Adipose Tissue as an Endocrine Organ: Leptin, Adiponectin and Metabolic Signalling
Adipose tissue is no longer regarded solely as an inert energy storage site. It is now recognised as a dynamic endocrine organ that secretes a wide range of hormones, known as adipokines, which influence appetite regulation, insulin sensitivity, inflammation, vascular health and reproductive function. Through these hormones, adipose tissue communicates metabolic status to the brain, liver, pancreas, skeletal muscle and immune system. This endocrine activity is central to understanding obesity, metabolic syndrome and the pathophysiology of insulin resistance. The endocrine functions of adipose tissue highlight how profoundly energy balance impacts whole-body physiology.
What You Need to Know
Adipose tissue is now recognised as an active endocrine organ rather than a passive energy store. It secretes multiple hormones and cytokines—collectively termed adipokines—that influence appetite regulation, insulin sensitivity, inflammation and cardiovascular risk. Through these signals, adipose tissue communicates nutritional status to the brain, liver, muscle and immune system.
Key endocrine products of adipose tissue include:
Leptin, which signals energy sufficiency to the hypothalamus and suppresses appetite
Adiponectin, which enhances insulin sensitivity and has anti-inflammatory and anti-atherogenic effects
Resistin and pro-inflammatory cytokines (eg TNF-α, IL-6), which impair insulin signalling and promote metabolic dysfunction
Leptin secretion rises with increasing fat mass, acting as a long-term regulator of energy balance rather than a meal-to-meal satiety signal. In contrast, adiponectin levels fall as adiposity increases, removing a key protective influence on glucose metabolism and vascular health. This inverse relationship helps explain why obesity is associated with insulin resistance, type 2 diabetes and cardiovascular disease.
Because adipose endocrine output changes with both excess fat and severe fat loss, disorders such as obesity, lipodystrophy, starvation and eating disorders all produce characteristic metabolic disturbances. Adipose tissue therefore functions as a dynamic endocrine sensor, linking nutritional state directly to whole-body metabolic regulation.
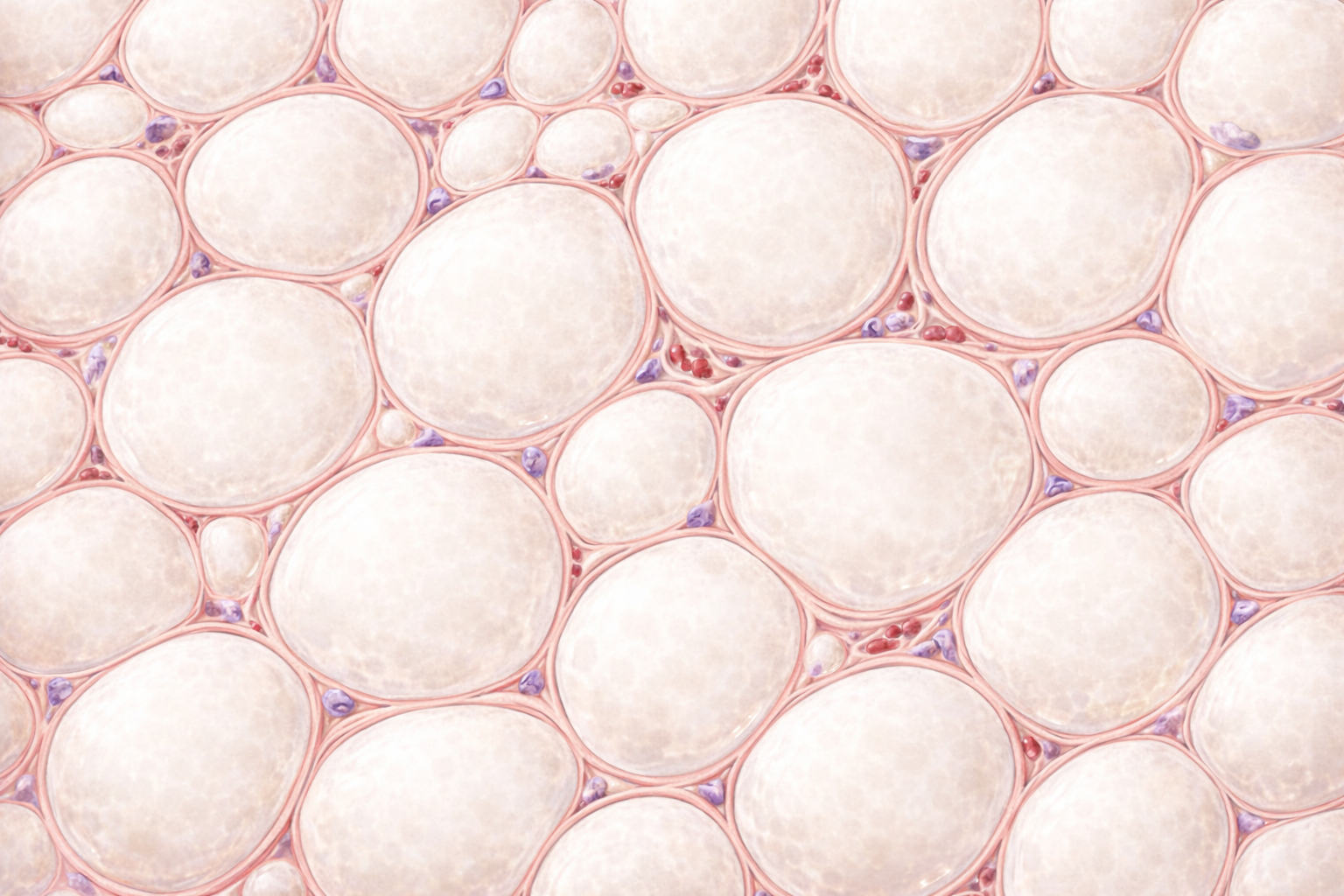
Image: Adipose tissue is composed of large adipocytes containing a single lipid droplet, which appears clear on histology. The nucleus is compressed and displaced to the cell periphery, giving each cell a characteristic signet ring appearance.
Beyond the Basics
Structural and Cellular Composition of Adipose Tissue
Adipose tissue is not a passive fat store but a highly active endocrine organ composed of multiple interacting cell types. In addition to adipocytes, adipose tissue contains immune cells, fibroblasts, endothelial cells and pre-adipocytes. This diverse cellular environment allows adipose tissue to respond dynamically to nutritional status, inflammation and hormonal signals, producing a complex endocrine output that changes with metabolic state.
White adipose tissue is the primary endocrine form and the main site of triglyceride storage. It is responsible for secreting most adipokines, including leptin, adiponectin and inflammatory mediators. Brown adipose tissue, although less abundant in adults, has a specialised role in thermogenesis. It generates heat through mitochondrial uncoupling and releases signalling molecules that influence whole-body energy expenditure, linking adipose tissue to temperature regulation and metabolic rate.
Leptin: Regulator of Appetite and Energy Balance
Leptin is produced by adipocytes in proportion to fat mass and serves as a long-term signal of energy sufficiency. It acts primarily on receptors in the hypothalamus to suppress appetite, increase energy expenditure and coordinate reproductive and thyroid hormone activity. In physiological conditions, rising leptin levels signal adequate energy stores and reduce food intake, while falling levels signal energy deficiency.
In obesity, leptin levels are chronically elevated, yet appetite suppression fails to occur. This reflects leptin resistance, a state in which central nervous system responsiveness to leptin is impaired. As a result, satiety signalling becomes ineffective despite abundant energy stores. In contrast, during starvation or significant weight loss, declining leptin levels suppress reproductive hormone secretion, reduce thyroid hormone activity and shift metabolism toward energy conservation, prioritising survival over growth or reproduction.
Adiponectin: Enhancer of Insulin Sensitivity
Adiponectin is secreted predominantly by adipocytes but displays an inverse relationship with fat mass. As adipose tissue expands, adiponectin secretion declines. This hormone enhances insulin sensitivity by promoting fatty acid oxidation, reducing hepatic glucose production and improving endothelial function, thereby supporting efficient glucose utilisation and vascular health.
High adiponectin levels are associated with reduced cardiovascular risk and metabolic stability, whereas low levels are characteristic of obesity, type 2 diabetes and metabolic syndrome. Its anti-inflammatory properties counteract the chronic low-grade inflammation that accompanies excess adiposity, highlighting its protective role in metabolic regulation.
Resistin and Pro-inflammatory Cytokines
As adipose tissue expands, immune cell infiltration increases and endocrine signalling shifts toward a pro-inflammatory profile. Resistin, along with cytokines such as tumour necrosis factor-alpha and interleukin-6, is produced by both adipocytes and resident immune cells. These mediators impair insulin signalling, reduce glucose uptake in peripheral tissues and promote systemic inflammation.
This inflammatory endocrine output provides a mechanistic link between obesity and insulin resistance, cardiovascular disease and metabolic dysfunction. Rather than being driven solely by excess energy storage, metabolic disease reflects altered hormonal and immune signalling originating within adipose tissue itself.
Endocrine Cross-Talk With Other Organs
Adipose-derived hormones exert widespread effects on multiple organ systems. Leptin influences pancreatic insulin secretion, reproductive hormone release and hypothalamic energy regulation. Adiponectin acts on the liver and skeletal muscle to enhance metabolic flexibility, improving the body’s ability to switch between glucose and lipid fuel sources.
Through these endocrine interactions, adipose tissue integrates nutritional status with whole-body metabolism, immune activity and vascular function. Disruption of this signalling network—whether through excess or deficient adiposity—has profound systemic consequences, reinforcing the role of adipose tissue as a central regulator of metabolic homeostasis.
Clinical Connections
Endocrine dysfunction of adipose tissue contributes to a wide range of metabolic and cardiovascular conditions. Adipose tissue is not merely a passive energy store but a hormonally active organ that communicates with the brain, liver, pancreas, muscle and vasculature. When this signalling becomes dysregulated, systemic metabolic control deteriorates.
In obesity, expanding adipose tissue undergoes structural and functional changes that alter its endocrine output. Leptin levels rise in proportion to fat mass, but chronic elevation leads to leptin resistance at hypothalamic receptors, blunting satiety signalling and perpetuating excess energy intake. At the same time, adiponectin secretion falls, removing an important protective influence on insulin sensitivity, endothelial function and inflammation.
Key endocrine shifts in dysfunctional adipose tissue include:
impaired leptin signalling despite high circulating leptin levels
reduced adiponectin availability, worsening insulin resistance
increased secretion of pro-inflammatory cytokines such as TNF-α and IL-6
These changes create a chronic low-grade inflammatory state that links obesity with type 2 diabetes, hypertension and cardiovascular disease, often years before overt hyperglycaemia develops. Metabolic disease therefore reflects qualitative dysfunction of adipose signalling rather than fat mass alone.
Lipodystrophy provides a powerful contrast that reinforces this concept. In the absence of adequate adipose tissue, leptin and adiponectin levels are profoundly low, leading to severe insulin resistance, hepatic steatosis and dyslipidaemia despite minimal or absent body fat. This demonstrates that normal metabolic regulation requires functioning adipose endocrine signalling, not simply reduced adiposity.
Therapeutically, this understanding has reshaped metabolic medicine. Leptin replacement improves glucose and lipid metabolism in lipodystrophy, while strategies aimed at improving adipose tissue function, reducing inflammation or enhancing adiponectin signalling are active areas of research in obesity-related disease.
Concept Check
Why is adipose tissue considered an endocrine organ rather than just an energy store
How does leptin regulate appetite and why does leptin resistance occur in obesity
Why is adiponectin protective against metabolic disease
How do inflammatory cytokines produced by adipose tissue contribute to insulin resistance
How does adipose–organ cross-talk influence metabolic homeostasis