GRANULOCYTES: Neutrophils, Eosinophils and Basophils
Granulocytes are a group of innate immune cells characterised by cytoplasmic granules that contain potent antimicrobial or inflammatory substances. These cells, neutrophils, eosinophils and basophils, play essential roles in early immune responses, inflammation and defence against a wide range of pathogens. Although they share a common origin in the bone marrow, each granulocyte subtype has highly specialised functions. Together, they form a rapid-response system that contains infection, combats parasites, contributes to allergic responses and shapes inflammation.
What You Need to Know
Granulocytes are a group of innate immune cells characterised by the presence of cytoplasmic granules, small membrane-bound packets inside the cell that store enzymes, antimicrobial proteins, and inflammatory signalling molecules. They develop from myeloid precursor cells in the bone marrow and circulate in the bloodstream under resting conditions. During infection, inflammation, or tissue injury, granulocytes are rapidly recruited into tissues, where their granule contents can be released almost immediately in response to immune activation.
Several shared features explain how granulocytes function as rapid first responders in immune defence:
Cytoplasmic granules contain pre-formed effector molecules, meaning antimicrobial substances do not need to be newly synthesised
Large circulating and bone marrow pools allow rapid mobilisation to sites of inflammation
A short lifespan is paired with high cytotoxic and inflammatory capacity
Although granulocytes share a common developmental lineage, neutrophils, eosinophils, and basophils perform distinct roles. Neutrophils are the most abundant circulating white blood cell and dominate early responses to bacterial infection. Eosinophils are specialised for defence against parasitic infections, particularly helminths, and contribute to allergic inflammation. Basophils are the least numerous but are powerful sources of histamine and other mediators that increase vascular permeability and influence immune cell recruitment.
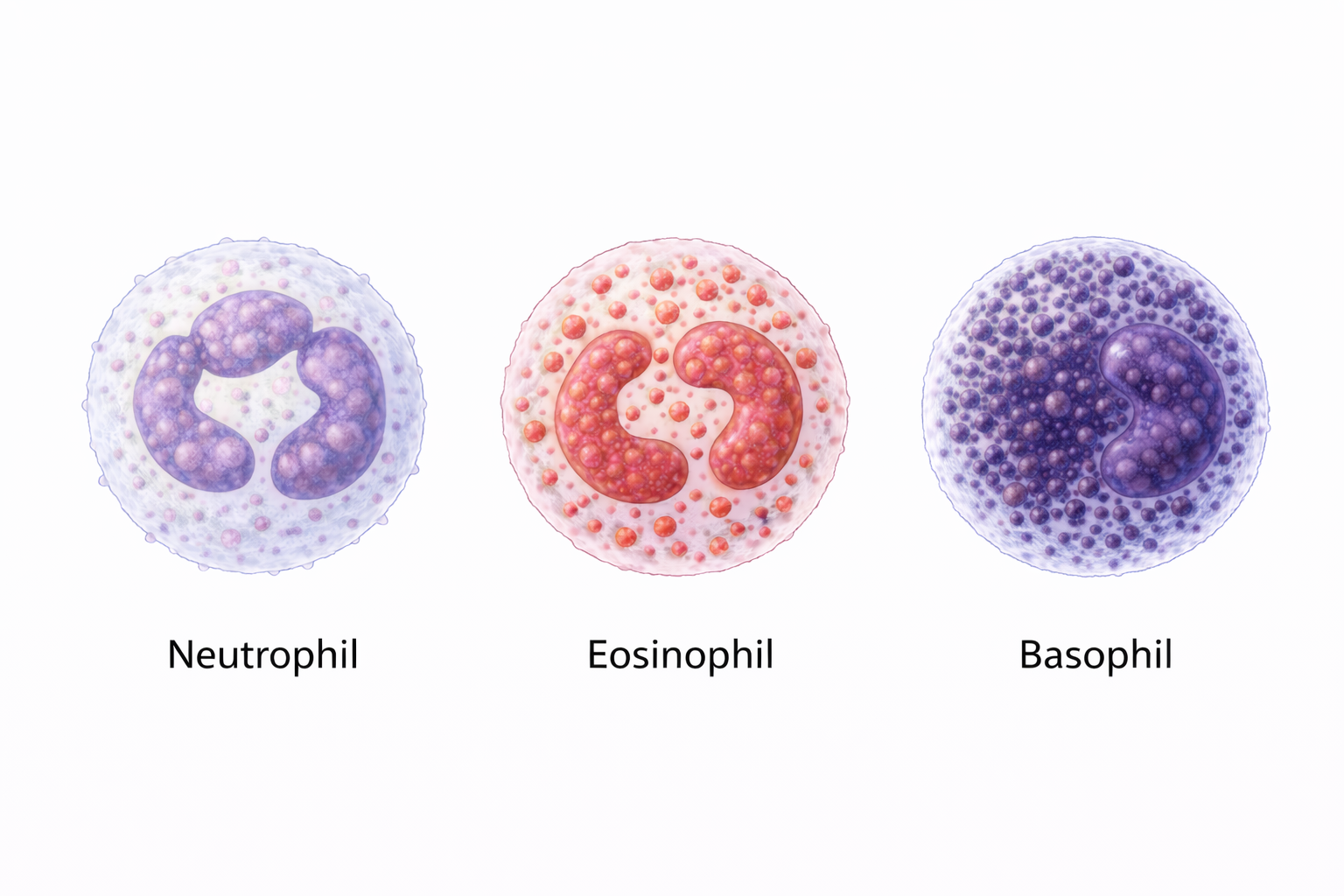
Image: Neutrophils, eosinophils, and basophils are granulocytes distinguished by their nuclear shape and cytoplasmic granules. Neutrophils have a multilobed nucleus and fine granules for phagocytosis, eosinophils have a bilobed nucleus with red-orange granules involved in parasitic and allergic responses, and basophils contain dense purple granules that can obscure the nucleus and release histamine during inflammatory reactions.
Beyond the Basics
Neutrophils: Rapid Phagocytic Effectors
Neutrophils account for approximately 50 to 70 percent of circulating white blood cells and are usually the first immune cells to arrive at sites of acute infection or tissue injury. They respond rapidly to chemotactic signals, chemical gradients released by bacteria, damaged tissues, and activated immune cells that guide neutrophils toward the source of inflammation. This early arrival places neutrophils at the centre of initial host defence, particularly in bacterial infections.
Once activated, neutrophils engulf pathogens through phagocytosis, enclosing them within intracellular vesicles called phagosomes. These fuse with lysosomes to form phagolysosomes, specialised compartments where enzymes, antimicrobial peptides, and reactive oxygen species act together to destroy the ingested organism. After completing their antimicrobial role, neutrophils commonly undergo apoptosis, a controlled form of cell death that limits ongoing inflammation and supports resolution of the immune response.
Neutrophils can also release neutrophil extracellular traps, or NETs, which are web-like structures composed of chromatin and antimicrobial enzymes expelled into the extracellular space. NETs trap and neutralise microbes outside the cell, particularly when pathogens are too numerous or too large to be efficiently phagocytosed. Excessive NET formation, however, is associated with tissue injury and thrombosis, making regulation of this response clinically important.
Eosinophils: Defence Against Parasites and Modulators of Allergy
Eosinophils make up approximately 1 to 4 percent of circulating leukocytes and are most abundant in mucosal tissues, particularly within the gastrointestinal tract where exposure to parasitic organisms is common. Their granules contain major basic protein, eosinophil cationic protein, and peroxidases, all of which are toxic to large multicellular parasites that cannot be internalised by phagocytosis.
Rather than engulfing pathogens, eosinophils act primarily through targeted degranulation, releasing cytotoxic proteins directly onto the surface of parasites. They also release cytokines and lipid mediators that influence vascular permeability, immune cell recruitment, and tissue remodelling, placing eosinophils at the intersection of host defence and inflammatory regulation.
In allergic disease, eosinophils contribute to chronic tissue inflammation. In asthma, for example, eosinophils accumulate in the airways and release mediators that damage the epithelium, increase mucus production, and promote bronchial hyperresponsiveness. These effects help explain why eosinophil-driven inflammation can persist even when the original allergen exposure is intermittent.
Basophils: Triggers of Inflammation and Allergy
Basophils are the least abundant granulocytes, accounting for less than 1 percent of circulating leukocytes, yet they exert powerful effects on inflammatory and allergic responses. Their cytoplasmic granules are rich in histamine, heparin, and proteases, mediators that rapidly alter vascular tone and tissue permeability when released.
Basophils express high-affinity IgE receptors on their surface and become activated when IgE-bound allergens cross-link, meaning a single allergen binds multiple IgE molecules simultaneously. This triggers degranulation, leading to histamine release and subsequent vasodilation, increased vascular permeability, and smooth muscle contraction. These physiological changes underpin many of the immediate features of allergic reactions, including oedema, bronchoconstriction, and pruritus.
Basophils also produce interleukin-4, a cytokine that promotes T helper 2 immune responses and supports IgE class switching in B cells. Through this pathway, basophils contribute not only to acute allergic reactions but also to the maintenance of allergic sensitisation over time.
Granulocyte Recruitment and Coordination
Recruitment of granulocytes to inflamed tissues depends on coordinated interactions between circulating immune cells and the vascular endothelium. In response to inflammatory signals, endothelial cells upregulate adhesion molecules that allow granulocytes to slow, adhere, and migrate out of the bloodstream through diapedesis, the process by which cells pass between endothelial junctions into tissues.
Neutrophils typically arrive first, guided by chemokines such as interleukin-8 that selectively attract them to sites of bacterial infection. Eosinophils are recruited by eotaxins, chemokines that direct eosinophil migration to mucosal surfaces and areas of allergic inflammation. Basophil recruitment occurs in contexts dominated by type 2 immune responses. This selective trafficking ensures that each granulocyte subtype is deployed where its effector functions are most effective.
Lifespan and Renewability
Granulocytes are short-lived cells, often surviving only hours to days once they enter tissues and become activated. Their brief lifespan limits prolonged tissue exposure to highly cytotoxic mediators and helps contain inflammation.
To maintain immune readiness, the bone marrow produces granulocytes continuously, releasing millions of new cells into the circulation every hour. During acute infection or systemic inflammation, production increases markedly, leading to detectable changes in peripheral blood counts. This rapid turnover allows the immune system to sustain effective early responses while replacing cells that have completed their effector function.
Clinical Connections
Changes in granulocyte number and function are common clinical indicators of underlying disease and are frequently used to guide assessment and decision-making. Neutrophilia, an increased neutrophil count, is a typical finding in acute bacterial infection and inflammatory states, driven by rapid mobilisation of neutrophils from the bone marrow into the circulation. In contrast, neutropenia significantly increases susceptibility to infection, particularly from opportunistic bacteria and fungi. This is a major concern in patients receiving chemotherapy, where reduced neutrophil production limits the body’s ability to mount an effective early immune response.
Several clinical patterns link granulocyte behaviour with disease presentation and risk:
Neutrophilia in acute infection, tissue injury, or systemic inflammation
Neutropenia associated with chemotherapy, bone marrow suppression, or severe infection
Eosinophilia in parasitic infection, allergic disease, and some autoimmune conditions
Basophil activation in immediate hypersensitivity reactions and anaphylaxis
Eosinophilia often occurs in settings of sustained immune activation at mucosal surfaces, particularly in allergic disease such as asthma or in parasitic infection where eosinophil-mediated cytotoxicity is required. Persistently elevated eosinophil counts can contribute to tissue injury through ongoing mediator release, linking laboratory findings with chronic inflammatory symptoms. Basophils, while rarely increased in number, have a central role in allergic reactions. IgE-mediated activation triggers rapid histamine release, leading to vasodilation, increased vascular permeability, and bronchoconstriction. Basophil activation tests are increasingly used in allergy diagnostics to support identification of specific triggers.
Granulocyte dysfunction, whether due to inherited disorders, medication effects, or systemic disease, compromises early immune defence and immune regulation. Impaired neutrophil phagocytosis or intracellular killing increases the risk of recurrent bacterial infections, while dysregulated eosinophil or basophil activity contributes to chronic inflammation and allergic pathology.
Concept Check
What are the primary roles of neutrophils in early immune defence?
How do eosinophils contribute to both parasitic defence and allergic disease?
Why are basophils important in IgE-mediated immunity?
What are neutrophil extracellular traps (NETs), and how do they assist in pathogen control?
How does the body rapidly replace granulocytes given their short lifespan?