Microscopic Structure of Lymphatic Tissues and Vessels
The lymphatic system is composed of a network of vessels, nodes and specialised tissues that maintain fluid balance and support immune function. Histology reveals how these structures are organised at the microscopic level and how their cellular components enable filtration, antigen presentation and lymphocyte activation.
Understanding lymphatic microanatomy bridges normal physiology with the clinical presentation of lymphatic disorders, infections and malignancies such as lymphoma.
What You Need to Know
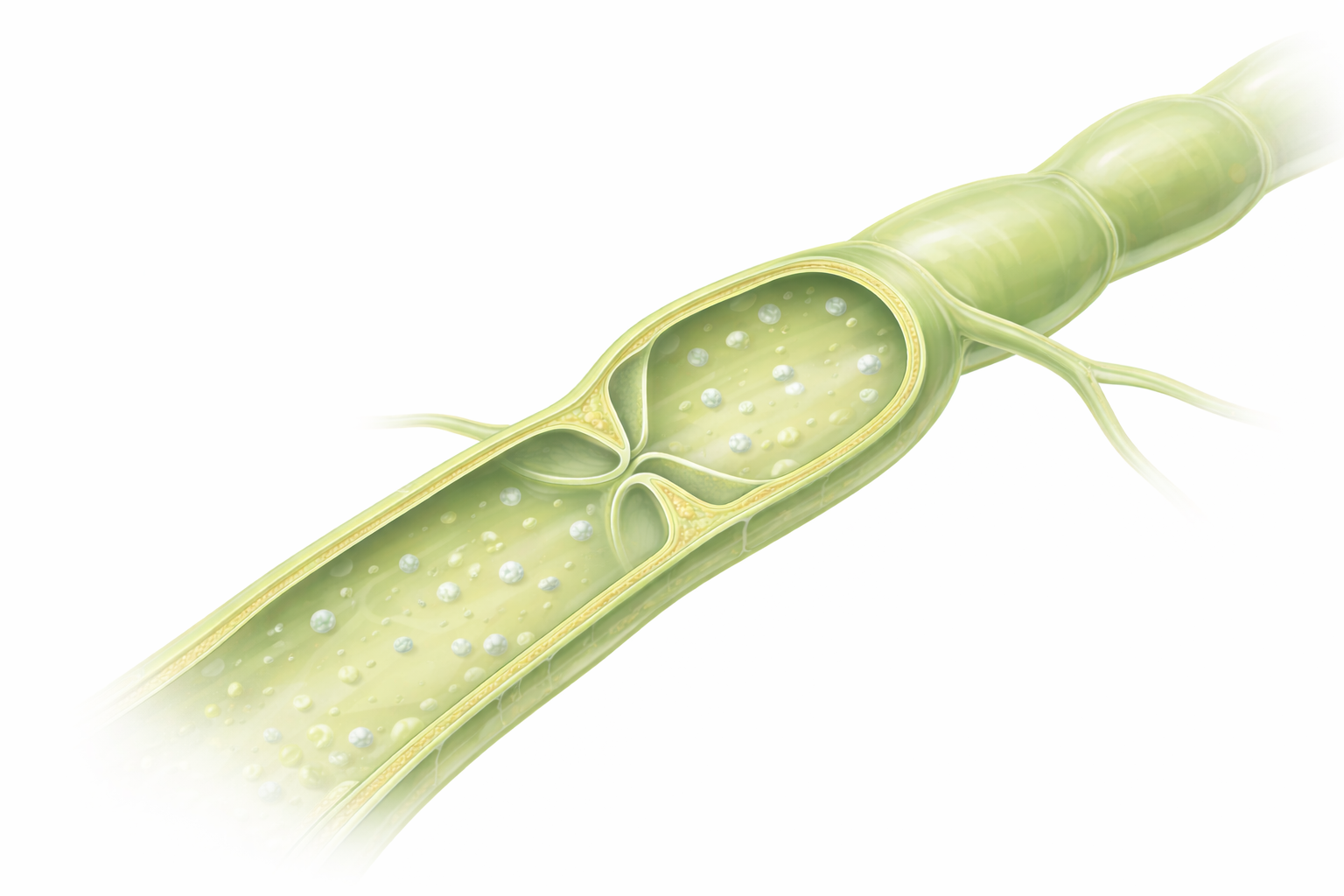
Lymphatic tissues and vessels have a distinctive microscopic structure that supports their role in fluid regulation, immune surveillance and cell transport. Lymphatic vessels begin as blind-ended capillaries embedded within tissues. Their walls are extremely thin and formed by overlapping endothelial cells that function like one-way entry flaps, opening when interstitial pressure rises and closing to prevent backflow. This design allows excess fluid, plasma proteins, immune cells and larger particles to enter the lymphatic system efficiently without requiring active transport.
As lymph moves away from tissues, lymphatic capillaries merge into larger collecting vessels. These vessels resemble small veins but contain more valves and a thinner wall structure. Smooth muscle within the vessel wall contracts rhythmically, helping propel lymph forward in combination with skeletal muscle movement and pressure changes from breathing. This low-pressure system ensures steady lymph flow despite the absence of a central pump.
Lymph nodes act as specialised filtration and processing centres, and their microscopic organisation reflects this function. Each node is enclosed by a fibrous capsule and divided internally into distinct regions, each supporting different immune activities:
The cortex contains lymphoid follicles rich in B cells, many with germinal centres where B cells proliferate and mature
The paracortex is dominated by T cells and dendritic cells, making it the primary site of antigen presentation to T lymphocytes
The medulla contains medullary cords and sinuses, populated by macrophages and plasma cells that filter lymph and secrete antibodies
Lymph enters nodes through afferent lymphatic vessels and flows slowly through a network of sinuses, allowing close contact between antigens and immune cells. This controlled flow increases the likelihood that pathogens, foreign particles or abnormal cells will be detected before lymph exits the node through efferent vessels. The organised microscopic layout ensures that immune responses are coordinated rather than random, supporting efficient activation while limiting unnecessary inflammation.
Image: Lymphatic vessel showing one-way valves and lymph fluid containing lymphocytes
Beyond the Basics
Lymphatic Vessels
Lymphatic capillaries are the smallest and most permeable vessels in the lymphatic system. They begin as blind-ended tubes embedded within connective tissue and are structurally distinct from blood capillaries. Their walls consist of a single layer of endothelial cells with overlapping edges that act as microscopic one-way flaps. When interstitial pressure rises, these overlaps separate slightly, allowing fluid and dissolved substances to enter. When pressure inside the vessel increases, the flaps close, preventing backflow.
Anchoring filaments connect the endothelial cells to surrounding connective tissue. As tissue swells, these filaments pull the endothelial flaps open rather than allowing the vessel to collapse. The basement membrane is absent or incomplete, which explains why lymphatic capillaries can take up large plasma proteins, immune cells and cellular debris that blood capillaries cannot reclaim. This structural design makes lymphatic capillaries essential for maintaining tissue fluid balance.
Collecting Vessels. As lymphatic capillaries converge, they form collecting vessels with a more organised wall structure. These vessels contain valves that enforce one-way flow and a thin layer of smooth muscle capable of rhythmic contraction. Although they resemble veins, their walls are thinner and their valves more frequent. Lymph movement through collecting vessels relies on intrinsic smooth muscle contraction, skeletal muscle activity, respiratory pressure changes and arterial pulsation in adjacent vessels. This coordinated movement ensures lymph progresses steadily toward lymph nodes and central ducts despite the absence of a central pump.
Lymphatic Trunks and Ducts. Collecting vessels unite to form large lymphatic trunks that drain major anatomical regions. These trunks merge into the thoracic duct or the right lymphatic duct, which represent the final conduits for lymph return to the bloodstream. Their walls contain more developed smooth muscle layers, allowing them to accommodate larger lymph volumes. Both ducts empty into the subclavian veins, completing the circuit and restoring fluid, proteins and immune cells to circulation.
Lymph Nodes
Lymph nodes are encapsulated lymphoid organs designed to slow lymph flow and maximise interaction between lymph contents and immune cells. A dense connective tissue capsule surrounds each node and sends inward extensions, called trabeculae, that provide structural support and guide lymph movement through the node.
The cortex lies just beneath the capsule and contains lymphoid follicles rich in B lymphocytes. Many follicles develop germinal centres, where B cells proliferate, undergo class-switch recombination and differentiate into antibody-secreting plasma cells. Follicular dendritic cells within these centres present antigen in a form optimised for B cell activation rather than phagocytosis.
Deeper within the node lies the paracortex, which is dominated by T lymphocytes and dendritic cells. This region contains high endothelial venules, specialised blood vessels that allow circulating lymphocytes to exit the bloodstream and enter the node. The paracortex functions as the primary site of T cell activation, particularly in response to antigens delivered by dendritic cells.
The medulla forms the innermost region and consists of medullary cords interspersed with medullary sinuses. Plasma cells within the cords secrete antibodies into the lymph, while macrophages within the sinuses remove pathogens and debris. By the time lymph reaches this region, it has been extensively filtered and processed before exiting through efferent lymphatic vessels.
Other Lymphatic Tissues
The spleen differs structurally from lymph nodes because it filters blood rather than lymph. Histologically, it is divided into white pulp, which contains lymphocytes organised around central arteries, and red pulp, which removes aged or damaged red blood cells. This architecture allows simultaneous immune surveillance and blood filtration.
The thymus has a unique microscopic structure reflecting its role in T cell development rather than immune response. Its cortex is densely packed with immature T cell precursors, while the medulla contains more mature T cells and characteristic Hassall’s corpuscles. The thymus lacks lymphoid follicles because B cell activation does not occur there.
Mucosa-associated lymphoid tissues, including Peyer’s patches and tonsils, are structurally distinct in that they are not encapsulated. Instead, lymphoid follicles are embedded directly within mucosal tissues and covered by specialised epithelium adapted for antigen sampling. Germinal centres are present, but lymph flow is not the organising principle. Instead, these structures are positioned to sample material directly from the external environment.
Clinical Connections
Microscopic changes in lymphatic tissues are often reflected in clinical findings. Reactive lymphadenopathy commonly occurs during infection as germinal centres within lymph node follicles enlarge due to rapid B cell proliferation. This process produces the tender, mobile lymph nodes frequently noted during acute illness and represents active immune engagement rather than malignancy.
Certain diseases selectively affect specific regions of lymphoid tissue:
In HIV infection, the paracortex, which is rich in CD4⁺ T cells, undergoes progressive depletion
In bacterial or viral infections, germinal centre expansion dominates the cortex
In lymphomas, malignant lymphoid cells may preserve normal nodal architecture early or distort it as disease progresses
In metastatic cancer, lymph node structure may be partially replaced by tumour cells
Lymphatic vessel disruption has significant clinical consequences. Obstruction, fibrosis or surgical removal of lymphatic vessels or nodes can impair lymph flow and lead to lymphoedema, particularly following cancer surgery or radiotherapy. This fluid accumulation reflects failure of lymphatic capillaries and collecting vessels to return interstitial fluid to circulation rather than primary vascular dysfunction.
Histological understanding of lymphatic tissues is essential for accurate interpretation of lymph node biopsies and surgical specimens. Distinguishing reactive changes from malignant transformation relies on recognising normal architecture and identifying where that organisation has been altered.
Concept Check
What structural feature of lymphatic capillaries allows them to absorb large molecules?
Which region of the lymph node contains germinal centres, and what occurs within them?
What is the function of high endothelial venules (HEVs)?
How do medullary cords and sinuses contribute to lymph filtration?
Which major differences distinguish the thymus histologically from lymph nodes?