PEYER’S PATCHES: Specialised Gut-Associated Lymphoid Tissue for Intestinal Immune Surveillance
Peyer’s patches are specialised clusters of lymphoid tissue located in the mucosa and submucosa of the small intestine, primarily the ileum. They form a crucial component of gut-associated lymphoid tissue (GALT) and play a central role in monitoring intestinal contents, detecting pathogens and supporting mucosal immunity.
Because the gastrointestinal tract is constantly exposed to billions of microbes, Peyer’s patches act as strategic immune sampling stations that help the body respond to harmful organisms while maintaining tolerance to food and commensal bacteria.
What You Need to Know
Peyer’s patches are organised clusters of lymphoid tissue embedded within the wall of the small intestine, most prominently in the ileum. Unlike encapsulated lymph nodes, they sit directly beneath the intestinal epithelium, positioning them to sample material passing through the gut lumen. Their location allows continuous monitoring of dietary antigens, microbes and microbial products as part of normal digestion. This close anatomical relationship between the gut lumen and lymphoid tissue makes Peyer’s patches a key structural component of gut-associated lymphoid tissue.
A defining feature of Peyer’s patches is their specialised epithelial covering. This surface contains M cells (microfold cells, specialised epithelial cells designed for antigen transport) that move intact antigens from the intestinal lumen to immune cells below. Once delivered, antigens are processed by dendritic cells and presented to lymphocytes within the follicles. This arrangement allows rapid assessment of luminal contents without relying on tissue damage or barrier breach.
Within Peyer’s patches, several coordinated processes occur:
B cells undergo activation and differentiation within lymphoid follicles
Class switching favours IgA production (the antibody specialised for mucosal surfaces)
T cells provide regulation and signalling rather than strong inflammatory responses
Dendritic cells shape responses toward either activation or tolerance
The outcome of antigen sampling in Peyer’s patches is not always immune activation. In many cases, repeated exposure to food antigens or commensal bacteria leads to immune tolerance rather than response. This balance limits unnecessary inflammation in the gut, where constant immune activation would disrupt absorption and barrier integrity. Through this combination of structural positioning and controlled immune signalling, Peyer’s patches support both surveillance and restraint, allowing the intestinal immune environment to remain responsive without becoming harmful.
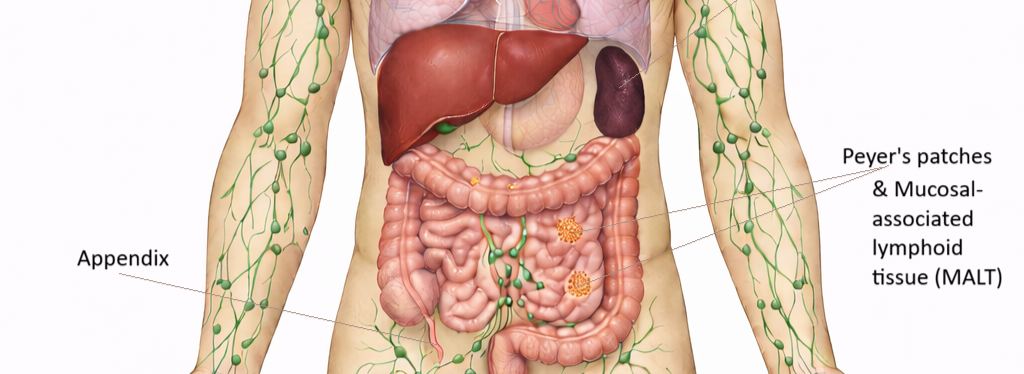
Image: Peyer’s patches are clusters of lymphoid tissue located in the ileum of the small intestine, concentrated along the distal intestinal wall. They form part of mucosa-associated lymphoid tissue (MALT), which is distributed throughout mucosal surfaces including the gastrointestinal, respiratory, and genitourinary tracts.
Beyond the Basics
Location and Structural Organisation
Peyer’s patches are located predominantly in the ileum, arranged along the antimesenteric border of the small intestine. This positioning places them opposite the mesenteric blood supply and allows broad exposure to intestinal contents. Structurally, they consist of multiple lymphoid follicles containing germinal centres, surrounded by interfollicular regions rich in T cells and dendritic cells. Above these immune cell clusters lies a specialised epithelial layer, beneath which sits the subepithelial dome, an area densely populated with antigen-presenting cells.
This close anatomical relationship between the intestinal lumen and lymphoid tissue allows Peyer’s patches to sample antigens efficiently without breaching the epithelial barrier. Rather than acting as a passive filter, they function as an active interface between the gut contents and the lymphatic system embedded within the intestinal wall.
M Cells and Controlled Antigen Transport
A defining feature of Peyer’s patches is the presence of M cells, or microfold cells, within the overlying epithelium. Unlike typical absorptive enterocytes, M cells have a thin glycocalyx and reduced microvilli, structural adaptations that facilitate antigen capture rather than nutrient absorption. They internalise luminal material and transport it intact across the epithelial layer through transcytosis, delivering antigens directly to immune cells in the subepithelial dome.
This mechanism allows immune surveillance to occur without relying on epithelial damage or inflammation. It is particularly important for early recognition of enteric pathogens such as Salmonella, norovirus and rotavirus, which are encountered within the gut lumen rather than the bloodstream.
B Cell Activation and IgA Specialisation
Within the lymphoid follicles of Peyer’s patches, B cells undergo activation and maturation in response to sampled antigens. This includes clonal expansion, somatic hypermutation, and class-switch recombination, with a strong bias toward IgA production. IgA is uniquely suited to mucosal environments because it neutralises microbes and toxins without activating complement or provoking tissue-damaging inflammation.
Once differentiated, IgA-producing plasma cells leave Peyer’s patches and migrate via lymphatic and blood vessels to the lamina propria throughout the intestinal tract. From there, IgA is transported across the epithelium into the gut lumen as secretory IgA, where it contributes to barrier protection and microbial containment rather than microbial elimination.
T Cell Regulation and Oral Tolerance
T cells within Peyer’s patches play a predominantly regulatory and coordinating role rather than driving aggressive immune responses. They assist B cell class switching, respond to antigen presentation by dendritic cells, and support the development of regulatory T cells. This environment favours controlled immune signalling over inflammation, an essential adaptation in a tissue exposed continuously to food antigens and commensal microbes.
Repeated exposure to non-harmful antigens often leads to oral tolerance, a state in which immune responses are actively suppressed rather than activated. Failure of this regulatory balance can contribute to pathological conditions such as food allergy, coeliac disease, or inflammatory bowel disease, where immune activation occurs inappropriately within the gut.
Interaction With the Intestinal Microbiome
Peyer’s patches exist in constant dialogue with the intestinal microbiome. Commensal bacteria influence follicle development, IgA production, and dendritic cell behaviour, helping calibrate immune responsiveness. Rather than triggering inflammation, normal microbial exposure promotes immune maturity and stability within gut-associated lymphoid tissue.
Disruption of microbial composition, whether through antibiotics, infection, or chronic illness, can alter Peyer’s patch activity and skew immune responses toward inflammation or impaired tolerance. This highlights the role of Peyer’s patches not only as immune sensors, but as adaptive lymphoid structures shaped by long-term environmental exposure within the gut.
Clinical Connections
Peyer’s patches are clinically important because their specialised antigen-sampling function can be exploited by certain pathogens. Some intestinal organisms, including Salmonella species and prions, gain access to deeper tissues by entering through M cells rather than crossing the epithelial barrier directly. This route allows pathogens to bypass surface defences and interact with immune cells within gut-associated lymphoid tissue, contributing to systemic spread or persistent infection.
During gastrointestinal infection or inflammation, Peyer’s patches may enlarge or become hyperactive as lymphocytes proliferate in response to antigen exposure. This can occur in conditions such as mesenteric adenitis or inflammatory bowel disorders, where immune activation is localised to the intestine rather than generalised throughout the lymphatic system. Enlargement of intestinal lymphoid tissue may be detected on imaging or inferred from associated symptoms such as abdominal pain and lymph node tenderness.
Several disease processes are linked to altered Peyer’s patch activity:
Entry of invasive pathogens through M cell transport pathways
Local lymphoid hyperplasia during enteric infection or inflammation
Dysregulated immune signalling in chronic gastrointestinal disease
Abnormal tolerance responses contributing to autoimmune pathology
Abnormal function of gut-associated lymphoid tissue, including Peyer’s patches, is thought to contribute to conditions such as Crohn’s disease, where immune responses to intestinal contents become excessive and poorly controlled. Structural and functional changes in these lymphoid aggregates may influence disease severity and progression.
Peyer’s patches are also central to the rationale behind oral and intranasal vaccine strategies. Vaccines delivered via mucosal routes target M cell uptake and local lymphoid activation, promoting IgA production and mucosal immune memory. Understanding the behaviour of Peyer’s patches helps explain how immunity can be generated at mucosal surfaces without relying on systemic inflammation, an approach increasingly used in modern vaccine design.
Concept Check
Where are Peyer’s patches located, and why is this location important?
What is the role of M cells in immune surveillance?
How do Peyer’s patches contribute to IgA production?
Why is oral tolerance important for gastrointestinal health?
How might Peyer’s patches be involved in the pathogenesis of intestinal infections?