PHAGOCYTOSIS: How Innate Immune Cells Detect, Engulf and Destroy Pathogens
Phagocytosis is a fundamental defence mechanism of the innate immune system. It allows specialised immune cells to engulf and destroy pathogens, clear cellular debris and initiate adaptive immune responses. This process is carried out primarily by neutrophils, macrophages and dendritic cells, cells designed to rapidly remove harmful organisms before they spread. Because phagocytosis is one of the first responses to infection, understanding how it works is essential for recognising early inflammation, immune activation, and the transition from innate to adaptive immunity.
What You Need to Know
Phagocytosis is a core mechanism of innate immunity that allows specialised immune cells to recognise, engulf and destroy pathogens, cellular debris and damaged cells. It provides a rapid, non-specific first line of defence and also supports activation of the adaptive immune system. The main phagocytic cells are neutrophils, macrophages and dendritic cells. Neutrophils act as fast, short-lived responders that rapidly ingest and kill microbes, while macrophages provide sustained phagocytic activity within tissues. Dendritic cells perform phagocytosis not only to eliminate pathogens, but also to initiate adaptive immune responses by processing and presenting antigens to lymphocytes.
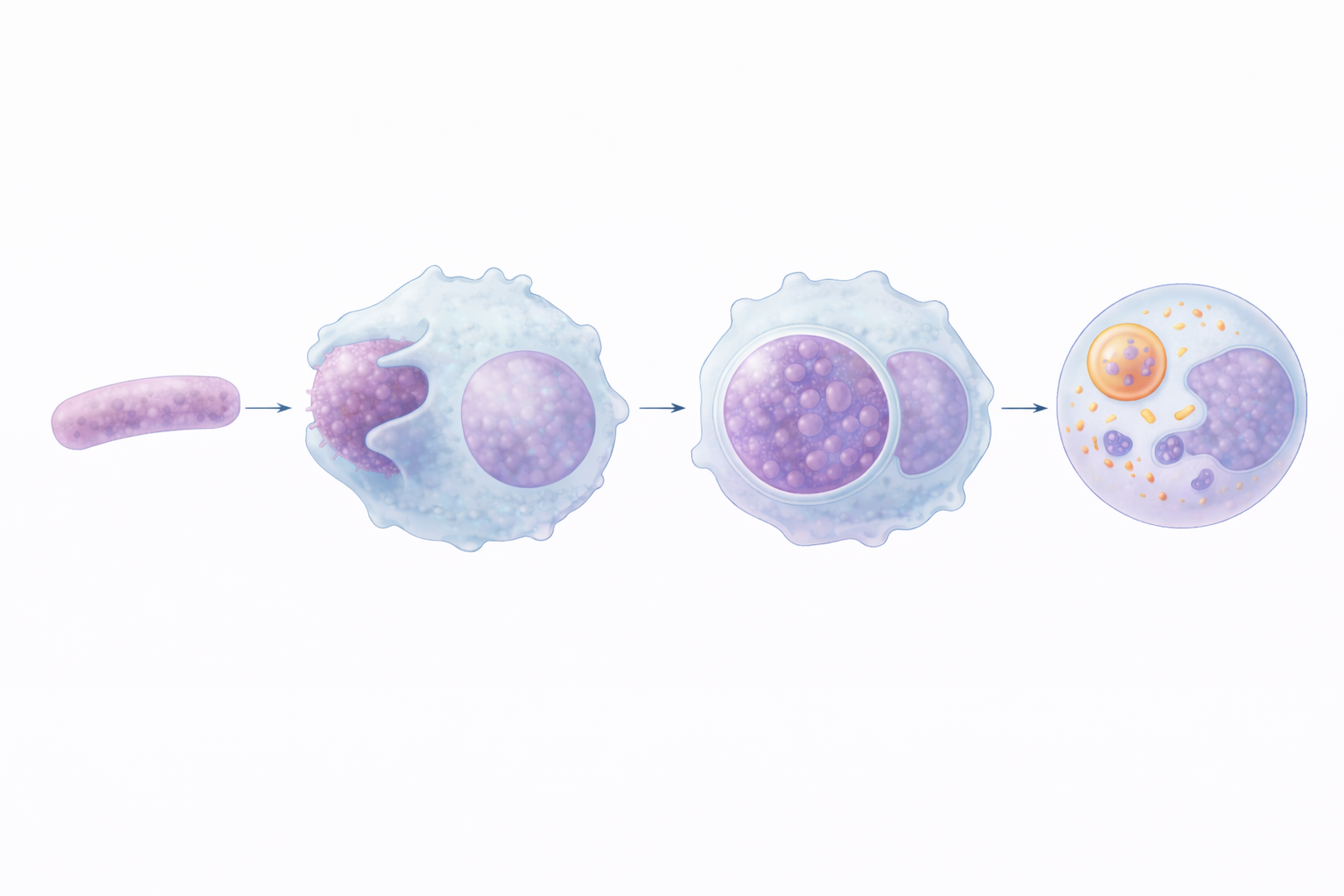
Phagocytosis begins with recognition and binding of a target. Phagocytes express surface receptors that detect conserved microbial patterns or host molecules attached to pathogens. This process is significantly enhanced by opsonisation, where antibodies or complement proteins coat the pathogen and improve binding efficiency. Once bound, the phagocyte surrounds the target with its membrane, enclosing it within a vesicle called a phagosome. The phagosome then fuses with lysosomes, forming a phagolysosome where the internal environment becomes acidic and hostile to microbial survival.
The process of phagocytosis can be summarised as a sequence of coordinated steps:
recognition of pathogens by surface receptors
strengthened attachment through opsonins such as antibodies or complement
engulfment into a membrane-bound phagosome
fusion with lysosomes to form a phagolysosome
enzymatic and oxidative destruction of the pathogen
Within the phagolysosome, pathogens are killed by acidic conditions, degradative enzymes and reactive oxygen or nitrogen species. Macrophages and dendritic cells retain peptide fragments after digestion, allowing them to present antigens on MHC molecules and activate T-cells. In this way, phagocytosis not only removes immediate threats but also links innate immune defence with longer-term, targeted immune responses.
Image: Phagocytosis is the process by which immune cells engulf and destroy pathogens. The cell extends pseudopodia to surround the bacterium, forming a phagosome, which then fuses with lysosomes to create a phagolysosome where enzymes break down and digest the pathogen.
Beyond the Basics
Step 1: Chemotaxis and Recruitment
Phagocytosis begins with recruitment of immune cells to the site of infection or tissue injury. Invading microbes, damaged cells and activated immune cells release chemical signals known as chemotactic factors. These include chemokines (immune signalling proteins), complement fragments such as C5a and bacterial products.
Together, these signals form a concentration gradient that guides phagocytes toward the affected area. Endothelial cells lining nearby blood vessels respond by expressing adhesion molecules, allowing circulating neutrophils and monocytes to slow, adhere and migrate between endothelial cells into surrounding tissue. This tightly regulated process ensures immune cells are delivered precisely where they are needed.
Step 2: Recognition and Attachment
Once at the site of infection, phagocytes must recognise and bind to their targets. Recognition occurs through two main mechanisms: pattern recognitions and opsonisation. In pattern recognition, phagocytes use pattern-recognition receptors (PRRs) to detect pathogen-associated molecular patterns (PAMPs), which are molecular structures shared by many microbes but absent from human cells.
In opsonisation, pathogens are coated with host molecules such as antibodies (IgG) or complement proteins (particularly C3b). These act as molecular “tags” that bind to specific receptors on phagocytes, greatly increasing the speed and efficiency of attachment.
Step 3: Engulfment and Phagosome Formation
Following attachment, the phagocyte reorganises its cytoskeleton to extend membrane projections called pseudopods around the pathogen. These extensions progressively surround and enclose the microbe. The cell membrane then fuses, internalising the pathogen within a membrane-bound vesicle known as a phagosome. At this stage, the pathogen is isolated from the extracellular environment but has not yet been destroyed.
Step 4: Phagolysosome Formation
The phagosome is transported deeper into the cell, where it fuses with lysosomes to form a phagolysosome. Lysosomes contain degradative enzymes, antimicrobial peptides and reactive molecules designed to destroy pathogens. In neutrophils, this fusion is accompanied by an oxidative burst, characterised by the rapid production of reactive oxygen species (ROS). These highly toxic molecules make neutrophils particularly effective at rapid microbial killing, although they also contribute to tissue inflammation.
Step 5: Killing and Degradation
Within the phagolysosome, pathogens are destroyed through multiple coordinated mechanisms. The internal environment becomes acidic, impairing microbial survival and enhancing enzyme activity. Reactive oxygen and nitrogen species damage proteins, membranes and genetic material. Enzymes such as proteases, lipases and lysozyme break pathogens down into small, harmless fragments. This controlled intracellular destruction ensures efficient pathogen elimination while minimising damage to surrounding tissue.
Step 6: Antigen Presentation (Macrophages and Dendritic Cells)
In macrophages and dendritic cells, phagocytosis also serves an adaptive immune function. After digestion, selected pathogen fragments are processed and presented on major histocompatibility complex (MHC) class II molecules on the cell surface. This antigen presentation activates helper T cells, linking innate immune defence with adaptive immunity. This step is essential for coordinating targeted immune responses and establishing long-term immune memory.
Clinical Connections
Defects in phagocytic function can significantly impair the body’s ability to control infection, particularly bacterial and fungal pathogens. Because phagocytes are central to early immune defence, even subtle dysfunction can lead to recurrent, severe or unusual infections. Laboratory findings related to phagocyte activity often provide early diagnostic clues in acute illness and immunodeficiency states.
Key clinical links related to impaired or exaggerated phagocytic activity include:
Chronic granulomatous disease, where defective reactive oxygen species (ROS) production severely reduces intracellular killing despite normal phagocyte numbers
Leukocytosis, particularly neutrophilia, which commonly reflects acute bacterial infection and active phagocytic recruitment
Opsonisation defects, such as antibody deficiency or low complement levels, which reduce phagocyte recognition and increase susceptibility to pneumonia, sepsis and skin infections
Excessive or prolonged inflammation, where ongoing phagocyte activation contributes to tissue damage rather than protection
Pus formation consists largely of dead neutrophils, digested microbes and cellular debris following intense phagocytic activity. Failure of effective clearance can prolong inflammation, delay healing and increase systemic spread of infection. For clinicians, recognising when phagocytosis is impaired, or overactive, helps guide investigation, antimicrobial decisions and assessment of immune competence.
Concept Check
What signals draw phagocytes to sites of infection?
How does opsonisation enhance phagocytosis?
What happens inside a phagolysosome?
Which cells use phagocytosis to initiate adaptive immunity, and how?
Why do defects in oxidative burst lead to recurrent infections?