THE THYMUS: Primary Site of T Cell Development, Selection and Immune Maturation
The thymus is a primary lymphoid organ essential for the development and maturation of T lymphocytes. Located in the anterior mediastinum, the thymus provides a highly specialised microenvironment where immature lymphocytes differentiate into functional T cells capable of recognising foreign antigens while avoiding harmful reactions against the body’s own tissues. Although the thymus is largest and most active in childhood, its influence on immune competency persists throughout life, as the T cells it produces seed lymphoid tissues and maintain long-term immune surveillance.
What You Need to Know
The thymus is a primary lymphoid organ responsible for the development, selection, and maturation of T lymphocytes. Immature T cell precursors originate in the bone marrow and migrate to the thymus, where they undergo a highly regulated education process that determines their survival and function. This process is essential for producing a functional T cell population capable of mounting immune responses without attacking self-tissues.
Within the thymus, developing thymocytes pass through two anatomically and functionally distinct regions:
The cortex, where thymocytes are tested for their ability to recognise antigens presented on self–MHC molecules
The medulla, where thymocytes are screened for excessive reactivity to self-antigens
Specialised thymic epithelial cells, dendritic cells, and macrophages present self-antigens and provide survival or death signals during these stages. Thymocytes that cannot recognise MHC molecules or that bind too strongly to self-antigens are eliminated through apoptosis. As a result, only a small fraction of thymocytes successfully complete development.
T cells that pass selection emerge as mature, naïve CD4⁺ or CD8⁺ T cells and exit the thymus to enter the circulation. They populate secondary lymphoid organs such as lymph nodes, the spleen, and mucosal lymphoid tissues, where they remain until they encounter their specific antigen. Through this stringent selection process, the thymus ensures immune competence while maintaining self-tolerance, a balance that is critical for effective and safe adaptive immunity.
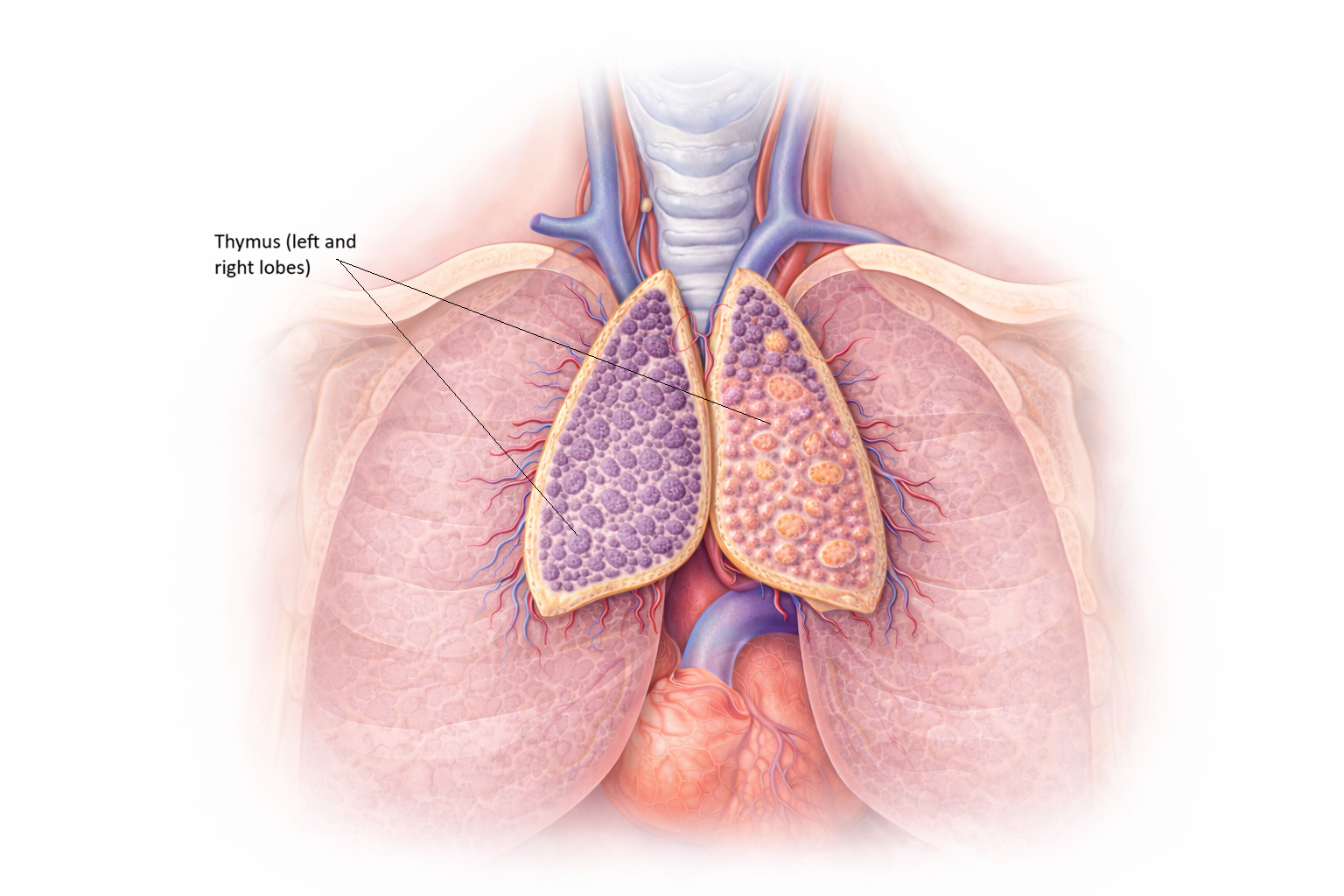
Image: The thymus sits in the anterior mediastinum, just behind the sternum and above the heart, between the lungs. It is a soft, bilobed gland that appears pale pink to grey and gradually shrinks and becomes fatty with age.
Beyond the Basics
Anatomy and Structure of the Thymus
The thymus is a bilobed, encapsulated organ located in the anterior mediastinum. A connective tissue capsule extends inward as septa, dividing the thymus into multiple lobules. Each lobule contains two functionally distinct regions, the cortex and the medulla, which together guide T cell development and selection.
The cortex forms the outer portion of each lobule and is densely packed with immature thymocytes. Cortical thymic epithelial cells provide survival signals and present self–MHC molecules to developing T cells. This environment supports early T cell differentiation and is the site where thymocytes are tested for their ability to recognise MHC molecules, a process known as positive selection.
The medulla forms the inner region of the lobule and contains fewer lymphocytes. It is enriched with medullary thymic epithelial cells, dendritic cells, and macrophages. Medullary epithelial cells express a broad array of tissue-specific self-antigens through the action of the autoimmune regulator gene. This allows developing T cells to be exposed to antigens normally found in peripheral organs, supporting elimination of self-reactive cells during negative selection.
Within the medulla are Hassall’s corpuscles, structures composed of concentrically arranged epithelial cells. Although once thought to be inert, these structures play an active role in shaping immune tolerance by influencing dendritic cell behaviour and supporting the development of regulatory T cells.
T Cell Development: From Thymocyte to Mature T Cell
T cell maturation within the thymus follows a tightly regulated sequence that progressively refines the T cell population.
Entry of Immature Thymocytes
Bone marrow–derived T cell precursors enter the thymus lacking CD4, CD8, and functional T cell receptors. These early cells are referred to as double-negative thymocytes. At this stage, they are committed to the T cell lineage but are not yet capable of antigen recognition.
T Cell Receptor Gene Rearrangement
Thymocytes undergo rearrangement of genes encoding the T cell receptor. This process generates immense receptor diversity, allowing the immune system to recognise a wide range of antigens. Only thymocytes that successfully form a functional T cell receptor progress further in development.
Positive Selection
Thymocytes that express both CD4 and CD8, known as double-positive cells, interact with self–MHC molecules displayed by cortical epithelial cells. Cells that fail to bind MHC undergo apoptosis. This step ensures that surviving T cells are capable of recognising antigens only when presented on MHC molecules. Interaction with MHC class II directs cells toward the CD4⁺ helper T cell lineage, while interaction with MHC class I commits cells to the CD8⁺ cytotoxic T cell lineage.
Negative Selection
Following positive selection, thymocytes migrate to the medulla, where they are exposed to a wide range of self-antigens. Cells that bind too strongly to these antigens receive death signals and are eliminated. This process removes potentially harmful self-reactive T cells and establishes central tolerance.
Export of Mature T Cells
Only a small proportion of thymocytes, approximately 1–3%, survive both selection processes. These cells exit the thymus as mature, naïve CD4⁺ or CD8⁺ T cells and enter the circulation. They then populate secondary lymphoid organs, where they remain until activated by their specific antigen.
Thymic Involution
The thymus is most active during childhood, when the immune system is establishing a diverse T cell repertoire. After puberty, thymic tissue gradually decreases in size and cellularity and is progressively replaced by adipose tissue, a process known as thymic involution.
Despite this structural change, immune competence is maintained in adulthood through long-lived naïve and memory T cell populations established earlier in life. Peripheral T cell proliferation and survival mechanisms compensate for reduced thymic output, allowing continued immune responsiveness even as thymic activity declines.
Clinical Connections
Normal thymic function is essential for establishing a competent and self-tolerant T cell population. When thymic development is disrupted, immune consequences are often severe and wide-ranging. Congenital absence or hypoplasia of the thymus, such as in DiGeorge syndrome, leads to marked T cell deficiency. Affected individuals have impaired cell-mediated immunity and are highly susceptible to viral, fungal, and opportunistic infections early in life.
Several clinical conditions are directly linked to thymic dysfunction:
Severe immunodeficiency due to reduced or absent T cell maturation
Increased infection risk following impaired thymic output
Autoimmune disease when negative selection fails
Altered immune recovery after chemotherapy or bone marrow transplantation
Failure of effective negative selection allows self-reactive T cells to escape into circulation, increasing the risk of autoimmune disease. These aberrant immune responses may target specific tissues and contribute to chronic inflammatory conditions that require long-term immunomodulatory therapy.
Thymomas, tumours arising from thymic epithelial cells, are frequently associated with autoimmune and neuromuscular disorders. Myasthenia gravis is the most well-known association, where abnormal immune regulation leads to autoantibody production against acetylcholine receptors at the neuromuscular junction. Removal of the thymoma or thymectomy can improve disease control in many patients.
Understanding thymic function is also important in transplant medicine and oncology. Recovery of T cell populations after chemotherapy or haematopoietic stem cell transplantation depends partly on residual thymic activity. Assessment of thymic output and immune reconstitution helps guide infection prophylaxis, vaccination timing, and long-term immune monitoring.
Concept Check
What are the two main regions of the thymus, and what occurs in each?
How does positive selection ensure proper T cell function?
Why is negative selection important for preventing autoimmunity?
What is the role of AIRE in thymic medullary epithelial cells?
Why does thymic involution not result in complete immune failure in adulthood?