The Skin Microbiome: Microbial Communities, Protective Functions and Host Interactions
The skin microbiome refers to the diverse population of microorganisms—bacteria, fungi, viruses and microscopic mites—that live symbiotically on the skin's surface. Far from being a passive layer of environmental contaminants, these microbial communities form an essential component of the skin’s defence system. They protect against pathogens, support immune regulation, contribute to barrier integrity and influence inflammatory responses. The composition of the microbiome varies across body regions depending on moisture, temperature, pH, hair density and glandular activity. Understanding the microbiome offers insight into skin health, wound healing, infection susceptibility and chronic dermatological conditions such as acne, eczema and psoriasis.
What You Need to Know
The skin microbiome refers to the collection of bacteria, fungi, viruses and mites that live on the skin surface and within hair follicles and glands. These organisms exist in a symbiotic relationship with the host rather than as passive contaminants. Dominant microbial groups vary by body site, reflecting differences in moisture, sebum content, oxygen availability and pH. Oily regions, moist folds and dry surfaces therefore support distinct microbial communities.
A healthy microbiome plays an active protective role. Commensal organisms limit pathogen overgrowth, reinforce the acid mantle and interact directly with skin and immune cells. Key protective functions include:
Competition with pathogens for nutrients and physical space
Production of antimicrobial substances and bacteriocins
Stimulation of keratinocytes to release antimicrobial peptides
Support of an acidic surface pH that discourages pathogenic growth
Immune “education,” promoting tolerance while maintaining vigilance
The microbiome helps prevent infection and inappropriate inflammation. This is why excessive cleansing, sterilisation or repeated antibiotic exposure can paradoxically increase skin vulnerability.
Microbiome composition changes across the lifespan and in response to environmental and physiological factors. Colonisation begins at birth, stabilises through childhood and alters again with ageing as sebum production, immune surveillance and barrier integrity decline. Hygiene practices, climate, occupation, occlusion, medications and chronic disease all influence microbial balance. When this equilibrium is disrupted (dysbiosis), barrier function weakens and the risk of inflammatory or infectious skin conditions increases, underscoring the microbiome’s central role in skin health.
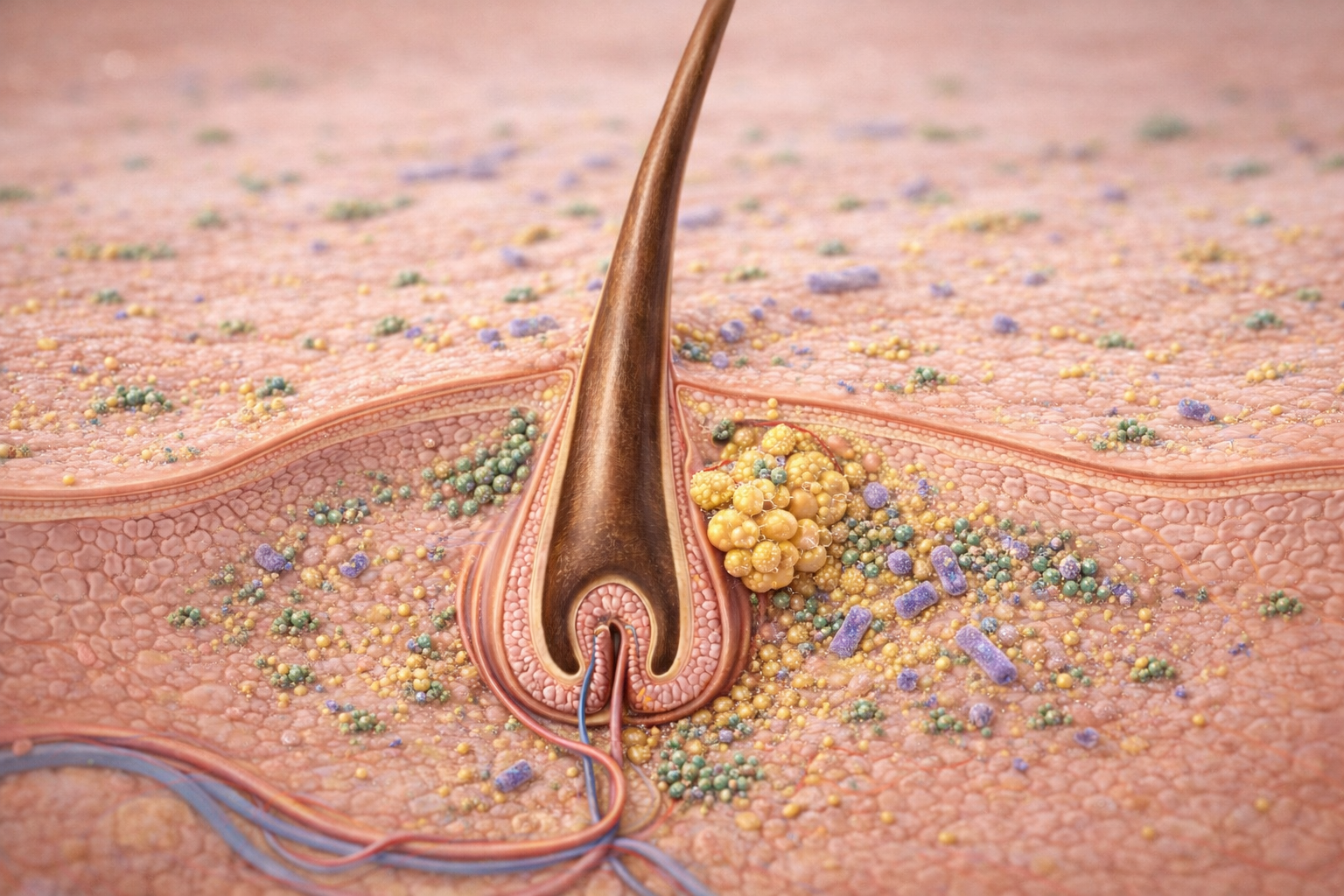
Image: The skin microbiome consists of diverse microorganisms that live primarily on the surface and within hair follicles, particularly in sebum-rich areas. These microbes form a dynamic ecosystem that contributes to skin health by interacting with the local environment and helping maintain barrier function.
Beyond the Basics
Diversity of Microbial Communities
The skin hosts a remarkably diverse range of microorganisms, with bacteria forming the largest proportion of this ecosystem. Common commensal species include Staphylococcus epidermidis, Cutibacterium acnes (formerly Propionibacterium acnes) and Corynebacterium species. These organisms are not randomly distributed. Each is adapted to specific skin environments shaped by moisture, lipid availability and oxygen levels. For example, C. acnes thrives within sebaceous follicles where lipids are abundant, while Staphylococcus species are more prominent on dry, exposed surfaces.
Bacteria coexist with other microbial groups that further enrich the skin ecosystem. Lipophilic fungi, particularly Malassezia species, metabolise skin lipids and interact closely with both keratinocytes and immune cells. Viruses are also present, including bacteriophages that infect resident bacteria and subtly regulate bacterial population balance. In addition, microscopic arthropods such as Demodex mites inhabit hair follicles and sebaceous glands. Together, these bacteria, fungi, viruses and mites form complex, interdependent communities that collectively constitute the skin microbiome.
The Microbiome and Barrier Defence
The skin microbiome is a key component of innate barrier defence. By occupying physical and nutritional niches on the skin surface, commensal organisms limit the ability of pathogens to establish themselves, a process known as colonisation resistance. This protective effect is both physical and biochemical.
Many commensal microbes actively produce antimicrobial peptides, fatty acids and metabolic by-products that inhibit the growth of harmful organisms. Sebum and sweat supply nutrients that shape which microbes dominate particular regions, while microbial metabolism in turn contributes to maintenance of the skin’s slightly acidic surface pH. This acidic environment is hostile to many pathogens and supports optimal barrier enzyme function, reinforcing the link between microbial balance and physical skin integrity.
Immune System Interactions
Microbial residents play an essential role in shaping and regulating cutaneous immune responses. Keratinocytes, Langerhans cells and other immune cells continuously sample microbial signals through pattern-recognition receptors. This allows the immune system to distinguish between harmless commensals and potentially dangerous invaders.
When the microbiome is balanced, these interactions promote immune tolerance and prevent unnecessary inflammation. When balance is disrupted—through antibiotics, harsh cleansing practices, barrier injury or disease-immune signalling can become exaggerated, leading to inflammation and tissue damage. Certain commensals, including Staphylococcus epidermidis, actively support wound healing by stimulating keratinocyte migration and coordinating early immune responses. This illustrates that the microbiome is not passive but deeply integrated into normal skin physiology and repair.
Microenvironmental Variation Across the Body
Microbial composition varies substantially across different body sites due to differences in temperature, moisture, sebum production and mechanical stress. Oily regions such as the face, scalp and upper back favour lipid-dependent organisms, while moist areas including the axillae, groin and inframammary folds support distinct bacterial and fungal populations. Dry regions, such as the forearms and lower legs, typically host fewer microbes but greater overall diversity.
These patterns are further shaped by individual behaviours and environmental exposure. Clothing, detergents, skincare products, climate, occupation and even habitual hand use influence microbial communities over time. Importantly, microbial composition can differ subtly between symmetrical body sites, reflecting the highly localised nature of skin ecology.
Dysbiosis and Disease
Disruption of the normal microbiome, referred to as dysbiosis, is increasingly recognised as a contributing factor in many skin diseases. Acne involves shifts in C. acnes strain composition and inflammatory signalling within sebaceous follicles rather than simple bacterial overgrowth. Atopic dermatitis is associated with reduced microbial diversity and dominance of Staphylococcus aureus, which worsens barrier dysfunction and inflammation.
Psoriasis is linked to altered microbial signatures that may influence immune activation, although cause-and-effect relationships remain under investigation. In wounds, dysbiosis reflects a transition from protective commensal populations to pathogenic dominance, often contributing to delayed healing. Understanding these patterns has led to emerging therapeutic approaches, including microbiome-friendly skincare, targeted antimicrobials, probiotics and experimental microbiome transplantation strategies.
Clinical Connections
The skin microbiome plays an increasingly important role in dermatology, wound care and general nursing practice. Disruption of resident microbial communities can impair barrier function, alter local immune responses and increase susceptibility to inflammation and infection, often before visible skin breakdown occurs.
Clinical factors that commonly disturb the skin microbiome include:
Frequent use of antimicrobial or alkaline cleansers that reduce microbial diversity and increase skin pH
Prolonged occlusion from dressings, incontinence products or medical devices that create warm, moist environments favouring pathogenic overgrowth
Broad-spectrum antibiotic exposure that disrupts commensal populations
Repeated friction, pressure or barrier injury that allows abnormal colonisation
In pressure injuries and chronic wounds, microbial imbalance is common and contributes to persistent inflammation, excessive protease activity and delayed granulation and epithelialisation. Importantly, dysbiosis may exist without overt signs of infection, complicating assessment and reinforcing the need for careful wound monitoring rather than reflex antimicrobial use.
Immunosuppressed individuals, including people with diabetes, those undergoing chemotherapy and older adults, are particularly vulnerable to microbiome shifts that permit pathogenic colonisation. In these populations, maintaining skin hydration, minimising unnecessary antimicrobial exposure and supporting barrier integrity are essential components of care.
Clinical practice is increasingly shifting from indiscriminate eradication toward preservation and support of protective microbial communities. This has implications for cleanser selection, dressing choice, infection prevention strategies and long-term skin health management.
Concept Check
How do commensal microbes contribute to the skin’s barrier function?
Why does microbiome composition vary between oily, moist and dry body regions?
How do keratinocytes and immune cells interact with the microbiome to maintain immune balance?
What factors commonly lead to dysbiosis, and how does this affect skin health?
Why are chronic wounds particularly susceptible to microbiome imbalance?