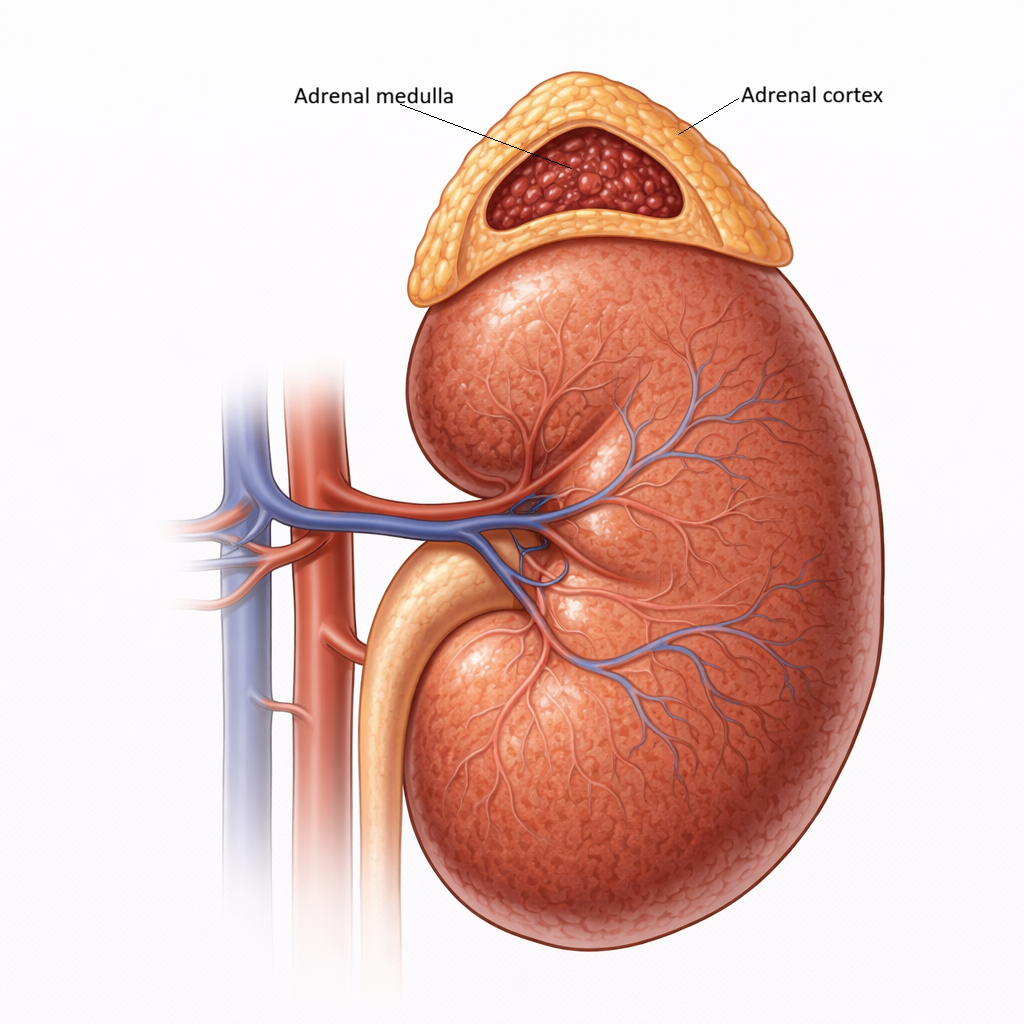
The Adrenal Medulla: Catecholamine Secretion and the Sympathetic Stress Response
The adrenal medulla is the inner region of the adrenal gland and functions as a specialised neuroendocrine organ. Derived from neural crest cells, it acts as an extension of the sympathetic nervous system by releasing adrenaline and noradrenaline directly into the bloodstream during stress. These catecholamines produce rapid physiological responses, supporting the body’s ability to react to threat, exertion or emotional arousal. Although smaller than the adrenal cortex, the medulla plays a crucial role in moment-to-moment cardiovascular and metabolic regulation.
What You Need to Know
The adrenal medulla is the inner region of the adrenal gland and functions as a specialised component of the sympathetic nervous system. It plays a key role in the body’s acute stress response by rapidly releasing catecholamines into the circulation. These hormones prepare multiple organ systems to respond to immediate physical or psychological stress by increasing cardiovascular output, mobilising energy stores, and redirecting blood flow to vital tissues.
Several core features distinguish adrenal medullary function from other endocrine systems:
Secretion of catecholamines, primarily adrenaline and noradrenaline
Direct neural control via pre-ganglionic sympathetic fibres
Rapid, short-acting systemic effects
The adrenal medulla is composed of chromaffin cells, which are modified post-ganglionic sympathetic neurons. Unlike most endocrine glands, the medulla is not regulated by pituitary hormones. Instead, chromaffin cells are stimulated directly by acetylcholine released from pre-ganglionic sympathetic nerves. This neural control allows catecholamines to be released within seconds, producing a fast, coordinated response that increases heart rate, blood pressure, ventilation, blood glucose availability, and skeletal muscle perfusion.
Although both adrenaline and noradrenaline are secreted, adrenaline predominates in humans. This is due to the presence of the enzyme phenylethanolamine N-methyltransferase, which converts noradrenaline into adrenaline within chromaffin cells. The dominance of adrenaline supports widespread metabolic and cardiovascular effects, reinforcing the adrenal medulla’s role in mounting a rapid, whole-body response to stress.
Image: The adrenal gland is composed of an outer cortex and an inner medulla. The cortex produces steroid hormones, while the medulla secretes catecholamines in response to sympathetic stimulation.
Beyond the Basics
Embryological origin and neural identity
The adrenal medulla develops from neural crest cells, the same embryological population that gives rise to much of the sympathetic nervous system. During development, these cells migrate into the forming adrenal gland and differentiate into chromaffin cells. This shared origin explains why the adrenal medulla behaves more like a neural structure than a traditional endocrine gland and why its responses are rapid and tightly linked to sympathetic activation.
Chromaffin cells retain many neural characteristics despite lacking axons. Instead of transmitting signals along nerve fibres, they release catecholamines directly into the bloodstream, functioning as endocrine cells. Their cytoplasm contains dense-core granules packed with adrenaline, noradrenaline, and co-released peptides. This structure reflects their dual identity, allowing fast signal transduction typical of neurons alongside hormone-mediated systemic effects.
Catecholamine synthesis and cortical interaction
Catecholamines are synthesised from the amino acid tyrosine through a well-defined enzymatic pathway within chromaffin cells. Tyrosine is first converted to DOPA and then to dopamine, which is transported into secretory granules. Within these granules, dopamine is converted to noradrenaline, the primary catecholamine produced by many sympathetic neurons.
In chromaffin cells that express phenylethanolamine N-methyltransferase, noradrenaline undergoes further conversion to adrenaline. Activity of this enzyme is stimulated by high local concentrations of cortisol delivered from the adjacent adrenal cortex via the portal circulation. This anatomical arrangement creates a functional link between the cortex and medulla, ensuring that adrenaline production is enhanced during states of increased cortisol secretion, such as stress. The result is a coordinated adrenal response that integrates slower hormonal adaptation with rapid sympathetic activation.
Neural control and hormone release
Catecholamine secretion from the adrenal medulla is controlled by direct neural input rather than by endocrine feedback loops. Pre-ganglionic sympathetic neurons release acetylcholine onto chromaffin cells, where it binds to nicotinic receptors on the cell membrane. This binding causes depolarisation and opening of voltage-gated calcium channels, allowing calcium to enter the cell.
The rise in intracellular calcium triggers exocytosis of catecholamine-containing granules into nearby capillaries. Because this pathway bypasses transcriptional regulation and hormone synthesis delays, catecholamine release can occur within seconds of stress detection. This mechanism allows the adrenal medulla to provide an immediate, whole-body response that complements slower endocrine systems.
Integrated physiological effects
Once released into the circulation, adrenaline and noradrenaline bind to adrenergic receptors distributed throughout the body. Their effects include increasing heart rate and myocardial contractility, redistributing blood flow by constricting vessels in less essential tissues while dilating those supplying skeletal muscle, and enhancing ventilation. At the metabolic level, catecholamines promote glycogen breakdown, gluconeogenesis, and lipolysis, rapidly increasing the availability of energy substrates.
These actions prepare the body for acute physical or psychological stress and are short-lived by design. Catecholamines act quickly and are rapidly metabolised, allowing the stress response to be finely tuned and reversible. Together with cortisol and other longer-acting hormones, adrenal medullary catecholamines form part of a layered stress-response system that balances speed, duration, and metabolic cost.
Clinical Connections
Disorders of the adrenal medulla are less common than those of the adrenal cortex, but when they occur, they tend to produce striking and sometimes episodic symptoms. Because catecholamines act rapidly and affect the cardiovascular, metabolic, and autonomic systems, excess or dysregulated secretion often presents with sudden, multisystem changes rather than gradual deterioration.
In clinical settings, adrenal medullary dysfunction is most often encountered in the following patterns:
Excess catecholamine release
Impaired or absent catecholamine responses during stress
Phaeochromocytomas are catecholamine-secreting tumours arising from chromaffin cells and are the classic example of adrenal medullary disease. They typically cause episodic hypertension accompanied by palpitations, sweating, headache, and a sense of anxiety or panic. These episodes reflect sudden surges of adrenaline and noradrenaline rather than sustained hormone elevation, which helps explain why blood pressure and symptoms can fluctuate dramatically between episodes.
Chronic catecholamine excess places ongoing strain on the cardiovascular and metabolic systems. Sustained exposure can contribute to persistent hypertension, weight loss, hyperglycaemia, and cardiomyopathy due to prolonged sympathetic stimulation. In contrast, reduced medullary function limits the body’s ability to mount an effective acute stress response. This may become apparent during illness, surgery, or in the context of autonomic dysfunction, where patients struggle to maintain blood pressure or respond appropriately to physiological stressors.
Understanding the structure and neural control of the adrenal medulla helps explain both the speed and severity of catecholamine-related conditions. It also clarifies why surgical removal of medullary tumours requires careful perioperative management, as sudden changes in catecholamine levels can lead to profound cardiovascular instability if not anticipated and controlled.
Concept Check
Why does the adrenal medulla release hormones in response to neural stimulation rather than pituitary control
How does cortisol from the adrenal cortex influence catecholamine synthesis in the medulla
Why is the adrenal medulla considered a modified sympathetic ganglion
How do catecholamines produce rapid cardiovascular and metabolic changes
What clinical features suggest a catecholamine-secreting tumour such as a phaeochromocytoma