INNATE IMMUNITY: The Body’s First and Immediate Line of Defence
Innate immunity is the body’s rapid, non-specific defence system that responds immediately to potential threats. Unlike adaptive immunity, which requires time to generate targeted responses, innate immunity provides broad protection from the moment a pathogen enters the body. This system relies on physical and chemical barriers, phagocytic cells, inflammatory responses, antimicrobial proteins and early signalling mechanisms that contain infection and recruit the adaptive immune system when needed. Innate immunity is foundational for survival. It controls the majority of infections before they cause disease, maintains tissue homeostasis and ensures that the body can respond quickly to danger.
What You Need to Know
Innate immunity provides the body’s immediate defence against infection and tissue injury. It is always active and responds within minutes to hours, forming the first protective layer before adaptive immunity is engaged. The initial barrier consists of physical and chemical defences that prevent pathogens from entering the body in the first place. Intact skin, mucous membranes, gastric acid, cilia in the respiratory tract, antimicrobial secretions, and normal microbiota work together to limit microbial attachment, survival, and colonisation at exposed surfaces.
When these external barriers are breached, innate immune responses are rapidly activated within tissues. Key components include phagocytic cells such as neutrophils, macrophages, and dendritic cells, as well as natural killer cells, complement proteins, interferons, and inflammatory mediators. These elements recognise conserved features of microbes rather than specific antigens, allowing rapid detection of a wide range of pathogens without prior exposure.
Several coordinated mechanisms drive this early response:
Phagocytosis, where immune cells engulf and destroy microbes
Inflammation, which increases blood flow and recruits immune cells to affected tissues
Complement activation, which enhances microbial clearance and cell lysis
Interferon signalling, which limits viral replication and alerts neighbouring cells
Innate immunity contains infection, limits early pathogen spread, and shapes subsequent adaptive immune responses. Signals generated during innate activation influence antigen presentation, T cell differentiation, and antibody production, ensuring that later immune responses are appropriate to the type and location of the threat. This early system therefore provides both immediate protection and critical instruction for longer-term immunity.
Beyond the Basics
Pattern Recognition and Early Detection
Innate immune cells detect danger through pattern recognition receptors, including Toll-like receptors. These receptors identify conserved molecular signatures shared by groups of microbes rather than unique antigens. Pathogen-associated molecular patterns are structures such as bacterial cell wall components, viral nucleic acids, or fungal carbohydrates. Damage-associated molecular patterns are signals released from stressed or injured host cells, such as ATP or nuclear proteins.
Recognition of these signals triggers intracellular signalling pathways that activate transcription factors responsible for cytokine and chemokine production. This early signalling alerts neighbouring cells, recruits immune cells from the bloodstream, and establishes an inflammatory environment that limits pathogen spread. Because these receptors are pre-formed and do not require prior exposure, innate responses begin almost immediately after detection.
Phagocytes and Cellular Defence
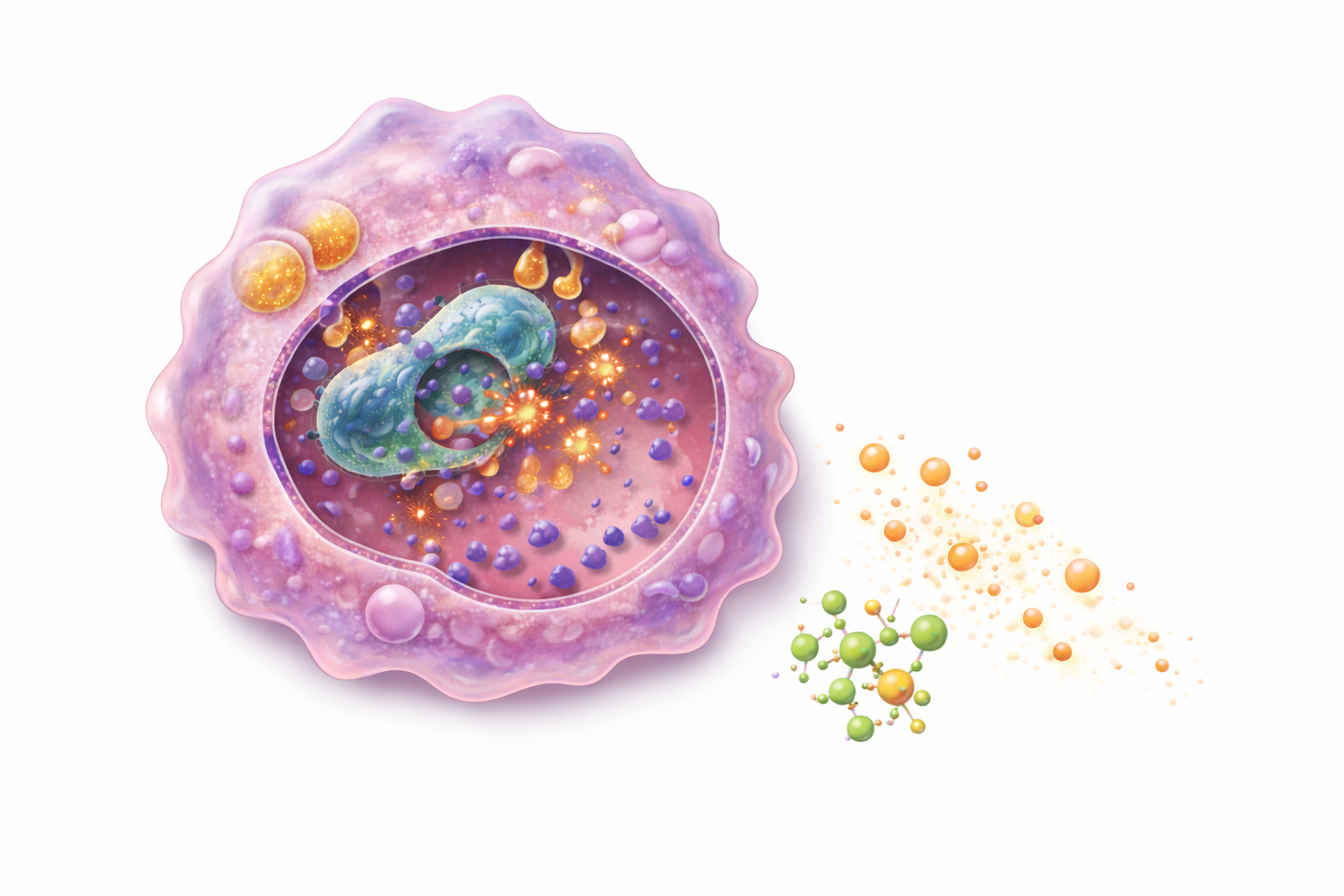
Neutrophils are the most abundant circulating white blood cells and are among the first cells recruited to infected or injured tissue. Guided by chemokines, they migrate out of blood vessels and engulf microbes through phagocytosis. Within the cell, microbes are destroyed using reactive oxygen species, chemically reactive molecules that damage proteins and membranes, and lysosomal enzymes that break down cellular components.
Macrophages are long-lived phagocytes that reside within tissues such as the lungs, liver, and spleen. In addition to clearing pathogens and debris, macrophages release cytokines that shape the inflammatory response and influence tissue repair. They also process and present antigens to T cells, providing a functional link between innate detection and adaptive immunity. Dendritic cells specialise in antigen capture and transport. After encountering pathogens in peripheral tissues, they migrate to lymph nodes to activate naïve T cells, initiating adaptive immune responses.
Image: Phagocytosis involves the engulfment of a pathogen into a membrane-bound vesicle (phagosome), which then fuses with lysosomes to form a phagolysosome. Within this compartment, antimicrobial mechanisms including reactive oxygen species, enzymes, and antimicrobial peptides break down the pathogen. At the same time, the cell releases cytokines that help coordinate the broader immune response.
Inflammation and Early Immune Signalling
Inflammation is a central feature of innate immunity and develops rapidly following infection or tissue injury. Cells at the site release mediators such as histamine, prostaglandins, and cytokines, which act locally on blood vessels and immune cells. Vasodilation increases blood flow, while increased vascular permeability allows plasma proteins and leukocytes to enter tissues.
These changes account for the classic features of inflammation: redness and warmth from increased blood flow, swelling from fluid leakage, and pain due to mediator effects on nerve endings. Inflammatory signalling concentrates immune activity at the site of damage, limits pathogen dissemination, and prepares the tissue environment for repair once the threat has been controlled.
Complement System and Antimicrobial Proteins
Complement proteins circulate in the blood in an inactive state and are rapidly activated in the presence of microbes or antibody-coated targets. Once triggered, complement fragments bind to pathogen surfaces, enhancing phagocytosis through opsonisation and recruiting immune cells through inflammatory signalling. The terminal components assemble into the membrane attack complex, which forms pores in microbial membranes and causes lysis, particularly in Gram-negative bacteria.
Interferons are another critical innate defence, especially during viral infection. Type I interferons induce expression of antiviral proteins within infected and neighbouring cells, limiting viral replication and spread. Natural killer cells contribute to early defence by identifying and killing virally infected or malignant cells. They target cells with reduced or abnormal MHC class I expression, inducing apoptosis without prior sensitisation.
Fever and Systemic Innate Responses
Innate immune activation can extend beyond local tissues to produce systemic responses. Cytokines such as interleukin-1 and tumour necrosis factor act on the hypothalamus to raise body temperature. Fever slows pathogen replication and enhances the efficiency of immune processes such as phagocytosis (engulfing and digesting pathogens) and antigen presentation (displaying pathogen fragments to activate other immune cells).
At the same time, the liver produces acute-phase proteins, including C-reactive protein (abbreviated to CRP in clinical practice and used as a marker for inflammation). These proteins bind to microbial structures, helping to label pathogens for destruction, enhance complement activation, and improve pathogen clearance. This is why CRP rises in blood tests during infection or inflammation. Fever and acute-phase responses support early containment of infection and signal that systemic immune activation is underway.
Clinical Connections
Innate immunity is critical during the earliest stages of infection and tissue injury. It determines how quickly pathogens are contained and how much inflammation develops before adaptive responses take over. When innate defences are compromised, even common infections can become severe or life-threatening. Neutropenia markedly increases the risk of bacterial and fungal infections, while complement deficiencies impair opsonisation and microbial clearance, particularly of encapsulated organisms.
Several common clinical patterns are closely linked to innate immune activity:
Fever, local redness, swelling, and pain during acute infection
Elevated inflammatory markers such as C-reactive protein during systemic inflammation
Severe infection in patients with neutropenia or complement dysfunction
Tissue damage resulting from prolonged or excessive inflammatory responses
Excessive activation of innate immunity contributes to inflammatory injury rather than protection. In sepsis, widespread cytokine release, complement activation, and endothelial dysfunction lead to hypotension, organ injury, and impaired perfusion. Even outside of sepsis, persistent innate activation can drive chronic inflammatory conditions and delay tissue healing.
Innate immune responses also explain why symptoms may persist after antimicrobial therapy is started. Antibiotics reduce bacterial burden but do not immediately switch off inflammatory signalling. Resolution requires clearance of inflammatory mediators, removal of cellular debris, and restoration of tissue homeostasis. This delay is particularly relevant in respiratory infections, soft tissue infections, and postoperative inflammation.
Innate immunity is also essential for effective vaccination. Vaccine components are detected by pattern recognition receptors, triggering cytokine production and antigen presentation that activate adaptive immune responses. Without this early innate signalling, long-term immune memory does not develop effectively. Understanding innate mechanisms therefore supports clinical decision-making relating to infection risk, fever management, inflammatory markers, and vaccine response across diverse patient populations.
Concept Check
How do pattern recognition receptors distinguish between pathogens and healthy tissues?
What roles do neutrophils and macrophages play during the early stages of infection?
How does inflammation support pathogen clearance and tissue repair?
What are the major functions of the complement system in innate immunity?
Why does fever enhance immune defence?