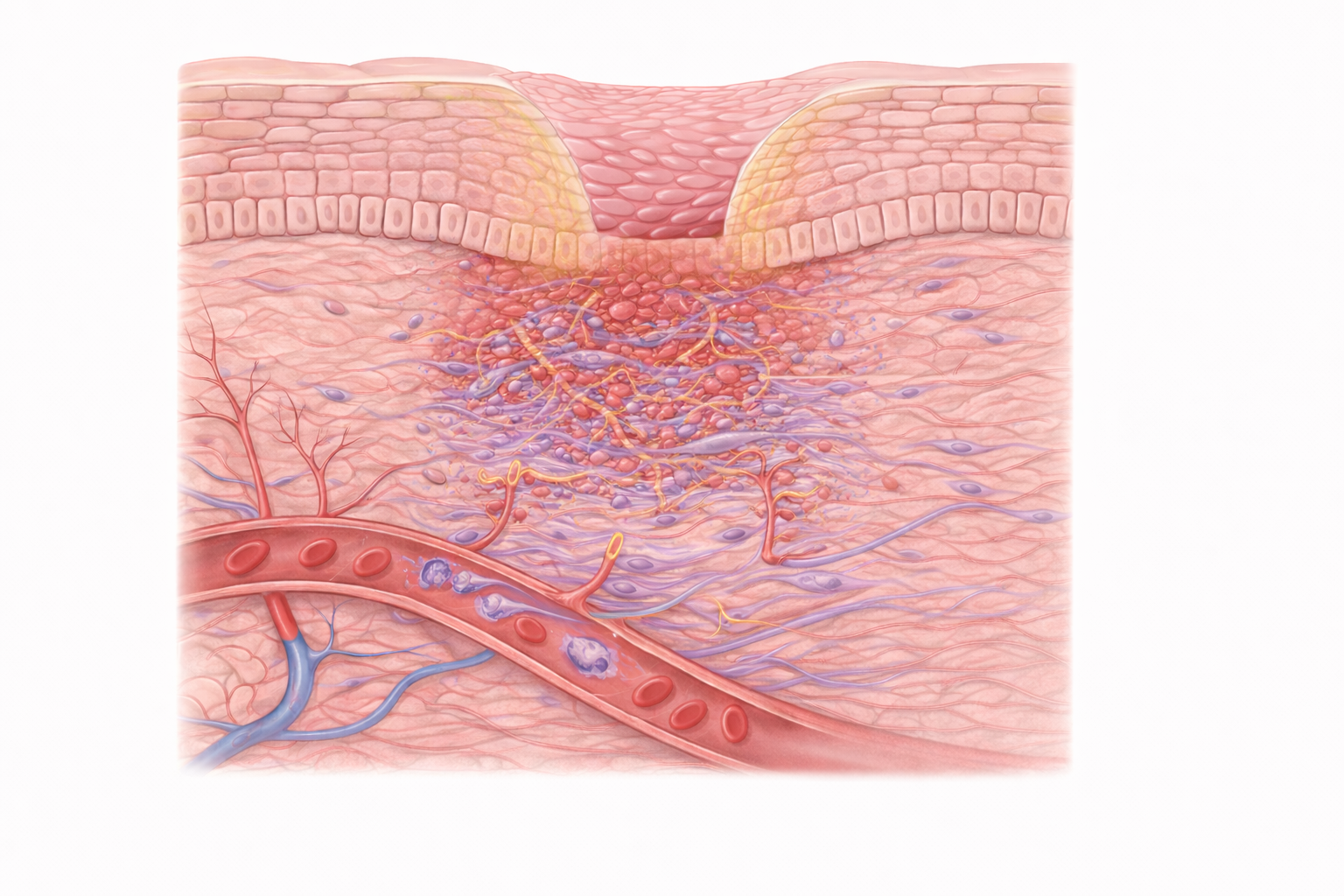
WOUND HEALING — Stage 3: Proliferation
The proliferative stage marks the transition from inflammation to active tissue reconstruction. Beginning around day 3 after injury and lasting several weeks, this phase is characterised by the coordinated activity of fibroblasts, endothelial cells, keratinocytes and myofibroblasts. The primary goals of proliferation are to rebuild structural support through granulation tissue, restore microvascular networks through angiogenesis, re-establish the epidermal barrier through re-epithelialisation and reduce wound size through contraction.
This is the stage where the wound visibly transforms, becoming red, granular and moist as new tissue fills the defect. The success of the proliferative phase determines the integrity of the final scar and the ability of the wound to resist infection, mechanical stress and desiccation.
What You Need to Know
The proliferative phase of wound healing focuses on rebuilding tissue and restoring surface continuity after inflammation has cleared debris and controlled contamination. This stage is driven largely by growth factors released from macrophages, including vascular endothelial growth factor, platelet-derived growth factor, transforming growth factor beta, and fibroblast growth factor. These signals activate fibroblasts, endothelial cells, and keratinocytes, coordinating repair across the wound bed.
Four key processes occur during proliferation and overlap in time:
Formation of granulation tissue by proliferating fibroblasts
Angiogenesis through endothelial cell sprouting and capillary growth
Re-epithelialisation via keratinocyte migration across the wound surface
Wound contraction mediated by myofibroblasts
Fibroblasts migrate into the wound and deposit a provisional extracellular matrix rich in collagen type III, fibronectin, and proteoglycans, forming granulation tissue. This tissue replaces the fibrin clot and provides a temporary scaffold that supports cell migration and new blood vessel growth. Angiogenesis rapidly increases oxygen and nutrient delivery, giving granulation tissue its characteristic red, granular appearance. At the surface, keratinocytes migrate from the wound edges and surrounding epidermis to restore epithelial continuity, while myofibroblasts generate contractile forces that draw wound edges inward and reduce the area requiring closure.
Although the tissue produced during the proliferative phase is structurally immature and mechanically weak, it is essential for successful healing. Granulation tissue establishes the vascular and cellular framework that supports epithelial closure and prepares the wound for the final remodelling phase, during which collagen is reorganised and tensile strength gradually increases.
Image: Keratinocytes migrate across the wound surface to restore the epidermis, while fibroblasts deposit new matrix and small blood vessels grow into the area, forming granulation tissue and supporting tissue repair.
Beyond the Basics
Granulation tissue as the scaffold for repair
Granulation tissue is the defining feature of the proliferative phase and represents the first true replacement of the provisional fibrin clot. It is composed of fibroblasts, newly formed capillaries, residual inflammatory cells, and a hydrated extracellular matrix rich in collagen type III, proteoglycans, and hyaluronic acid. Fibroblasts migrate into the wound in response to growth factors such as platelet-derived growth factor and transforming growth factor beta, using fibrin strands from the original clot as a temporary migration scaffold.
Once established in the wound bed, fibroblasts synthesise extracellular matrix components that gradually replace the fibrin framework. Early granulation tissue is loose and highly permeable, allowing rapid movement of cells and diffusion of nutrients. As matrix deposition continues, the tissue becomes progressively denser and more organised, providing mechanical support and forming the template for later collagen remodelling.
Angiogenesis and restoration of blood supply
Angiogenesis is essential for sustaining granulation tissue and supporting the high metabolic demands of repair. Signals released during inflammation, particularly vascular endothelial growth factor, fibroblast growth factor, and angiopoietins, stimulate endothelial cells in nearby vessels to degrade their basement membranes, proliferate, and sprout into the wound.
These newly formed capillary loops give healing wounds their characteristic red, granular appearance. Although fragile and leaky, they deliver oxygen and nutrients to fibroblasts and keratinocytes and remove metabolic waste. Local hypoxia and lactate accumulation within the wound further stimulate angiogenic signalling, creating a positive feedback loop that promotes vascular ingrowth. Without adequate angiogenesis, granulation tissue cannot be sustained and the wound fails to progress.
Fibroblast activity and provisional collagen deposition
Fibroblasts are the primary matrix-producing cells during proliferation and act as the architects of early tissue repair. Under the influence of transforming growth factor beta, fibroblasts synthesise type III collagen, which forms the initial structural framework of the healing tissue. This collagen is thinner and more flexible than the type I collagen that predominates in uninjured skin, allowing the wound to remain adaptable during active cell migration and vascular growth.
In addition to collagen, fibroblasts produce fibronectin, a key adhesive glycoprotein that anchors cells to the extracellular matrix and guides directional migration. As the wound environment matures, fibroblasts respond to mechanical tension and biochemical cues by differentiating into myofibroblasts, linking matrix production with force generation.
Myofibroblasts and wound contraction
Myofibroblasts play a central role in reducing wound size through contraction. These cells contain actin and myosin filaments similar to those found in smooth muscle, enabling them to generate contractile force. By anchoring to the extracellular matrix via integrins and fibronectin, myofibroblasts pull the wound edges inward and align collagen fibres along lines of tension.
Wound contraction can significantly reduce the surface area requiring re-epithelialisation, particularly in large or deep wounds. The extent of contraction is influenced by tissue type, wound location, and mechanical tension within the surrounding skin. When contraction is excessive or poorly regulated, particularly in burns or wounds over joints, functional impairment and contractures may develop.
Re-epithelialisation and restoration of the surface barrier
Re-epithelialisation restores the epidermal barrier and is driven by keratinocyte migration from the wound edges and from surviving skin appendages such as hair follicles and sweat glands. Keratinocytes undergo cytoskeletal reorganisation that allows them to flatten and migrate as a coordinated sheet across the granulation tissue. During migration, they release proteases that clear residual debris and temporarily loosen cell-to-cell adhesions.
Once the wound surface is covered, keratinocytes re-establish tight junctions, stratify, and differentiate to recreate the layered structure of the epidermis. This process is highly dependent on adequate oxygenation and a moist wound environment. Excessive dryness impairs keratinocyte movement, while excessive exudate can destabilise the fragile new epithelial layer.
Integration and timing of proliferative processes
Although granulation, angiogenesis, collagen deposition, contraction, and re-epithelialisation are often described separately, they occur concurrently and are tightly coordinated. Fibroblasts remodel the matrix as keratinocytes migrate, capillaries grow in response to metabolic demand, and myofibroblasts contract the wound as collagen is deposited and reorganised.
The balance and timing of these processes determine whether healing proceeds efficiently or becomes dysregulated. Disruption of any component can delay progression or predispose to abnormal scarring. Successful completion of the proliferative phase establishes a stable, vascularised tissue bed that can enter the final remodelling stage, where strength and function are gradually restored.
Clinical Connections
Successful progression through the proliferative phase depends on a supportive local and systemic environment. Adequate oxygen delivery, sufficient nutritional substrates, effective perfusion, and resolution of excessive inflammation are all required for fibroblasts, endothelial cells, and keratinocytes to function effectively. When these conditions are not met, proliferation becomes inefficient and the wound may stall.
In clinical practice, impaired proliferation most commonly presents through the following patterns:
Poor or fragile granulation tissue formation
Excessive granulation that interferes with epithelial migration
Disrupted moisture balance affecting cell movement and tissue integrity
Systemic conditions strongly influence these outcomes. Diabetes impairs angiogenesis and fibroblast activity, while anaemia and peripheral vascular disease reduce oxygen delivery to the wound bed. Smoking further compromises healing by causing vasoconstriction and reducing oxygen availability. Together, these factors lead to weak, pale granulation tissue that lacks the vascular support needed for sustained repair.
Local wound conditions also play a critical role. Hypergranulation occurs when granulation tissue rises above the level of the surrounding skin, physically blocking keratinocyte migration and delaying surface closure. In contrast, insufficient granulation leaves underlying tissue exposed, increasing susceptibility to infection and mechanical trauma. Moisture balance is equally important, as dry wound beds inhibit keratinocyte movement, while excessive moisture causes maceration and breakdown of newly formed tissue.
Careful assessment during this phase helps determine whether healing is progressing appropriately. Observing granulation tissue colour, texture, vascularity, and coverage provides insight into perfusion, oxygenation, and cellular activity. These observations guide decisions around dressing selection, moisture management, and the need for interventions that support angiogenesis, control inflammation, or protect fragile new tissue as the wound transitions toward remodelling.
Concept Check
What signalling mechanisms stimulate fibroblasts to migrate into the wound and begin building granulation tissue?
Why is angiogenesis essential for sustaining the proliferative phase?
How do myofibroblasts contribute to reducing wound size, and what complications can arise from excessive contraction?
What cellular changes allow keratinocytes to migrate across the wound bed during re-epithelialisation?
How do oxygenation and moisture levels influence the speed and quality of proliferation?