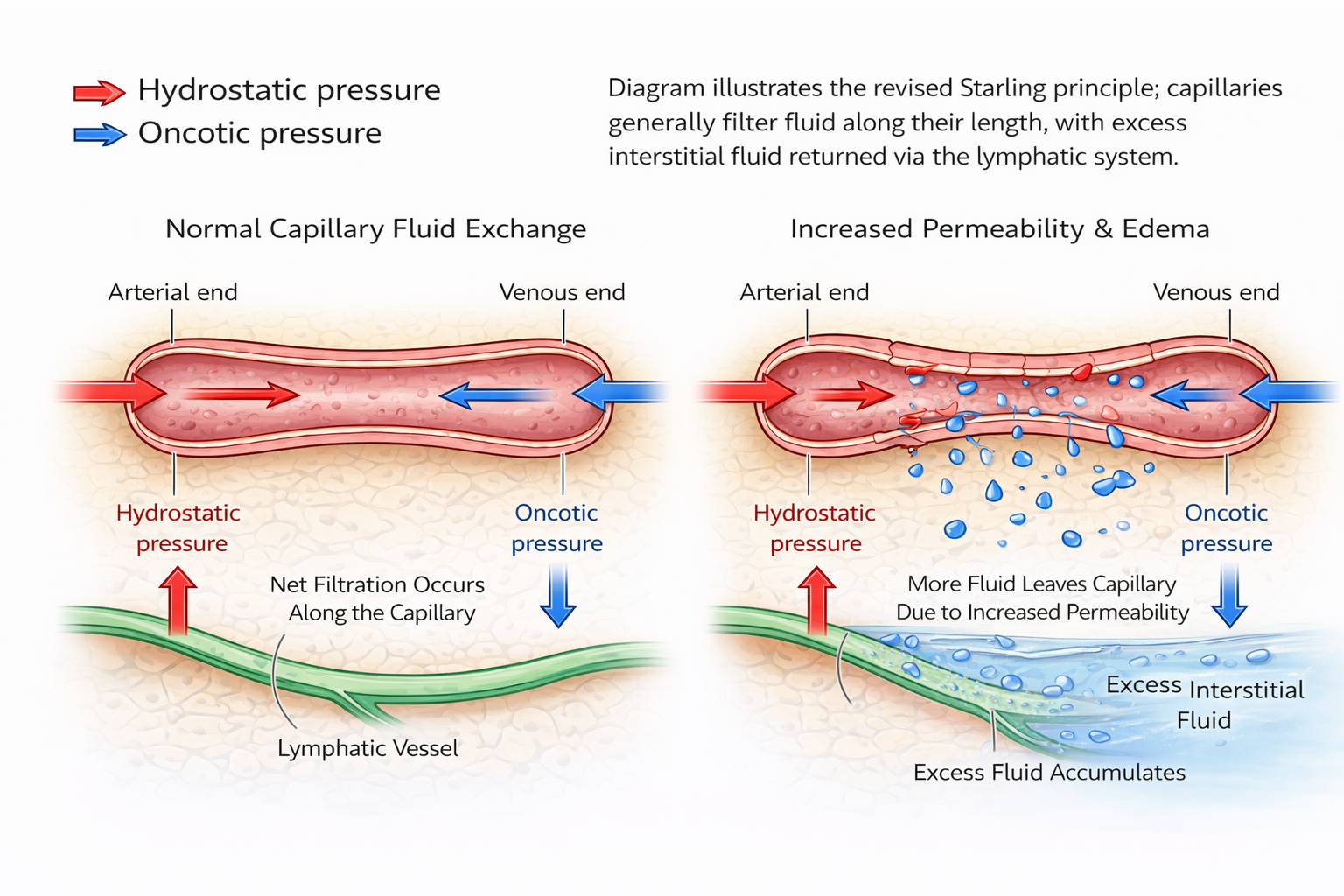
At the arterial end of a capillary, hydrostatic pressure is higher and fluid is pushed into the tissues. At the venous end, oncotic pressure is stronger and fluid returns to the bloodstream. This balance ensures that tissues receive nutrients without accumulating excess fluid.
Image: Capillary fluid exchange. Fluid is filtered from capillaries into the interstitial space, with excess returned via the lymphatic system.
Beyond the Basics
Starling Forces and Capillary Exchange
Starling forces govern the movement of fluid across the capillary wall and reflect the balance between hydrostatic and oncotic pressures within the microcirculation. Under normal conditions, capillary hydrostatic pressure drives fluid out of the capillary at the arterial end, while plasma oncotic pressure generated largely by albumin promotes reabsorption at the venous end. This dynamic equilibrium allows continuous exchange of fluid while preventing excessive accumulation in the interstitial space.
Capillary permeability is a critical modifier of these forces. The endothelial barrier normally restricts the movement of plasma proteins, preserving oncotic pressure within the capillary. When endothelial integrity is disrupted, permeability increases and proteins leak into the interstitial fluid. This reduces the oncotic gradient that would normally draw fluid back into the capillary, shifting the balance toward net filtration and promoting oedema formation.
Endothelial Dysfunction and Increased Permeability
Inflammation profoundly alters endothelial function. Inflammatory mediators such as histamine, cytokines, and bradykinin cause endothelial cells to contract or reorganise their junctions, creating gaps through which fluid and proteins can escape. This response is protective in localised inflammation, allowing immune cells and plasma proteins to access injured tissue, but becomes pathological when widespread or sustained.
In systemic inflammatory states such as sepsis or extensive burns, this increased permeability occurs throughout the circulation. The resulting loss of plasma proteins into the interstitial space reduces effective circulating oncotic pressure, leading to diffuse oedema and intravascular volume depletion. Despite adequate total body fluid, effective perfusion pressure falls, compromising tissue oxygen delivery.
Oncotic Pressure and Plasma Protein Balance
Plasma oncotic pressure is determined primarily by albumin concentration. Albumin’s relatively small size and high abundance make it the dominant contributor to colloid osmotic pressure. When albumin levels fall, the inward-pulling force opposing capillary filtration is reduced, favouring movement of fluid into tissues.
Hypoalbuminaemia therefore shifts Starling forces toward net filtration even when capillary permeability is normal. In this setting, oedema develops not because hydrostatic pressure is elevated, but because reabsorptive forces are insufficient to return fluid to the circulation. This illustrates how alterations in plasma protein composition alone can significantly disrupt fluid balance.
Venous Pressure and Impaired Reabsorption
Venous pressure plays an important role in determining capillary hydrostatic pressure at the venous end of the capillary bed. When venous pressure is elevated, capillary hydrostatic pressure remains high along the entire length of the capillary. This reduces or eliminates the normal reabsorptive phase of capillary exchange.
In conditions such as heart failure, sustained elevation of venous pressure promotes continuous filtration of fluid into the interstitial space. Because reabsorption is impaired, excess fluid accumulates preferentially in dependent tissues where venous pressures are highest. This mechanism highlights how changes downstream of the capillary can profoundly affect microvascular fluid dynamics.
Lymphatic Drainage and Fluid Homeostasis
The lymphatic system provides an essential safety mechanism for maintaining interstitial fluid balance. It returns excess filtered fluid and escaped plasma proteins to the venous circulation, preventing progressive fluid accumulation. Under normal conditions, lymphatic flow increases in response to elevated interstitial pressure, compensating for increased capillary filtration.
When lymphatic drainage is impaired, this compensatory mechanism fails. Obstruction, damage, or congenital absence of lymphatic vessels prevents removal of interstitial fluid and proteins, leading to chronic lymphoedema. Because proteins remain trapped in the interstitial space, oncotic pressure within tissues rises, drawing in additional fluid and perpetuating swelling.
Microcirculatory Failure and Shock States
Microcirculatory dysfunction plays a central role in shock, particularly distributive forms such as septic shock. In sepsis, widespread endothelial activation increases permeability while simultaneously disrupting normal vasoregulatory responses. Fluid shifts from the intravascular to the interstitial compartment, reducing effective circulating volume despite aggressive fluid administration.
At the same time, maldistribution of blood flow at the capillary level impairs oxygen extraction by tissues. Even when global blood pressure appears adequate, cellular hypoxia may persist due to failure of effective microvascular perfusion. These changes illustrate how disruption of Starling forces and endothelial function can directly contribute to organ dysfunction.
Integration of Capillary Exchange Mechanisms
Fluid balance at the tissue level depends on the coordinated interaction between capillary pressures, endothelial permeability, plasma protein concentration, venous drainage, and lymphatic return. Alteration in any one of these components shifts Starling forces and predisposes to oedema formation.
Understanding these mechanisms provides a framework for appreciating why oedema can arise from diverse pathophysiological processes and why restoring circulating volume alone may not correct microvascular dysfunction.
Clinical Connections
Microcirculatory and Starling force disturbances result in many of the most common fluid balance problems seen in clinical practice. Oedema develops whenever filtration out of capillaries exceeds reabsorption back into them, and this can occur through several different mechanisms depending on the disease process.
In heart failure, raised venous pressure increases capillary hydrostatic pressure, pushing fluid out into tissues. This explains dependent leg oedema in ambulant patients and sacral oedema in bed-bound patients, as well as pulmonary oedema when the same process occurs in the lungs. Diuretics, venodilators, and positive pressure ventilation all improve symptoms by lowering capillary pressure and restoring fluid reabsorption.
In liver disease, reduced albumin synthesis lowers plasma oncotic pressure, weakening the force that normally pulls fluid back into the circulation. Even if hydrostatic pressure is normal, fluid leaks into the peritoneal cavity and tissues, producing ascites and peripheral oedema. Giving albumin or restricting sodium directly targets this imbalance in Starling forces.
In burns, sepsis, and major trauma, capillary permeability increases due to endothelial injury. Fluid and plasma proteins escape into the interstitium, causing dramatic intravascular volume loss despite total body fluid overload. This is why these patients may be swollen but hypotensive at the same time, and why early aggressive IV fluid replacement is lifesaving.
At the bedside, impaired microcirculation presents with signs of poor tissue perfusion:
cool or mottled skin
delayed capillary refill
reduced urine output
altered mental state
rising lactate
An understanding Starling forces helps to interpret why a patient may be oedematous yet intravascularly depleted, and why fluid, albumin, diuretics, or vasopressors are chosen in different clinical scenarios.
Concept Check
Explain how hydrostatic and oncotic pressures influence fluid movement in capillaries.
Why does oedema occur in patients with hypoalbuminaemia?
How does heart failure alter Starling forces?
What happens to microcirculation during sepsis?
Why is the lymphatic system essential for fluid balance?