THE SPLEEN: STRUCTURE & FUNCTION
The spleen is the largest lymphatic organ and performs vital roles in immune surveillance, blood filtration and the removal of aged or damaged red blood cells. Unlike lymph nodes, which filter lymph, the spleen filters blood directly, making it essential for detecting blood-borne pathogens. Positioned in the upper left abdominal quadrant, the spleen contains specialised tissues that allow it to simultaneously carry out immune functions and maintain healthy blood composition. Its dual role makes it indispensable in both haematological and immunological physiology.
What You Need to Know
The spleen is the largest secondary lymphoid organ and plays a unique role in immune surveillance of the bloodstream. Unlike lymph nodes, which filter lymph, the spleen continuously monitors blood for pathogens, abnormal cells, and cellular debris. Its structure allows immune cells to rapidly detect blood-borne antigens and initiate appropriate responses while also maintaining red blood cell integrity.
Structurally, the spleen is divided into two distinct but interconnected regions:
White pulp, which contains organised clusters of lymphocytes positioned around central arteries to detect antigens circulating in blood
Red pulp, which filters blood, removes senescent or damaged red blood cells and platelets, and clears cellular debris through macrophage activity
White pulp supports adaptive immune responses by enabling interaction between antigen-presenting cells, T cells, and B cells. This arrangement allows rapid activation of lymphocytes when blood-borne pathogens are detected. Red pulp macrophages perform continuous quality control of circulating blood cells, ensuring damaged or aged cells are removed efficiently.
In addition to immune defence, the spleen contributes to haematological regulation. It acts as a temporary reservoir for blood components and can release stored red blood cells and platelets during acute physiological stress, such as haemorrhage. Through these combined immune and filtration functions, the spleen plays a central role in maintaining systemic immune readiness and blood homeostasis.
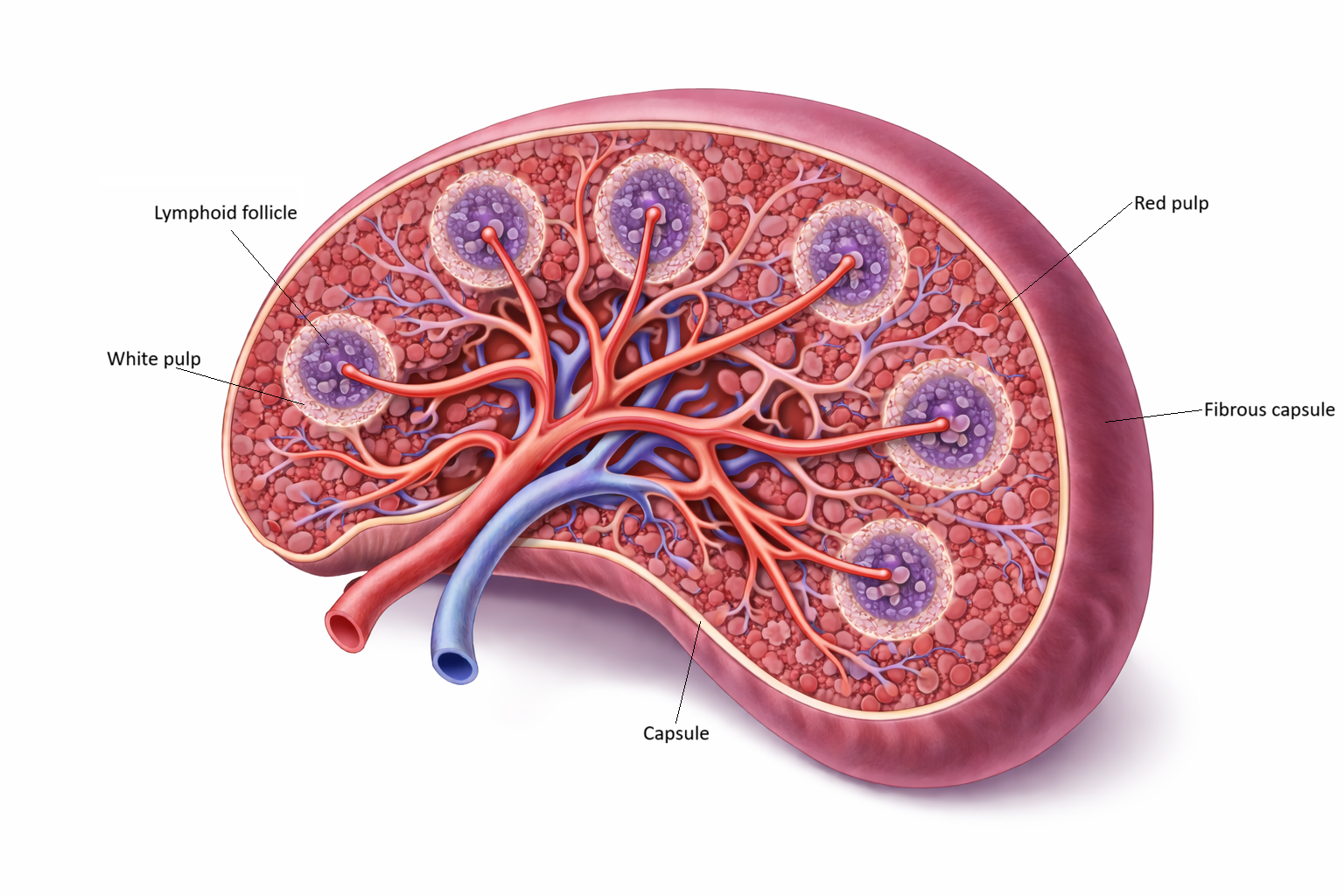
Image: The spleen is enclosed by a fibrous capsule and organised into two main regions: white pulp and red pulp. White pulp consists of lymphoid tissue arranged around central arterioles, where immune cells monitor the blood for pathogens. Surrounding this, red pulp contains vascular channels and splenic cords that filter blood, remove aged or damaged red blood cells, and recycle cellular components.
Beyond the Basics
Anatomical Organisation of the Spleen
The spleen is enclosed by a fibromuscular capsule that provides structural support and allows limited contraction. Extensions of this capsule, known as trabeculae, divide the organ into functional compartments and carry blood vessels deep into splenic tissue. Blood enters through the splenic artery, branches into central arterioles, and then passes through specialised vascular channels before draining into venous sinuses and exiting via the splenic vein. This circulation pattern slows blood flow, maximising contact between blood components and immune cells.
White Pulp
White pulp is the immunologically active region of the spleen and is organised around central arteries. Its structure resembles lymphoid follicles found in lymph nodes, but is adapted for monitoring blood rather than lymph.
Key components of white pulp include:
Periarteriolar lymphoid sheaths (PALS), which are rich in T lymphocytes positioned around central arterioles
Lymphoid follicles, which contain predominantly B lymphocytes and may develop germinal centres during active antibody responses
As blood flows through these regions, it enters the white pulp via central arterioles, where it is slowed and exposed to dense populations of lymphocytes. Antigens carried in the blood are sampled by antigen-presenting cells, allowing close interaction with both T cells in the periarteriolar lymphoid sheath and B cells within adjacent follicles. This organised arrangement increases the likelihood that rare antigen-specific lymphocytes will encounter their target, enabling rapid activation of adaptive immune responses to blood-borne pathogens.
Red Pulp
Red pulp makes up the majority of splenic tissue and is specialised for blood filtration and cell turnover. It consists of splenic cords, also called cords of Billroth, which contain macrophages, plasma cells, and reticular connective tissue, alongside venous sinuses that act as filtration channels.
Blood entering the red pulp must pass through narrow slits in the venous sinuses. Healthy red blood cells deform and pass through, while aged or damaged cells become trapped. Macrophages then phagocytose these cells, removing them from circulation and preventing accumulation of dysfunctional erythrocytes.
Filtration of Blood-Borne Pathogens
Because the spleen filters blood directly, it plays a critical role in systemic immune defence. Macrophages and dendritic cells capture circulating microbes and present antigens to lymphocytes in the white pulp. This localised immune activation supports rapid antibody production and cellular responses before pathogens disseminate further through the bloodstream.
This function is particularly important for organisms that circulate freely in blood, including encapsulated bacteria that resist direct phagocytosis without antibody or complement assistance.
Removal of Aged Red Blood Cells
As red blood cells age, their membranes lose flexibility and structural integrity. Passage through the narrow sinusoidal spaces of the red pulp acts as a mechanical quality-control process. Cells that fail to deform appropriately are retained and broken down by macrophages, which recognise these less flexible cells as abnormal, engulf them, and digest them internally, allowing haemoglobin to be broken down so that iron can be recovered and reused while the remaining components are safely removed.
Within these macrophages, haemoglobin is degraded and iron is recovered for reuse in erythropoiesis. This recycling process supports efficient red blood cell production and prevents iron loss. Cells that fail to deform appropriately are retained and broken down by macrophages, which recognise and engulf these damaged cells through phagocytosis, degrade them within lysosomes, and break down haemoglobin into its components so that iron can be recovered for reuse while the remaining components are processed and removed
Blood Reservoir and Platelet Storage
The spleen stores a substantial proportion of the body’s platelets and a smaller volume of red blood cells. Smooth muscle fibres within the splenic capsule allow contraction during acute physiological stress, such as haemorrhage, releasing stored blood components into circulation. This mechanism provides short-term support for haemodynamic stability while definitive control of blood loss occurs.
Immune Support and Antibody Production
The spleen is a major site of antibody production against blood-borne antigens. It is especially important for generating IgM and IgG responses to encapsulated bacteria such as Streptococcus pneumoniae and Neisseria meningitidis. These organisms are difficult to clear because their outer capsule masks surface structures that immune cells would normally recognise. Effective clearance relies on opsonisation, a process in which antibodies (such as IgG) or complement proteins bind to the surface of a pathogen, coating it and making it more visible to phagocytic cells. This coating acts like a signal that allows macrophages and neutrophils to recognise, attach to, and more efficiently engulf the pathogen.
Loss of splenic function significantly impairs this aspect of immunity, increasing susceptibility to overwhelming infection. The spleen therefore serves as both a filter and an immune amplifier for systemic defence.
Clinical Connections
Loss of splenic function has serious clinical consequences because the spleen plays a central role in filtering blood and mounting immune responses to circulating pathogens. Individuals with asplenia or functional hyposplenism are particularly vulnerable to overwhelming infection, especially from encapsulated bacteria that rely on splenic macrophages and antibody-mediated opsonisation for clearance. Even minor infections in these patients can progress rapidly and require urgent assessment.
Key clinical risks associated with impaired splenic function include:
Severe infection with encapsulated organisms such as Streptococcus pneumoniae and Neisseria meningitidis
Rapid progression from mild illness to sepsis
Reduced antibody responses to blood-borne pathogens
Increased reliance on vaccination and early antimicrobial treatment
Patients without a spleen typically require lifelong vaccination strategies, including additional pneumococcal, meningococcal, and Haemophilus influenzae type b vaccines. Education around early presentation for fever or signs of infection is critical, as prompt treatment significantly reduces morbidity and mortality.
Splenomegaly occurs when splenic workload increases or blood flow is altered. It is commonly associated with systemic infection, chronic liver disease with portal hypertension, haematological disorders such as haemolytic anaemia, and malignancies including lymphoma and leukaemia. An enlarged spleen may become hyperactive, leading to excessive removal of blood cells and resulting cytopenias.
Trauma to the spleen is a major clinical concern due to its high vascularity. Blunt abdominal trauma can cause splenic rupture and life-threatening internal bleeding. Careful monitoring, imaging, and sometimes surgical intervention are required, with splenic preservation preferred where possible to maintain long-term immune protection.
Concept Check
What are the two main regions of the spleen, and what does each region do?
How does the spleen filter blood differently from lymph nodes?
Why are individuals without a spleen more susceptible to encapsulated bacteria?
How does the spleen remove aged or damaged red blood cells?
What is the function of periarteriolar lymphoid sheaths (PALS)?