The Immune System - Overview
The immune system is the body’s sophisticated defence network, designed to protect against infection, eliminate harmful pathogens, recognise foreign substances, and maintain internal balance. It consists of specialised cells, tissues, organs, and molecular pathways working together to detect threats and mount appropriate responses. Unlike most organs, the immune system is distributed throughout the entire body, continuously surveying for danger. It is capable of rapid, nonspecific responses as well as highly specific, long-lasting protection through immunological memory.
What You Need to Know
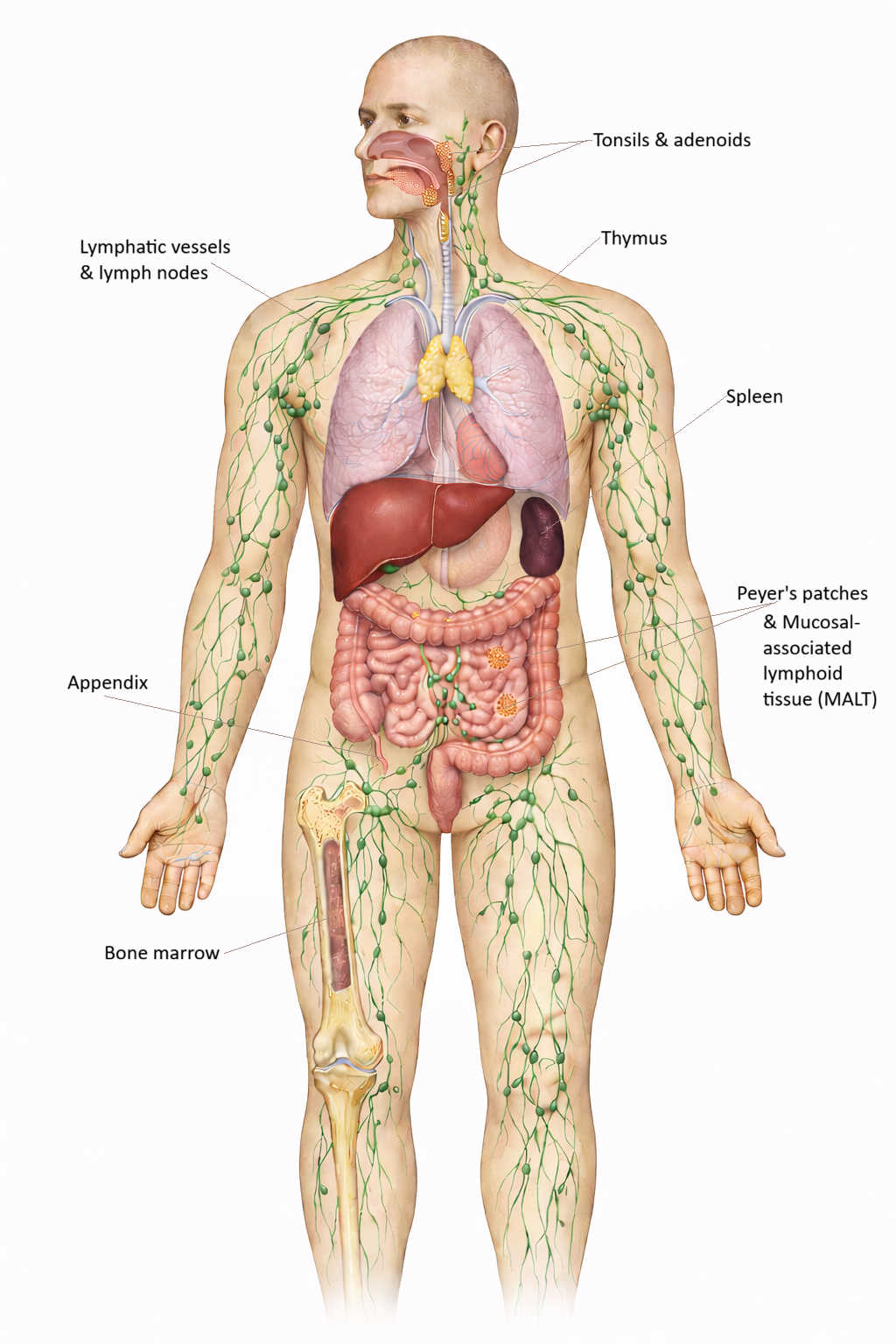
The immune system is organised through specialised organs, cells, and signalling pathways that work together to protect the body from infection and injury. Immune cells originate in the bone marrow through haematopoiesis, the process that generates all blood cells. Some lymphocytes mature in the bone marrow, while others migrate to the thymus, where T cells undergo development and selection to ensure they can respond to foreign antigens without targeting self-tissues.
Once mature, immune cells circulate through secondary lymphoid organs, which act as sites of immune surveillance and activation:
Lymph nodes filter lymphatic fluid and coordinate immune responses to tissue-derived antigens
The spleen monitors the bloodstream for pathogens and removes damaged red blood cells
Mucosa-associated lymphoid tissues protect surfaces exposed to the external environment
These structures provide organised environments where immune cells interact, become activated, and form memory populations.
Immune defence operates through two tightly integrated systems. The innate immune system provides immediate, non-specific protection using physical barriers, inflammation, and rapid-acting cells such as neutrophils, macrophages, and natural killer cells. These responses limit infection early and shape subsequent immune activity. The adaptive immune system develops more slowly but provides antigen-specific responses through B cells and T cells. Antibodies, cytotoxic activity, and immune memory allow precise targeting of pathogens and long-term protection.
Communication between immune components is mediated by cytokines, chemokines, complement proteins, and cell-to-cell interactions. These interactions allow the immune response to be rapid when needed, controlled to limit tissue damage, and adaptive to future threats.
Image: The immune system is a body-wide network of organs, tissues, and vessels. Immune cells develop in the bone marrow and thymus, then circulate through lymphatic vessels and lymphoid organs such as lymph nodes, the spleen, and MALT, where pathogens are detected and immune responses are initiated.
Beyond the Basics
Immune Tolerance and Autoimmunity
Immune function extends beyond pathogen defence and includes the ability to tolerate self-antigens (molecules, such as proteins, fats, or sugars, produced by an individual’s own cells). Immune tolerance allows lymphocytes to remain unresponsive to the body’s own tissues while retaining the capacity to respond to infection. This balance is maintained through central tolerance during lymphocyte development and peripheral tolerance mechanisms that regulate immune activity in tissues, meaning potentially harmful self-reactive cells are either removed early as they develop or kept under control later in the body.
Failure of tolerance leads to activation of self-reactive T and B cells, leading to autoimmune disease. Conditions such as systemic lupus erythematosus (SLE), rheumatoid arthritis, type 1 diabetes, and multiple sclerosis arise when immune responses are directed against specific self-antigens. In many cases, genetic susceptibility, particularly certain human leukocyte antigens (HLA) types (cell-surface proteins that allow the immune system to distinguish between self and foreign cells), combines with environmental triggers to initiate disease.
Immunoregulation and Chronic Inflammation
Immune responses must be actively regulated to prevent excessive or prolonged inflammation. Regulatory T cells play a central role in suppressing immune activation through direct cell contact and secretion of anti-inflammatory cytokines, such as interleukin-10 and transforming growth factor beta. These mechanisms limit tissue damage once a threat has been controlled.
When immunoregulation is impaired, inflammation may persist even in the absence of infection. Chronic inflammatory states contribute to diseases such as inflammatory bowel disease, chronic pain syndromes, atherosclerosis, and some malignancies. Sustained cytokine production and immune cell infiltration alter tissue structure and function over time, leading to progressive pathology.
Neuroendocrine–Immune Interactions
The immune system is closely integrated with the nervous and endocrine systems. Stress responses influence immune activity through hormones such as cortisol, which suppress inflammatory signalling and lymphocyte function. Short-term stress responses may be protective, but chronic stress reduces immune surveillance and increases susceptibility to infection.
Neural signalling and immune mediators are closely interconnected, forming part of a broader neuroimmune network that allows the body to coordinate physiological and behavioural responses to threat. Cytokines released during immune activation can act on the central nervous system, either directly or via signalling pathways such as the vagus nerve, to influence mood, cognition, and behaviour.
This contributes to the characteristic fatigue, reduced motivation, and social withdrawal seen during illness, often referred to as sickness behaviour. These interactions demonstrate that immune activity is not confined to local tissue responses but has widespread systemic effects, influencing whole-body physiology and behavioural adaptation.
The Microbiome and Immune Modulation
The gut microbiome exerts a major influence on immune development, tolerance, and inflammatory control. Commensal microbes (microorganisms that live on the human body) interact with epithelial cells, dendritic cells, and regulatory T cells to shape immune responses from early life onward. Microbial metabolites, such as short-chain fatty acids produced by the normal microbiota, support epithelial barrier integrity and help regulate immune responses, particularly at mucosal surfaces like the gut, where the immune system must balance tolerance and defence.
Disruption of the microbiome through antibiotics, infection, or dietary imbalance alters immune signalling and barrier function. These changes are associated with increased risk of allergic disease, autoimmune conditions, and chronic inflammation. Maintenance of microbial diversity is therefore an important contributor to immune stability.
Immune Surveillance and Cancer
The immune system continuously monitors tissues for abnormal or transformed cells, a process known as immune surveillance. Cytotoxic T cells and natural killer cells identify and eliminate cells expressing abnormal antigens or altered MHC expression. Many potential malignancies are controlled at this early stage without clinical detection.
Cancer cells may evade immune surveillance by suppressing antigen presentation, producing inhibitory signals, or altering the tumour microenvironment. Modern immunotherapies aim to overcome these mechanisms. Checkpoint inhibitors enhance T cell activity by removing inhibitory signalling, while CAR-T cell therapy engineers patient T cells to recognise tumour antigens directly. These approaches utilise immune specificity and memory to improve cancer control.
Clinical Connections
Immune dysfunction presents across a wide clinical spectrum, from transient infection to chronic inflammatory and immune-mediated disease. Acute infections activate strong inflammatory responses that produce fever, redness, swelling, and pain as immune cells and mediators are rapidly recruited to affected tissues. Chronic infections place ongoing demands on the immune system and may lead to prolonged inflammation, tissue injury, or immune exhaustion, particularly when pathogens persist within cells or evade clearance.
Several common clinical patterns signal altered immune function:
Allergic reactions caused by immune responses directed against harmless antigens
Autoimmune disease resulting from loss of tolerance to self-tissues
Recurrent or severe infections associated with immunodeficiency
Prolonged inflammation contributing to tissue damage and chronic disease
Allergic disease occurs when immune responses are inappropriately directed toward non-threatening substances such as pollen, foods, or medications. These responses range from mild local symptoms to systemic anaphylaxis, driven by IgE-mediated mast cell activation. Autoimmune diseases develop when tolerance mechanisms fail, allowing sustained immune attack against self-antigens. Management often requires long-term immunosuppression, which reduces tissue damage but increases susceptibility to infection.
Immunodeficiency may be congenital or acquired. HIV infection remains the most recognised acquired cause, but immune suppression also occurs in patients receiving chemotherapy, those with malnutrition, advanced age, or chronic disease. Reduced immune capacity increases infection risk, delays recovery, and alters responses to vaccination and treatment. Careful assessment of immune status is therefore essential in many clinical settings.
In clinical practice, immune impairment has broad implications. Fever patterns assist in distinguishing bacterial from viral illness. Neutropenia following chemotherapy necessitates strict infection control and early intervention. Effective wound healing depends on coordinated immune activity. Vaccination history informs risk assessment and patient education. Subtle clinical signs such as lymphadenopathy, recurrent infections, unexplained fatigue, or persistent rashes may indicate underlying immune dysfunction and warrant further investigation. Understanding immune responses supports early recognition, appropriate escalation, and safe patient care across all clinical environments.
Concept Check
Explain how the innate and adaptive immune systems differ in speed, specificity, and memory, and describe how they work together during infection.
How do the bone marrow, thymus, lymph nodes, and spleen each contribute to immune function, and why are they considered essential components of the system?
What happens during inflammation, and why is this process both protective and potentially harmful?
How does immune tolerance work, and what occurs when this mechanism fails?
A patient presents with recurrent infections, prolonged wound healing, and persistent fatigue. Which immune disorders should you consider, and why?