The Pineal Gland: Melatonin Secretion, Circadian Rhythms and Neuroendocrine Regulation
The pineal gland is a small, cone-shaped endocrine organ located near the centre of the brain, between the superior colliculi. Although modest in size, it plays a crucial role in regulating the body’s circadian rhythms through the secretion of melatonin. Acting as a neuroendocrine transducer, the pineal gland converts neural signals related to light exposure into hormonal output that influences sleep–wake cycles, seasonal physiology and reproductive function. Its intimate connection with the visual system and autonomic pathways highlights its role as an environmental timekeeper within the endocrine network.
What You Need to Know
The pineal gland is a small neuroendocrine structure that plays a central role in translating environmental light–dark signals into hormonal rhythms. Its primary hormone, melatonin, is synthesised from serotonin and secreted in a circadian pattern tightly linked to darkness. Secretion increases shortly after sunset, peaks during the night and falls toward morning, creating a hormonal signal of “biological night” for the entire body.
Light exposure suppresses melatonin release through a precisely organised neural pathway. Photoreceptors in the retina transmit signals to the suprachiasmatic nucleus (SCN) of the hypothalamus, the body’s central circadian clock. From the SCN, signals travel through the brainstem and spinal cord to sympathetic fibres innervating the pineal gland. When light is present, this pathway inhibits melatonin synthesis; when darkness falls, inhibition is removed and melatonin secretion rises.
Melatonin acts widely despite being produced in small amounts. Its key physiological roles include:
synchronising circadian rhythms across organ systems
promoting sleep onset and regulating sleep–wake timing
modulating reproductive hormone secretion, particularly gonadotropins
exerting antioxidant and neuroprotective effects
Through these actions, melatonin helps align metabolism, hormone secretion, immune function and behaviour with the external light–dark cycle. Although melatonin does not drive acute metabolic changes in the way hormones such as cortisol or insulin do, it provides essential temporal organisation, ensuring that physiological processes occur at the most appropriate time of day.
Disruption of melatonin signalling—through shift work, jet lag, excessive nighttime light exposure or neurological disease—leads to circadian misalignment, contributing to sleep disorders, metabolic dysregulation and altered neuroendocrine function.
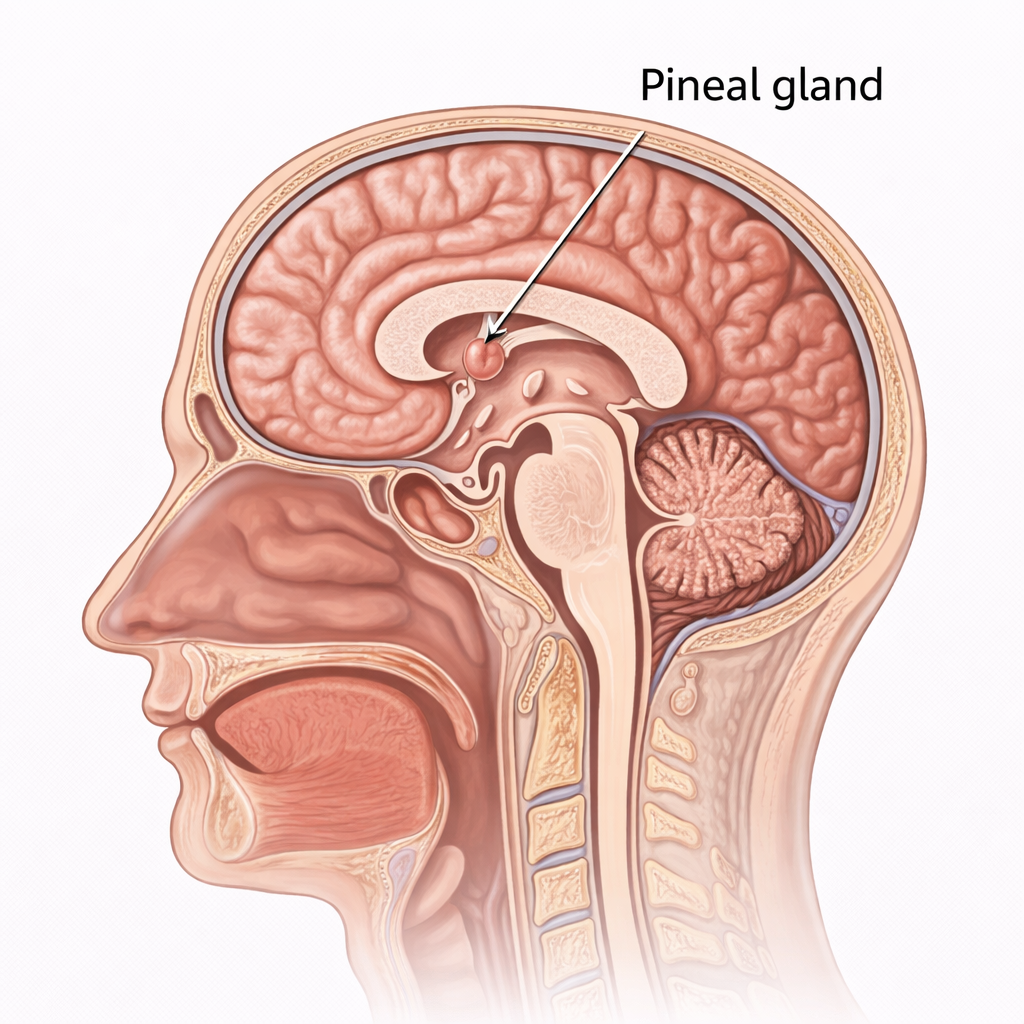
Image: The pineal gland is a small endocrine structure located deep within the brain, positioned near the midline between the two hemispheres, just posterior to the third ventricle.
Beyond the Basics
Structural Organisation
The pineal gland consists of pinealocytes, the primary hormone-secreting cells, intermixed with glial cells and supported by connective tissue septa. Pinealocytes contain abundant secretory vesicles and long cytoplasmic processes that extend toward nearby capillaries, allowing rapid diffusion of melatonin into the circulation. With increasing age, calcified deposits known as corpora arenacea (“brain sand”) accumulate within the gland. Although their physiological function is unclear, they are clinically useful as radiographic landmarks for identifying pineal position.
Neural Control of Melatonin Secretion
Melatonin secretion is regulated by a precisely organised neural pathway known as the retinohypothalamic–pineal tract. Light detected by specialised retinal ganglion cells is transmitted to the suprachiasmatic nucleus, the brain’s central circadian pacemaker. From there, signals descend through the spinal cord and synapse in the superior cervical ganglion, whose sympathetic fibres innervate the pineal gland.
In darkness, sympathetic activity increases, stimulating pinealocytes to convert serotonin into melatonin via the rate-limiting enzyme N-acetyltransferase. Exposure to light suppresses this pathway, rapidly reducing melatonin production. This neural–endocrine arrangement ensures that melatonin secretion accurately reflects environmental light–dark conditions rather than internal metabolic state.
Physiological Actions of Melatonin
Melatonin functions primarily as a circadian timing signal rather than a fast-acting regulatory hormone. Its key effects include:
signalling “biological night” to peripheral tissues
promoting sleep onset and regulating sleep–wake timing
influencing core body temperature and alertness
modulating reproductive hormone secretion
providing antioxidant and immunomodulatory protection
Through these actions, melatonin synchronises endocrine, metabolic and behavioural processes with the external day–night cycle. While its reproductive effects are pronounced in seasonally breeding animals, in humans they are more subtle and are most evident during early development and puberty.
Age and Environment-Related Changes
Melatonin secretion is highest in childhood and declines progressively with age, contributing to lighter, more fragmented sleep in older adults. Environmental factors such as artificial light exposure, night-shift work and rapid travel across time zones disrupt normal melatonin rhythms, leading to circadian misalignment, insomnia and fatigue. Over time, persistent disruption of melatonin signalling is associated with metabolic disturbance, impaired immune regulation and altered neuroendocrine function.
Clinical Connections
The clinical relevance of the pineal gland reflects both its anatomical position and its role in circadian regulation. Structural abnormalities such as tumours, cysts or excessive calcification can impair melatonin secretion or compress adjacent midbrain structures. Pineal tumours may present with disrupted sleep–wake cycles, impaired upward gaze due to compression of the dorsal midbrain (Parinaud syndrome), or obstructive hydrocephalus if cerebrospinal fluid flow through the cerebral aqueduct is compromised.
Melatonin dysregulation is also clinically significant even in the absence of overt structural disease and commonly presents as:
• difficulty initiating or maintaining sleep
• circadian rhythm misalignment (eg shift work or jet lag)
• age-related sleep fragmentation due to reduced melatonin secretion
Reduced endogenous melatonin production contributes to insomnia and circadian rhythm sleep disorders, while inappropriate timing of melatonin release is central to jet lag and shift-work disorder.
Exogenous melatonin is widely used in clinical practice, but its effectiveness depends on correct timing rather than dose alone. Supplementation taken too early or too late can worsen circadian misalignment instead of correcting it. Understanding normal pineal physiology explains why melatonin acts as a time cue rather than a sedative and why light exposure, especially blue light in the evening, can counteract its effects.
Concept Check
How does the pineal gland convert light exposure into hormonal signals
Why is melatonin secretion highest at night and inhibited by light
What structural features of pinealocytes support their role in hormone secretion
What physiological processes outside sleep regulation are influenced by melatonin
How can environmental light exposure disrupt endocrine timing mechanisms