WOUND HEALING — Stage 2: Inflammation
The inflammatory stage of wound healing begins immediately after haemostasis and typically lasts from several hours to several days. Although often portrayed simply as a period of “cleaning,” inflammation is a highly organised and indispensable biological response that determines whether healing will proceed normally, stagnate or fail entirely.
This phase recruits immune cells to the wound bed, eliminates pathogens and debris, and establishes the biochemical environment required for tissue regeneration. Inflammation is therefore both protective and preparatory, ensuring microbial control while initiating the signalling networks that drive proliferation and angiogenesis. A balanced, time-limited inflammatory response is essential. Too little inflammation increases infection risk and delays granulation tissue formation. Too much, or inflammation that persists, contributes to chronic, non-healing wounds.
What You Need to Know
The inflammatory phase of wound healing begins immediately after haemostasis and is essential for clearing contamination and preparing the wound for repair. Chemical signals released from platelets, damaged tissue, and activated complement proteins create a strong chemotactic gradient that draws immune cells into the wound site. This response is protective and time-limited, designed to remove threats rather than rebuild tissue.
Several coordinated processes define this phase:
Rapid recruitment of neutrophils to phagocytose bacteria and cellular debris
Transition to macrophage dominance for ongoing clearance and regulation
Increased vascular permeability allowing plasma proteins to enter the wound
Neutrophils are the first immune cells to arrive, appearing within minutes to hours. They provide early defence through phagocytosis and release of antimicrobial enzymes and reactive oxygen species. As inflammation progresses, macrophages become the predominant cell type. In addition to clearing debris, macrophages act as key regulators by releasing growth factors that activate fibroblasts, stimulate new blood vessel formation, and promote keratinocyte migration. At the same time, increased vascular permeability allows immunoglobulins, fibrinogen, and complement proteins to enter the wound, producing the exudate commonly seen in early healing.
The inflammatory phase therefore represents a critical transition from containment to repair. While redness, warmth, swelling, and exudate are normal features of this stage, prolonged or excessive inflammation can impair healing. Effective progression through this phase is essential for successful entry into the proliferative stage, where tissue regeneration and wound closure begin.
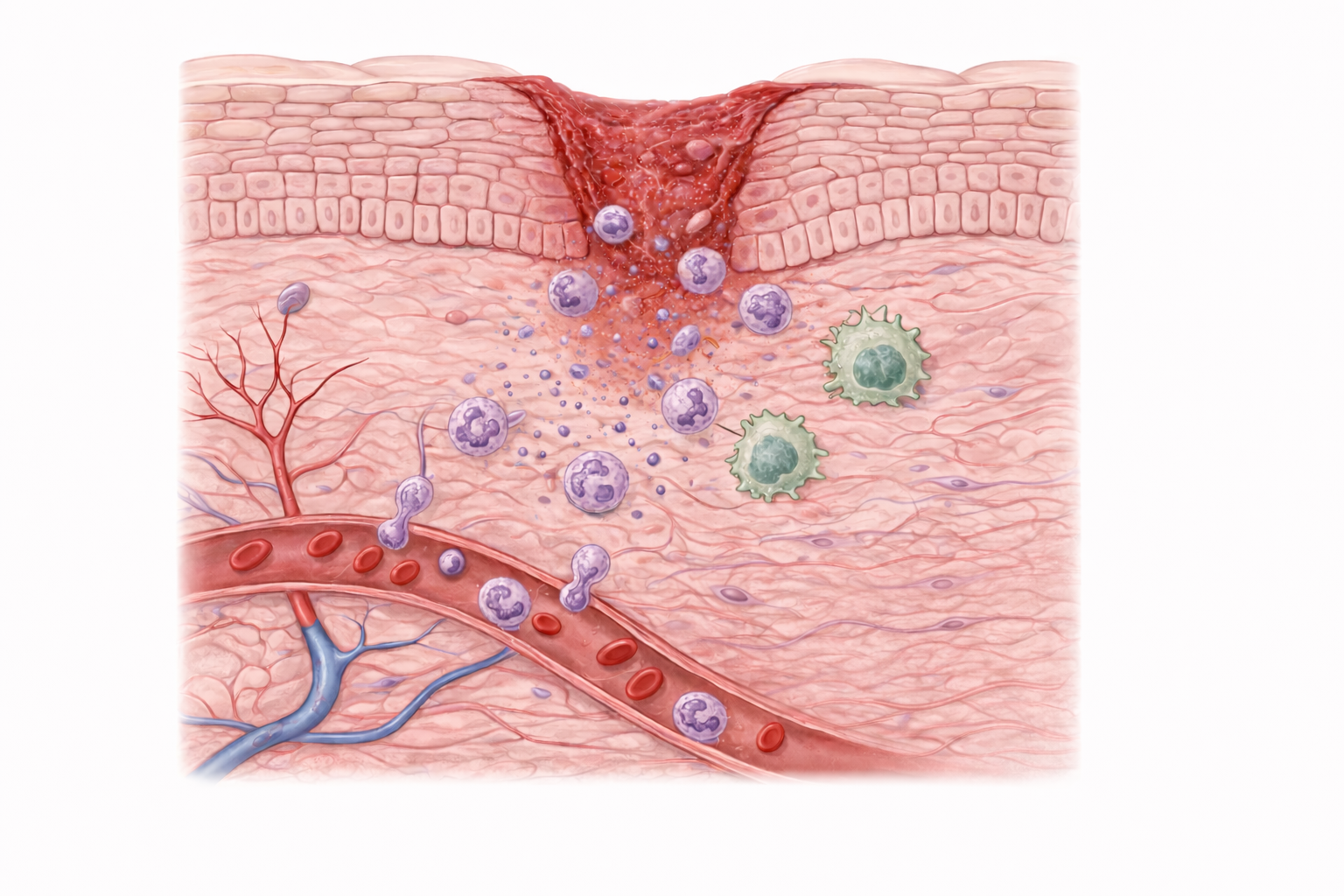
Image: Neutrophils (purple cells) leaving dilated blood vessels and migrating toward the wound, where they begin clearing debris and microorganisms as part of the inflammatory response. Macrophages (large green cells) continue clearing debris while releasing signals that regulate inflammation and initiate tissue repair.
Beyond the Basics
Activation of the inflammatory response
The inflammatory response is initiated almost immediately by signals originating from the haemostatic clot and injured tissue. Platelets trapped within the fibrin clot release a range of growth factors and cytokines, including platelet-derived growth factor, transforming growth factor beta, interleukin-1, and chemokines that establish a strong chemotactic gradient. At the same time, damaged keratinocytes release intracellular danger signals known as alarmins, such as HMGB1, which alert the immune system to tissue injury rather than infection alone.
Complement activation during clot formation further amplifies this response. Complement fragments such as C3a and C5a act as powerful chemoattractants for neutrophils and increase vascular permeability. Together, these signals ensure that immune cells are rapidly recruited in a controlled and spatially localised manner, allowing the wound to be protected from infection while avoiding excessive or systemic inflammation.
Neutrophil recruitment and early wound defence
Neutrophils are the first immune cells to arrive at the wound site, typically within minutes to hours. Their entry follows a well-coordinated sequence involving margination along the vessel wall, rolling mediated by selectins, firm adhesion through integrins, and migration across the endothelium by diapedesis. This tightly regulated process allows neutrophils to exit the circulation precisely where they are needed.
Once within the wound bed, neutrophils act as short-lived but highly effective defenders. They phagocytose bacteria and cellular debris, release reactive oxygen species to destroy pathogens, and secrete proteolytic enzymes that degrade damaged extracellular matrix. In settings of severe contamination, neutrophils may also form neutrophil extracellular traps, which immobilise and kill microbes. Although these actions cause collateral tissue damage, this is an expected and necessary component of early wound cleaning. Crucially, neutrophils undergo programmed apoptosis after completing their task, signalling that the wound is ready to transition away from destructive inflammation.
Macrophages and regulation of the inflammatory phase
Macrophages are the central coordinators of wound healing and represent the pivotal cell type of the inflammatory phase. They enter the wound as circulating monocytes and differentiate in response to local cues. Early in inflammation, macrophages adopt a pro-inflammatory phenotype that supports continued debris clearance and microbial defence through phagocytosis and production of inflammatory mediators such as tumour necrosis factor alpha and interleukin-1 beta.
As neutrophils undergo apoptosis and platelet-derived growth factors accumulate, macrophages shift toward a reparative phenotype. This phenotypic transition marks a decisive turning point in healing. Reparative macrophages reduce inflammatory signalling and instead release growth factors that drive tissue repair. These include vascular endothelial growth factor to stimulate angiogenesis, transforming growth factor beta to promote fibroblast recruitment and collagen synthesis, fibroblast growth factor and platelet-derived growth factor to support fibroblast proliferation, and interleukin-10 to actively suppress ongoing inflammation. Without this macrophage transition, wounds remain trapped in an inflammatory state and fail to progress.
Vascular permeability and wound exudate
Inflammation is accompanied by pronounced vascular changes driven by mediators such as histamine, prostaglandins, and bradykinin. These mediators increase capillary permeability, allowing plasma proteins to leak into the wound space. Immunoglobulins, complement proteins, and fibrinogen enter the wound bed, contributing to immune defence and structural support.
The resulting wound exudate plays an important functional role in acute healing. It creates a moist environment that facilitates immune cell movement, supports enzymatic breakdown of necrotic tissue, and prevents desiccation of the wound surface. However, while exudate is beneficial in the short term, excessive or persistent exudate reflects prolonged inflammation and is a hallmark of chronic, non-healing wounds.
Transition from inflammation to proliferation
Resolution of inflammation is not passive but actively regulated. As neutrophils are cleared and macrophages adopt a reparative phenotype, pro-inflammatory cytokine levels decline and growth factor signalling becomes dominant. Fibroblasts are recruited into the wound, angiogenesis is initiated, and keratinocyte migration is stimulated.
This transition represents the handover from immune defence to tissue reconstruction. Failure of this shift, whether due to ongoing infection, ischaemia, repeated trauma, or systemic disease, results in wounds that remain locked in the inflammatory phase. Understanding this transition explains why chronic wounds are characterised not by an absence of healing signals, but by persistent inflammation that overwhelms the normal progression to repair.
Clinical Connections
The inflammatory phase of wound healing is particularly vulnerable to disruption by systemic factors, and when inflammation becomes ineffective or prolonged, healing stalls. Many patients with non-healing wounds do not lack an inflammatory response; rather, the response is poorly regulated, excessive, or unable to transition into repair.
In clinical practice, altered inflammation most often presents through the following patterns:
Impaired immune cell function and delayed inflammatory resolution
Persistent inflammatory signalling with excessive protease activity
Failure to transition from inflammation to proliferation
Systemic conditions strongly influence these patterns. Diabetes impairs neutrophil chemotaxis and phagocytosis, disrupts macrophage signalling, and delays the critical shift from inflammatory to reparative macrophage phenotypes. As a result, inflammation persists without effectively clearing debris or initiating repair. Ageing and malnutrition blunt inflammatory responses through reduced cellular activity and diminished cytokine production, while corticosteroid therapy suppresses immune cell recruitment and growth factor release, further delaying progression through this phase.
Chronic wounds such as venous leg ulcers and diabetic foot ulcers are characterised by a dysregulated inflammatory environment rather than a normal, time-limited response. These wounds contain excessive numbers of neutrophils, persistently high levels of pro-inflammatory cytokines, and elevated protease activity that continuously degrades extracellular matrix and growth factors. This ongoing tissue breakdown prevents fibroblast activity, angiogenesis, and epithelial migration, effectively trapping the wound in inflammation.
Recognising where a wound sits within the inflammatory phase is essential for guiding management. Clinical decisions around debridement, infection control, moisture balance, and dressing selection should aim to reduce excessive inflammation while supporting the transition to repair. Interventions that reset the wound environment allow inflammatory processes to resolve appropriately, creating the conditions necessary for progression into the proliferative stage and successful healing.
Concept Check
Why is neutrophil apoptosis essential for the transition to later healing stages?
How do macrophages regulate both inflammation and repair?
What structural and biochemical features define wound exudate during inflammation?
How does complement activation contribute to early inflammatory responses?
Why is prolonged inflammation a hallmark of chronic wounds?