LYMPH NODES: STRUCTURE & FUNCTION
Lymph nodes are small, bean-shaped organs distributed along the lymphatic vessels. They act as biological “checkpoints,” filtering lymph and monitoring it for pathogens, foreign material and abnormal cells. As lymph passes through a node, specialised immune cells identify and respond to potential threats. This makes lymph nodes essential not only for fluid filtration but for initiating adaptive immune responses. Their strategic placement ensures that harmful substances entering tissues do not reach the bloodstream without immune scrutiny.
What You Need to Know
Lymph nodes are small, encapsulated organs positioned along lymphatic vessels that function as filtration and immune coordination centres. As lymph drains from tissues, it enters lymph nodes through afferent lymphatic vessels and flows slowly through an internal network of sinuses. This slowed movement maximises contact between lymph contents and immune cells, allowing pathogens, abnormal cells and debris to be identified before lymph returns to circulation.
Each lymph node has a highly organised internal structure that supports different stages of immune activation. The outer cortex contains lymphoid follicles rich in B cells, where antibody-producing responses are initiated. Deeper within the node, the paracortex is dominated by T cells and dendritic cells and serves as the primary site for antigen presentation and T cell activation. The medulla forms the final processing zone, containing macrophages that remove debris and plasma cells that secrete antibodies into the lymph.
This compartmentalisation allows lymph nodes to perform several linked functions:
Filter lymph and trap pathogens, foreign particles and malignant cells
Activate and expand specific T and B lymphocyte populations
Coordinate local and systemic immune responses through cytokine signalling
Lymph enters a node through multiple afferent vessels but exits through a single efferent vessel at the hilum. This arrangement further slows lymph flow and ensures thorough immune surveillance. When immune activation occurs, lymph nodes enlarge due to lymphocyte proliferation and increased cellular traffic, making them important indicators of infection, inflammation or malignancy on clinical examination.
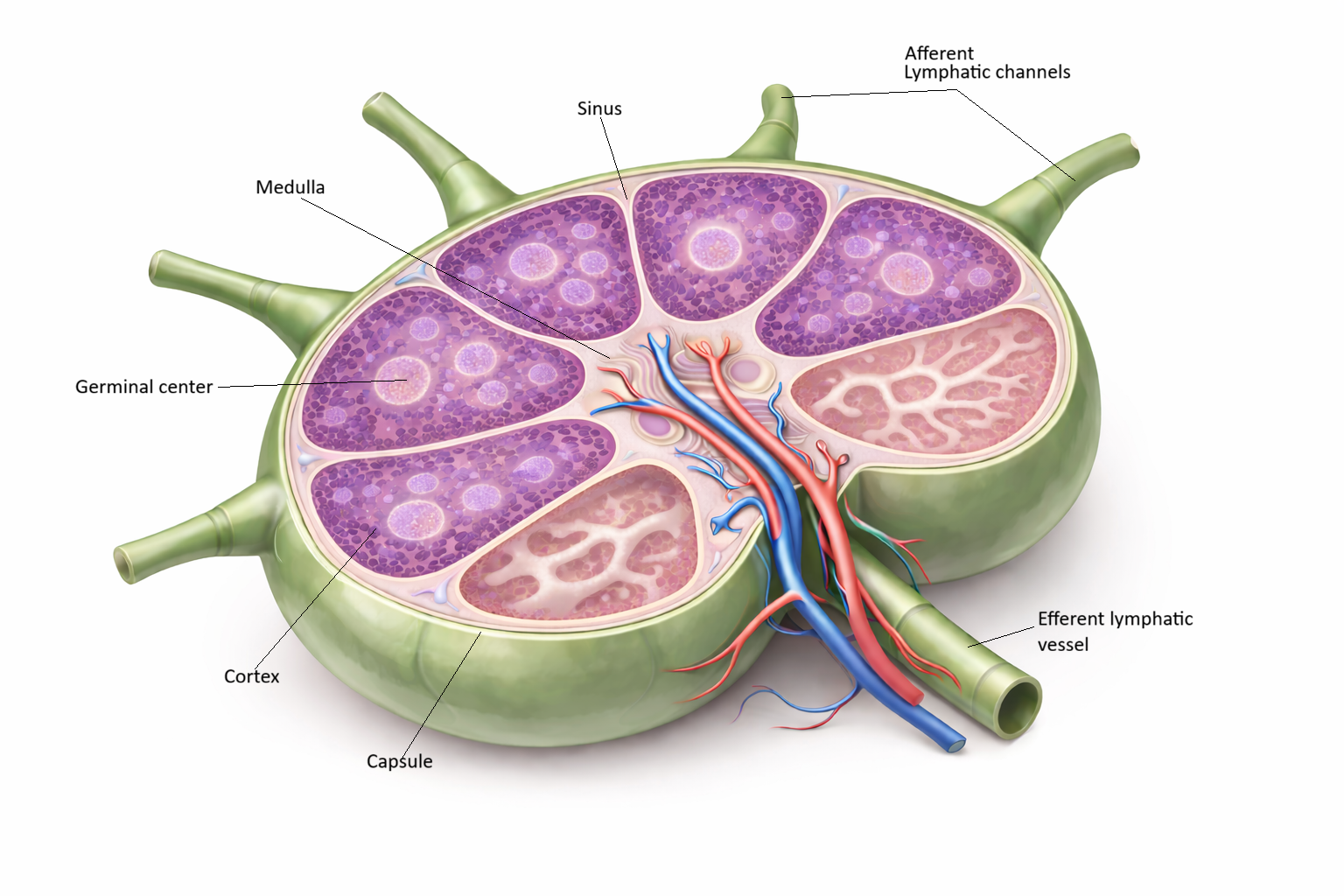
Image: A cross-sectional view of a lymph node showing its organised internal structure. The outer cortex contains lymphoid follicles with pale germinal centres, while the inner medulla consists of branching sinuses and cords. Lymph enters via afferent vessels on the convex surface, filters through the node, and exits at the hilum via the efferent lymphatic vessel.
Beyond the Basics
Anatomical Organisation of a Lymph Node
Lymph nodes are encapsulated, highly organised structures designed to maximise interaction between lymph and immune cells. A dense connective tissue capsule surrounds each node and provides protection and shape. From this capsule, trabeculae extend inward, creating a supportive framework that guides lymph through the internal compartments rather than allowing it to pass straight through unchecked.
Just beneath the capsule lies the cortex, which contains lymphoid follicles populated predominantly by B lymphocytes. Some follicles develop germinal centres, dynamic regions where B cells proliferate, undergo affinity maturation and differentiate into plasma cells. These processes allow the immune system to refine antibody responses and produce antibodies that bind antigens with increasing precision over time.
Deeper within the node is the paracortex, a region rich in T lymphocytes and dendritic cells. This area functions as the primary site of T cell activation. Dendritic cells that have captured antigen in peripheral tissues migrate to the paracortex, where they present antigen fragments to naïve T cells. Successful interactions here lead to T cell activation, clonal expansion and differentiation into effector and memory cells, forming the backbone of adaptive cellular immunity.
The medulla forms the innermost region of the lymph node and contains medullary cords interspersed with medullary sinuses. Plasma cells within the cords secrete antibodies directly into the lymph, while macrophages remove remaining debris, pathogens and abnormal cells. By the time lymph reaches this region, it has already been extensively sampled and processed, making the medulla the final checkpoint before lymph exits the node.
Directed Lymph Flow and Filtration
Lymph enters each lymph node through multiple afferent lymphatic vessels that penetrate the capsule. From there, it flows into the subcapsular sinus and then slowly percolates through cortical and medullary sinuses. This controlled, low-velocity movement ensures prolonged exposure of lymph contents to immune cells at each stage. Filtered lymph exits the node through a single efferent vessel at the hilum, reinforcing the node’s role as a bottleneck for immune surveillance rather than a passive conduit.
Immune Surveillance and Activation
As lymph moves through the node, macrophages capture and phagocytose foreign particles, while dendritic cells present processed antigens to T cells in the paracortex. B cells encountering antigen within follicles migrate into germinal centres, where they undergo proliferation, somatic hypermutation and class-switch recombination. These tightly regulated interactions allow lymph nodes to generate targeted immune responses that are both specific and adaptable, rather than indiscriminate.
High Endothelial Venules and Lymphocyte Recirculation
Lymph nodes also receive a continuous supply of naïve lymphocytes directly from the bloodstream. High endothelial venules, specialised blood vessels located primarily in the paracortex, enable lymphocytes to exit circulation and enter the node. This constant recirculation ensures that a diverse pool of T and B cells is always present and ready to respond to antigens arriving via lymph. The combination of lymph-borne antigen delivery and blood-borne lymphocyte entry makes lymph nodes highly efficient immune coordination centres.
Clinical Connections
Changes in lymph node size, texture and tenderness provide valuable clues about underlying pathology. Lymphadenopathy, whether localised or generalised, often reflects active immune responses as lymphocytes proliferate and traffic through nodes during infection or inflammation. Tender, mobile nodes are more commonly associated with acute infection, whereas nodes that are firm, fixed or painless raise concern for malignancy, including lymphoma or metastatic disease spreading along predictable lymphatic pathways.
Patterns of lymph node involvement help narrow differential diagnoses:
Localised enlargement linked to infection or inflammation within a specific drainage region
Generalised lymphadenopathy seen in systemic conditions such as viral illness, autoimmune disease or haematological malignancy
Firm, non-tender nodes suggesting malignant infiltration or chronic disease
Matted or fixed nodes indicating possible tumour spread or fibrosis
Disruption of lymph node structure or removal of nodes has important consequences for lymph flow. Surgical lymph node dissection and radiotherapy, commonly used in cancer treatment, can interrupt normal drainage pathways and lead to lymphoedema. This complication is particularly relevant after breast, pelvic or gynaecological cancer treatment and requires ongoing monitoring, patient education and preventive care to reduce infection risk.
Concept Check
What are the primary regions of a lymph node, and which immune cells reside in each?
Why is lymph flow deliberately slow within a lymph node?
What occurs within germinal centres during an immune response?
How do high endothelial venules contribute to lymphocyte circulation?
What is the functional difference between afferent and efferent lymphatic vessels?