Osteomalacia
Osteomalacia is a metabolic bone disorder characterised by impaired mineralisation of bone matrix, resulting in structurally weak and deformable bone. Unlike osteoporosis, which involves loss of bone mass and microarchitecture, osteomalacia reflects failure to adequately harden newly formed bone. Understanding the pathophysiology explains why bone pain and muscle weakness dominate the clinical picture, why fractures may occur despite preserved bone volume, and why biochemical abnormalities often precede radiographic change.
What You Need to Know
Osteomalacia develops when newly formed bone matrix fails to mineralise adequately, resulting in bone that is structurally weak despite preserved volume. Under normal conditions, osteoblasts produce an organic collagen scaffold that is subsequently hardened by deposition of calcium–phosphate crystals. In osteomalacia, this second step is impaired. Osteoid continues to be laid down, but insufficient mineral incorporation leaves bone flexible rather than rigid, reducing its ability to withstand normal mechanical load.
Several interrelated processes contribute to this failure of mineralisation:
Inadequate calcium and phosphate availability to mineralise osteoid
Impaired intestinal absorption of minerals, most commonly due to vitamin D deficiency
Secondary hormonal responses that prioritise serum calcium over skeletal strength
Vitamin D deficiency is the most frequent underlying cause and limits absorption of both calcium and phosphate from the gastrointestinal tract. As circulating calcium falls, parathyroid hormone secretion increases, stimulating bone resorption to stabilise serum levels. This compensatory response maintains extracellular calcium needed for neuromuscular and cellular function, but it further depletes skeletal mineral stores. Over time, the combination of unmineralised osteoid and increased resorption produces bone that appears intact in size but lacks mechanical strength, predisposing to pain, deformity and fracture under low levels of stress.
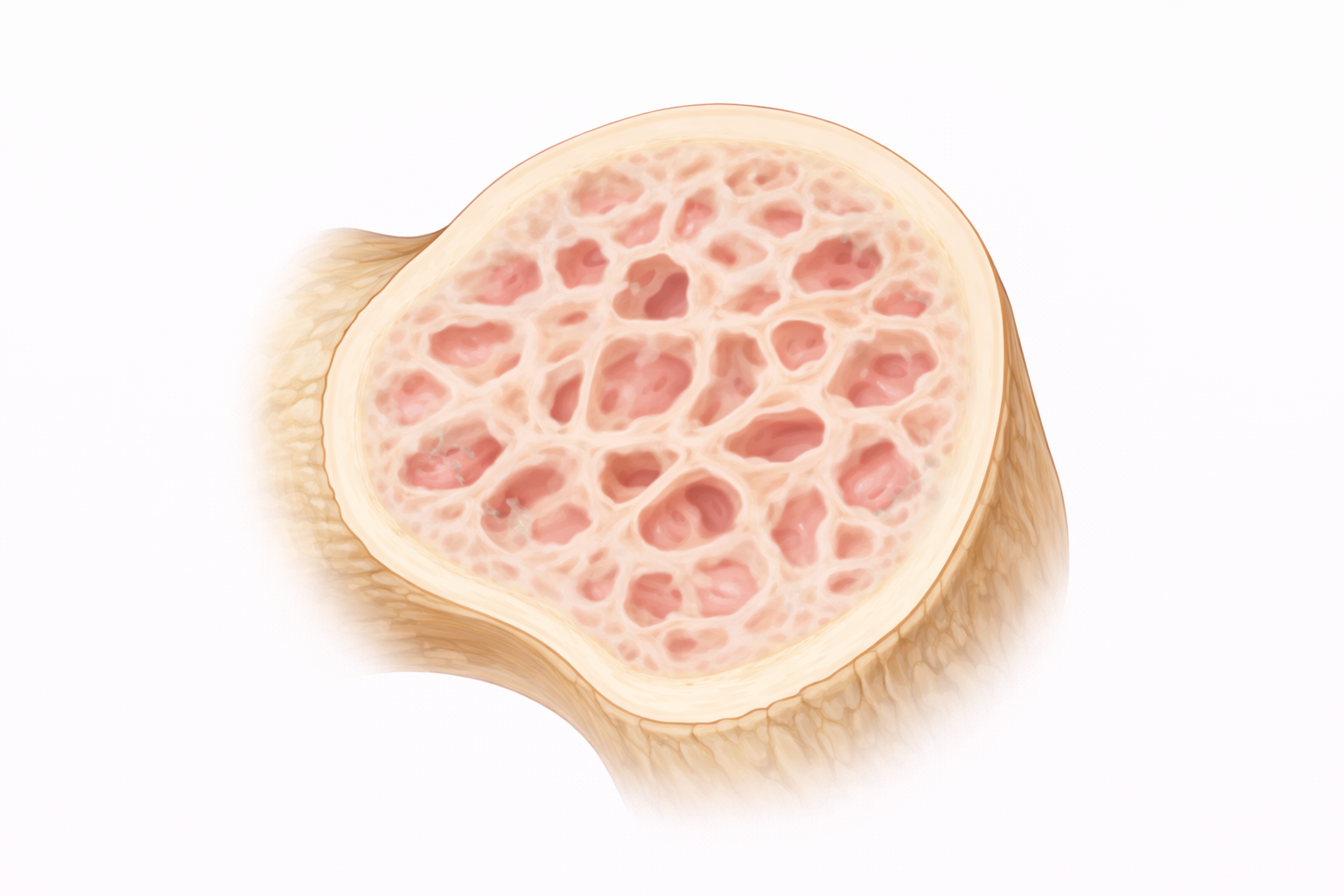
Image: Osteomalacia is characterised by impaired mineralisation of bone, resulting in a softer, weaker structure. The trabecular framework remains present but appears less dense and poorly defined, reflecting reduced calcium deposition and decreased mechanical strength.
Beyond the Basics
Failure of mineralisation rather than bone loss
In osteomalacia, the fundamental problem is impaired mineral deposition rather than reduced production of bone matrix. Osteoblasts continue to synthesise osteoid, the collagen-rich framework of bone, but inadequate availability of calcium–phosphate crystals prevents this matrix from hardening. As a result, osteoid accumulates and bone remains soft and pliable instead of rigid. This distinction explains why bone density measurements may appear only mildly reduced, as bone volume is preserved even though its mechanical strength is markedly compromised.
Because bone lacks adequate rigidity, it deforms under normal physiological load. Weight-bearing activities place stress on softened bone, leading to bone pain, local tenderness and gradual skeletal deformation. These effects are most evident in areas exposed to high load, including the pelvis, femur and vertebrae, where bending and compression forces are greatest.
Vitamin D, calcium, and phosphate interdependence
Effective mineralisation requires coordinated availability of calcium and phosphate, both of which depend heavily on vitamin D. Vitamin D promotes intestinal absorption of these minerals, maintaining the substrate needed for hydroxyapatite formation within the osteoid matrix. When vitamin D levels are low, absorption falls and mineral supply becomes insufficient to support normal mineralisation.
Phosphate deficiency is particularly significant because mineral deposition cannot proceed without it, even when calcium levels are relatively preserved. Disorders such as renal phosphate wasting, malabsorption syndromes and chronic kidney disease can disrupt phosphate balance independently of vitamin D intake. In these settings, osteomalacia develops through impaired mineral availability rather than failure of matrix production.
Secondary hyperparathyroidism and adaptive bone resorption
As mineral availability declines, parathyroid hormone secretion increases to maintain serum calcium within a narrow physiological range. PTH promotes bone resorption and enhances renal calcium reabsorption, stabilising extracellular calcium levels required for neuromuscular and cardiac function.
This adaptive response comes at a skeletal cost. Increased resorption further depletes already poorly mineralised bone, while ongoing osteoid production continues. The result is bone that is abundant in matrix but deficient in mineral, producing a structurally unsound skeleton. Progressive pain and fracture risk therefore develop despite active hormonal compensation.
Muscle weakness and altered biomechanics
Calcium and vitamin D are also essential for normal muscle contraction and neuromuscular signalling. Deficiency contributes to proximal muscle weakness, particularly affecting the hip and thigh muscles, leading to altered gait and reduced postural stability. These changes increase falls risk and expose softened bone to abnormal loading patterns.
The combination of reduced bone rigidity and impaired muscle function amplifies mechanical stress across the skeleton. Difficulty rising from a chair or climbing stairs often appears early, preceding overt fractures. This interaction between skeletal softness and muscle weakness explains why functional decline may be an early feature of osteomalacia, even before significant radiographic abnormalities are identified.
Clinical Connections
Osteomalacia commonly presents with diffuse, deep bone pain that is often poorly localised and worsens with weight-bearing. Pain arises because inadequately mineralised bone bends under normal load, stimulating periosteal nerves rather than from acute structural failure alone. Proximal muscle weakness, particularly affecting the hip and thigh muscles, contributes to fatigue, altered gait and difficulty rising from a chair or climbing stairs. These features may precede fracture and are frequently misattributed to general deconditioning or musculoskeletal strain.
Several clinical patterns point toward impaired mineralisation rather than primary bone loss:
Widespread bone tenderness with minimal trauma history
Proximal muscle weakness and gait disturbance preceding fracture
Stress fractures in weight-bearing bones, such as the femur, pelvis or tibia, without a clear injury
Diagnosis is supported by the clinical picture alongside biochemical abnormalities, typically low or low-normal calcium, reduced phosphate, elevated alkaline phosphatase and low vitamin D levels, with secondary hyperparathyroidism often present. Imaging may show stress fractures or Looser zones, areas of incomplete fracture caused by repeated bending of softened bone. Bone density testing may underestimate disease severity because bone volume can be preserved despite poor mineralisation.
Management targets restoration of mineral availability and correction of the underlying cause. Vitamin D replacement improves intestinal absorption of calcium and phosphate, while supplementation restores the substrate needed for proper mineral deposition within osteoid. Identification and treatment of contributing factors such as malabsorption, chronic kidney disease or renal phosphate wasting are essential, as bone cannot regain strength if mineral deficits persist. Improvement in bone pain and muscle strength typically follows correction of mineral imbalance, reducing fracture risk and preventing progression to deformity.
Concept Check
How does osteomalacia differ mechanistically from osteoporosis?
Why can bone volume be preserved despite reduced bone strength in osteomalacia?
How does vitamin D deficiency impair bone mineralisation?
Why does secondary hyperparathyroidism worsen skeletal demineralisation?
How does muscle weakness contribute to fracture risk in osteomalacia