Endocrine Pancreas: Islet Structure, Cell Types and Microvascular Organisation
The endocrine pancreas plays an essential role in maintaining metabolic homeostasis. Its hormone-producing cells are organised into clusters called the islets of Langerhans, which regulate blood glucose, nutrient storage and energy availability. These small but highly specialised structures operate as an integrated network, allowing rapid, coordinated hormonal responses to changing metabolic demands. Because insulin, glucagon, somatostatin and pancreatic polypeptide influence nearly every organ system, even minor disruptions to islet function can lead to profound clinical consequences such as diabetes mellitus and metabolic instability.
What You Need to Know
The endocrine pancreas consists of clusters of hormone-secreting cells known as pancreatic islets, which are dispersed throughout the exocrine pancreas. These islets play a central role in regulating blood glucose and overall metabolic balance by sensing circulating nutrients and releasing hormones directly into the bloodstream. Their structure is specialised to allow rapid detection of changes in blood glucose and coordinated hormone secretion.
Several key cell types work together within each islet:
Beta cells, which secrete insulin
Alpha cells, which secrete glucagon
Delta cells, which secrete somatostatin
PP cells, which secrete pancreatic polypeptide
Although the exact arrangement varies between species, beta cells are typically located toward the centre of the islet, with alpha and other cell types more peripherally distributed. This organisation supports close communication between cells, allowing hormones to modulate each other’s release and maintain glucose within a narrow physiological range. Delta cell–derived somatostatin, for example, acts locally to dampen both insulin and glucagon secretion, preventing excessive swings in blood glucose.
Pancreatic islets are supplied by a dense and highly specialised microvascular network that delivers glucose rapidly to endocrine cells and carries secreted hormones efficiently into the circulation. Blood flow through the islet often follows a defined pattern, exposing different cell populations sequentially to changing glucose and hormone levels. This microvascular organisation reinforces tight functional integration within the islet and underpins the precise, moment-to-moment regulation of glucose and metabolic homeostasis.
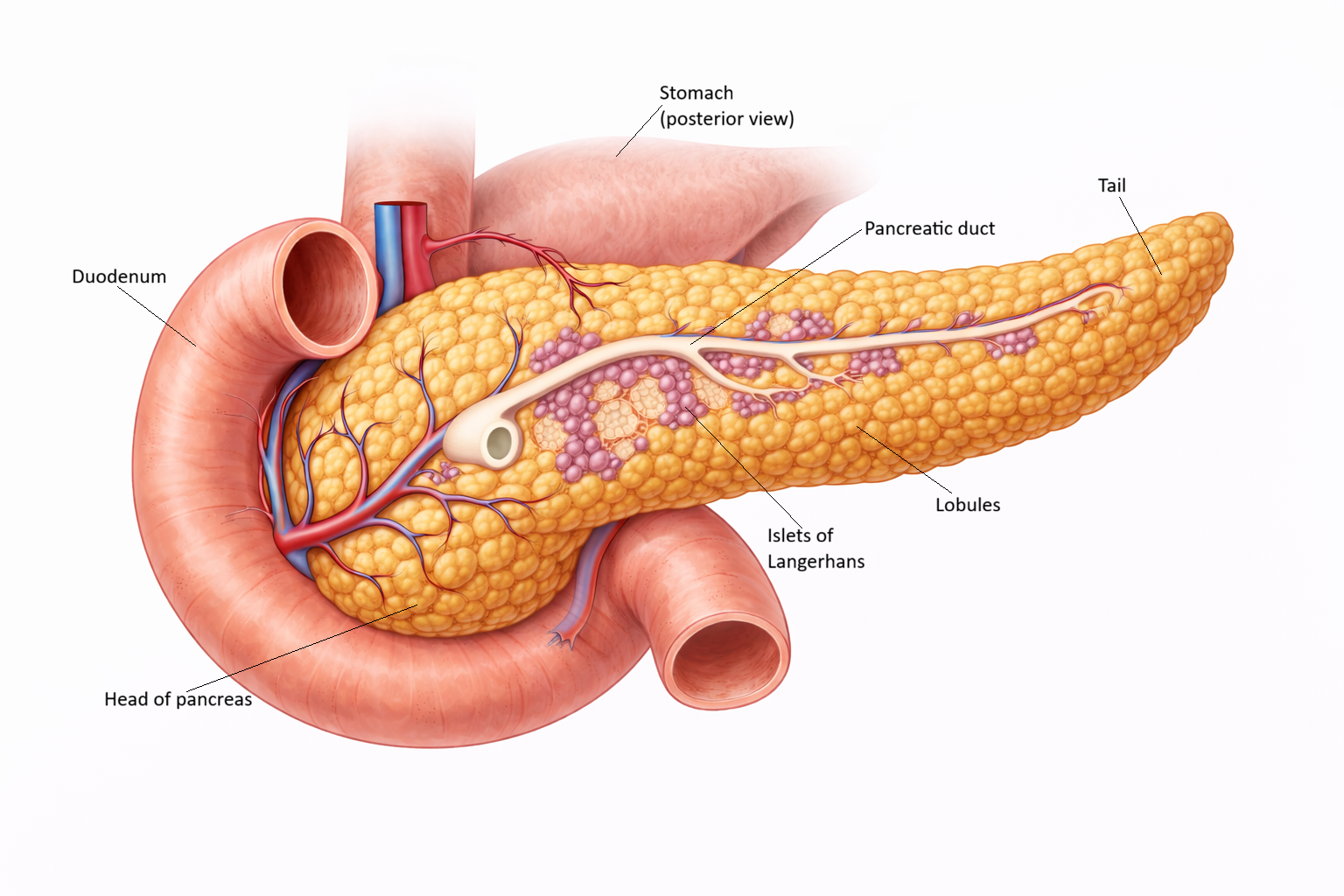
Image: The pancreas lies within the curve of the duodenum and has both exocrine and endocrine functions. Exocrine tissue produces digestive enzymes, while scattered islets of Langerhans secrete hormones such as insulin and glucagon.
Beyond the Basics
Islet microanatomy and cellular arrangement
Each pancreatic islet is composed of tightly packed endocrine cells supported by a fine reticular connective tissue framework and enclosed by a thin capsule. In humans, beta cells are the dominant cell type, typically accounting for around half to two-thirds of the total islet cell population. Alpha cells make up a substantial minority, while delta and pancreatic polypeptide–secreting cells are present in smaller numbers. This cellular composition reflects the central role of insulin in glucose regulation, while still allowing for counter-regulatory and modulatory influences.
The spatial organisation of cells within the islet is functionally significant. Blood flow generally enters through beta cell–rich central regions before passing toward the periphery, exposing alpha and delta cells to high local concentrations of insulin and other beta cell–derived peptides. This directional flow creates an internal hierarchy of hormonal signalling, where insulin can exert inhibitory effects on glucagon secretion before glucagon enters the systemic circulation. The arrangement supports fine control of hormone release and helps stabilise blood glucose levels during changing metabolic conditions.
Vascular and neural supply
Pancreatic islets receive a disproportionately high blood supply relative to their size, often several times greater than that of the surrounding exocrine tissue. This dense microvascular network allows endocrine cells to sense circulating glucose rapidly and ensures that secreted hormones are delivered efficiently into the bloodstream. The close association between capillaries and endocrine cells minimises diffusion distance and supports rapid, moment-to-moment regulation of glucose homeostasis.
In addition to their vascular specialisation, islets receive autonomic innervation from both sympathetic and parasympathetic fibres. Sympathetic activation tends to suppress insulin secretion and promote glucagon release, supporting glucose availability during stress or fasting. Parasympathetic input, particularly during feeding, enhances insulin secretion in anticipation of rising blood glucose. This neural control links islet hormone release to behavioural state, nutrient intake, and energy demand.
Intercellular communication within the islet
Islet function depends not only on individual hormone secretion but also on continuous communication between neighbouring cells. Beta cells are electrically coupled through gap junctions, allowing coordinated depolarisation and synchronous insulin release across the islet. This coupling ensures that insulin secretion occurs as a unified response rather than as isolated activity from individual cells.
Paracrine signalling further refines hormone output. Insulin released from beta cells suppresses glucagon secretion from alpha cells, while glucagon can enhance insulin release under certain conditions, creating a balanced feedback relationship. Somatostatin released from delta cells acts as a local inhibitor of both insulin and glucagon secretion, preventing excessive hormonal swings. Together, these interactions allow the islet to function as an integrated regulatory unit rather than a collection of independent endocrine cells.
Functional integration with the exocrine pancreas
Although the endocrine and exocrine pancreas serve different primary functions, they are closely integrated anatomically and functionally. Hormones released from islets influence exocrine pancreatic activity, including enzyme secretion and local blood flow. At the same time, digestive activity and nutrient absorption generate neural and hormonal signals that feed back to the islets, shaping endocrine responses after meals.
This bidirectional communication helps coordinate digestion, nutrient handling, and glucose regulation as part of a unified metabolic response. Rather than operating in isolation, the endocrine pancreas is embedded within a broader digestive and metabolic network, allowing precise adjustment of hormone secretion to match nutritional state and energy availability.
Clinical Connections
Damage to pancreatic islets disrupts glucose regulation in predictable ways because each islet cell type contributes to maintaining stable blood glucose. Clinical presentations often reflect which cell population is affected and whether hormone deficiency, excess, or impaired responsiveness is the dominant problem. Linking symptoms and laboratory findings back to islet structure helps explain both disease progression and treatment strategies.
In clinical practice, islet dysfunction is most commonly encountered in the following patterns:
Loss of insulin-producing beta cells
Progressive beta-cell dysfunction with insulin resistance
Hormone excess due to islet cell tumours
Autoimmune destruction of beta cells leads to type 1 diabetes mellitus, resulting in absolute insulin deficiency. Without insulin, glucose cannot be effectively taken up by tissues, leading to hyperglycaemia and reliance on fat metabolism for energy. This explains the risk of rapid metabolic decompensation and the need for lifelong insulin replacement. In contrast, type 2 diabetes develops more gradually, as insulin resistance increases and beta cells become unable to meet rising insulin demands. Over time, declining beta-cell function contributes to worsening glycaemic control despite initially preserved insulin secretion.
Islet cell tumours produce clinical syndromes that reflect the hormone secreted by the affected cell type. Insulinomas cause inappropriate insulin release and recurrent hypoglycaemia, while glucagonomas and somatostatinomas produce metabolic disturbances linked to excess glucagon or somatostatin. These conditions highlight the normally tight paracrine balance within the islet and the systemic consequences when that balance is lost.
Understanding islet architecture also informs modern therapeutic approaches. Treatments such as islet transplantation aim to restore functional beta-cell mass, while incretin-based therapies enhance glucose-dependent insulin secretion by acting on intact islet signalling pathways. Closed-loop insulin delivery systems attempt to replicate physiological insulin release patterns, reinforcing how closely clinical management is tied to the normal structure and function of pancreatic islets.
Concept Check
How does the vascular organisation of pancreatic islets support rapid metabolic regulation
Why does the spatial arrangement of beta and alpha cells enhance hormonal control of glucose
How do paracrine interactions within the islet stabilise blood glucose levels
Why does autonomic innervation play a key role in islet hormone secretion
How does islet damage lead to the clinical manifestations of diabetes mellitus