Compartment Syndrome
Compartment syndrome is a limb-threatening condition caused by increased pressure within a closed osteofascial compartment, resulting in impaired tissue perfusion and ischaemia. Because skeletal muscle and nerves are enclosed by relatively non-compliant fascia, any increase in compartment volume can rapidly compromise circulation and cellular viability. Compartment syndrome can develop suddenly after injury, pain is often disproportionate to clinical findings, and delayed recognition can lead to irreversible tissue damage. The condition therefor requires urgent escalation and treatment.
What You Need to Know
Compartment syndrome occurs when rising pressure within a closed muscle compartment compromises tissue perfusion. Skeletal muscle groups are enclosed by dense, non-compliant fascia that allows little outward expansion. When bleeding, oedema, or reperfusion swelling increases the volume within this fixed space, intracompartmental pressure rises. As pressure approaches capillary perfusion pressure, blood flow to muscles and nerves falls, limiting oxygen delivery and removal of metabolic waste.
Reduced perfusion rapidly disrupts cellular metabolism. Muscle and nerve cells switch to anaerobic energy production, ATP levels fall, and intracellular ion balance is lost. This process causes increasing pain and sensory disturbance before irreversible damage occurs. Because large arteries may remain patent, distal pulses can be present despite severe microvascular compromise, and early imaging may appear unremarkable.
Several features define compartment syndrome as a time-critical physiological process:
Rising intracompartmental pressure that impairs capillary blood flow rather than large vessel occlusion
Progressive tissue hypoxia leading to muscle necrosis and nerve injury if pressure is not relieved
Clinical deterioration that can occur despite normal pulses and initially reassuring investigations
Without timely decompression (fasciotomy), prolonged ischaemia results in irreversible muscle death, permanent nerve damage, and long-term functional loss. The severity of outcome depends on both the magnitude and duration of pressure elevation, making early recognition and intervention central to preventing permanent disability.
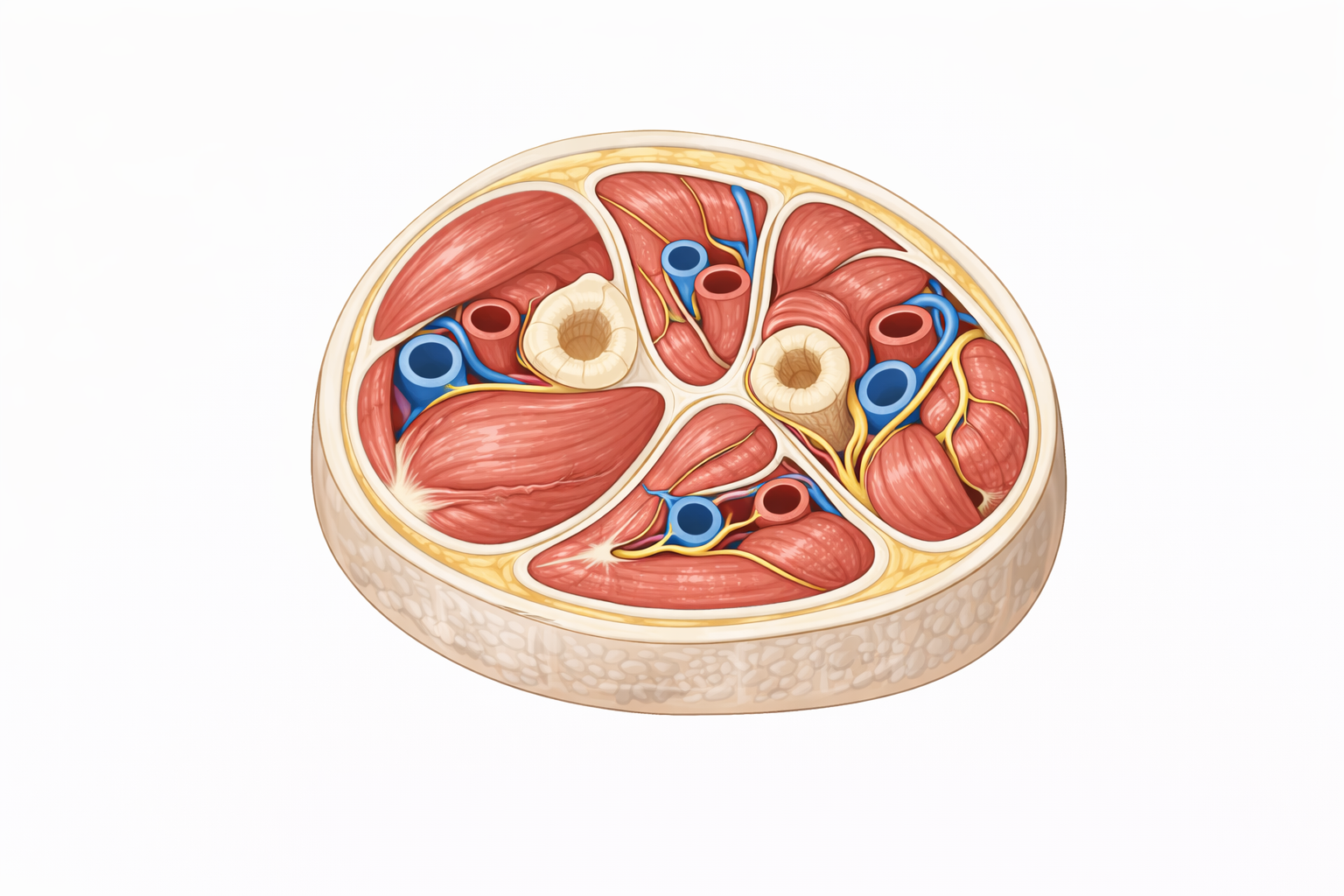
Image: Muscles of the limb are enclosed within rigid fascial compartments that limit expansion. When swelling or bleeding occurs within a compartment, pressure rises, compressing blood vessels and reducing tissue perfusion. This restricted space is what underlies compartment syndrome, where increasing pressure can lead to ischaemia and tissue damage if not relieved.
Beyond the Basics
Pressure–perfusion mismatch
Tissue viability depends on continuous capillary blood flow that delivers oxygen and nutrients while removing metabolic waste. In compartment syndrome, rising intracompartmental pressure compresses venous outflow first. As venous pressure increases, the pressure gradient between arterial inflow and venous drainage narrows, reducing effective perfusion even before arteries are directly compressed.
As pressure continues to rise, arterial inflow becomes progressively compromised. Capillaries collapse under external pressure, preventing oxygen and glucose from reaching cells and blocking removal of carbon dioxide and metabolic by-products. This mismatch between tissue metabolic demand and available perfusion is the central mechanism driving cellular injury in compartment syndrome, rather than large-vessel occlusion.
Ischaemia, oedema, and the vicious cycle
Ischaemia initiates cellular injury by disrupting membrane integrity and endothelial function. Damaged capillaries become increasingly permeable, allowing plasma to leak into the interstitial space. This fluid accumulation increases compartment volume, further elevating intracompartmental pressure.
A self-amplifying cycle develops in which ischaemia promotes oedema, oedema increases pressure, and rising pressure worsens ischaemia. Once this cycle is established, tissue injury can progress rapidly even in the absence of ongoing bleeding or external compression. At this stage, spontaneous resolution is unlikely without active pressure relief.
Muscle vulnerability and necrosis
Skeletal muscle has a high metabolic demand and limited tolerance for reduced oxygen delivery. Within hours of sustained ischaemia, ATP depletion impairs ion pump function, allowing sodium and calcium to accumulate inside muscle fibres. Elevated intracellular calcium activates proteolytic enzymes that degrade structural proteins and cell membranes.
As injury progresses, muscle fibres swell and undergo necrosis. Necrotic muscle releases myoglobin and potassium into the circulation, creating a direct link between compartment syndrome, rhabdomyolysis and acute kidney injury when large muscle volumes are involved or treatment is delayed.
Nerve ischaemia and sensory change
Peripheral nerves are particularly sensitive to pressure and hypoxia. Early nerve ischaemia disrupts impulse conduction, producing paraesthesia, burning pain and altered sensation. These changes often precede overt motor weakness and serve as early indicators of neural compromise.
With prolonged compression, nerves develop demyelination and axonal injury. Unlike muscle tissue, neural tissue has limited regenerative capacity. Persistent sensory loss or motor deficit indicates irreversible damage rather than delayed recovery, even after compartment pressure is relieved.
Causes and triggers
Compartment syndrome most commonly follows fractures, particularly of the tibia and forearm, due to bleeding and post-injury swelling within confined compartments. It may also occur after crush injury, reperfusion following prolonged ischaemia, burns, overly tight casts or dressings, and prolonged external compression such as immobilisation or entrapment.
Importantly, compartment syndrome can develop without fracture and may arise after apparently successful reduction or immobilisation. Ongoing swelling and reperfusion-related oedema mean risk persists beyond the initial injury period.
Acute vs chronic compartment syndrome
Acute compartment syndrome is a time-critical surgical emergency in which sustained pressure elevation leads to progressive tissue necrosis unless promptly relieved. Symptoms worsen over time and do not resolve with rest or analgesia.
Chronic exertional compartment syndrome involves transient increases in compartment pressure during activity. Symptoms develop predictably with exercise and resolve when activity ceases, as perfusion is restored before irreversible injury occurs. While both conditions involve pressure-related perfusion impairment, differentiation relies on symptom persistence and progression rather than pressure measurements alone.
Clinical Connections
Compartment syndrome often presents with severe pain that is disproportionate to the apparent injury and poorly relieved by analgesia. Pain is typically exacerbated by passive stretch of the involved muscle group, as stretching further increases intracompartmental pressure and worsens perfusion. The affected compartment may feel tense or firm on palpation, and early sensory disturbance such as paraesthesia indicates evolving nerve ischaemia. Motor weakness may develop as pressure and hypoxia progress.
Key clinical features that support early recognition include:
Escalating pain out of proportion to injury and resistant to analgesia
Pain on passive stretch of muscles within the affected compartment
Tense swelling with early sensory change, even when distal pulses are present
Assessment relies on repeated clinical examination rather than a single observation. The limb should be inspected and palpated for increasing firmness, assessed for pain on passive movement, and monitored for evolving sensory or motor change over time. Compartment pressure measurement may be used when the diagnosis is uncertain or in patients unable to report pain, but a normal reading does not exclude early disease in a deteriorating limb.
Late findings such as pallor, paralysis and pulselessness indicate advanced injury and should not be awaited. Large-vessel flow may be preserved, allowing distal pulses to remain palpable despite severe microvascular compromise. Normal vascular observations therefore do not rule out compartment syndrome.
Definitive management requires urgent surgical fasciotomy to decompress the compartment and restore perfusion. Delay allows ongoing ischaemia to progress to muscle necrosis, nerve injury and secondary infection, markedly increasing the risk of permanent disability or limb loss.
Several management principles follow directly from the underlying mechanisms:
Analgesia may be ineffective because pain is driven by ischaemia and nerve compression rather than inflammation alone
Limb elevation can reduce arterial inflow and worsen tissue hypoxia in an underperfused compartment
Time to decompression is the dominant determinant of outcome, as prolonged pressure leads to irreversible tissue injury
Concept Check
Why does increased compartment pressure impair tissue perfusion before arterial pulses are lost?
How does ischaemia contribute to worsening oedema in compartment syndrome?
Why are skeletal muscle and nerves particularly vulnerable to raised compartment pressure?
How does compartment syndrome link mechanistically to rhabdomyolysis?
Why is pain on passive stretch an early and important clinical sign?