Osteoporosis
Osteoporosis is a systemic skeletal disorder characterised by reduced bone strength and increased susceptibility to fracture. It develops when bone remodelling becomes imbalanced, leading to progressive loss of bone mass and deterioration of bone microarchitecture. Although often clinically silent until fracture occurs, osteoporosis reflects long-standing disruption of normal bone physiology. Fractures may occur with minimal trauma, women of 50 and adults over 70 are at higher risk.
What You Need to Know
Osteoporosis develops when the normal balance between bone resorption and bone formation is persistently disrupted. In healthy bone, osteoclasts remove old or microdamaged bone and osteoblasts replace it with new bone, maintaining strength and structural integrity over time. In osteoporosis, this coordinated process becomes uncoupled. Bone breakdown continues, but replacement is incomplete, leading to gradual loss of bone mass and deterioration of internal structure across repeated remodelling cycles.
This imbalance arises from interacting biological changes rather than a single defect. Osteoblast activity declines, osteoclasts remain active for longer, and hormonal regulation that normally restrains bone loss becomes less effective. As remodelling continues under these conditions, trabecular bone becomes thinner and loses connectivity, while cortical bone becomes progressively thinner and more porous. These changes reduce the skeleton’s ability to absorb and distribute load, making bone more susceptible to fracture during everyday activities.
Several structural features explain why fracture risk increases even before bone density appears markedly reduced:
Thinning and loss of trabecular connections reduce internal support within bone
Cortical thinning and porosity weaken resistance to bending and impact
Repeated incomplete repair allows microdamage to accumulate over time
As a result, bone strength declines disproportionately to changes seen on bone density measurement alone. Osteoporosis is therefore a disorder of bone quality as well as quantity, with microarchitectural failure playing a central role in fragility and fracture risk even when overall bone size appears preserved.
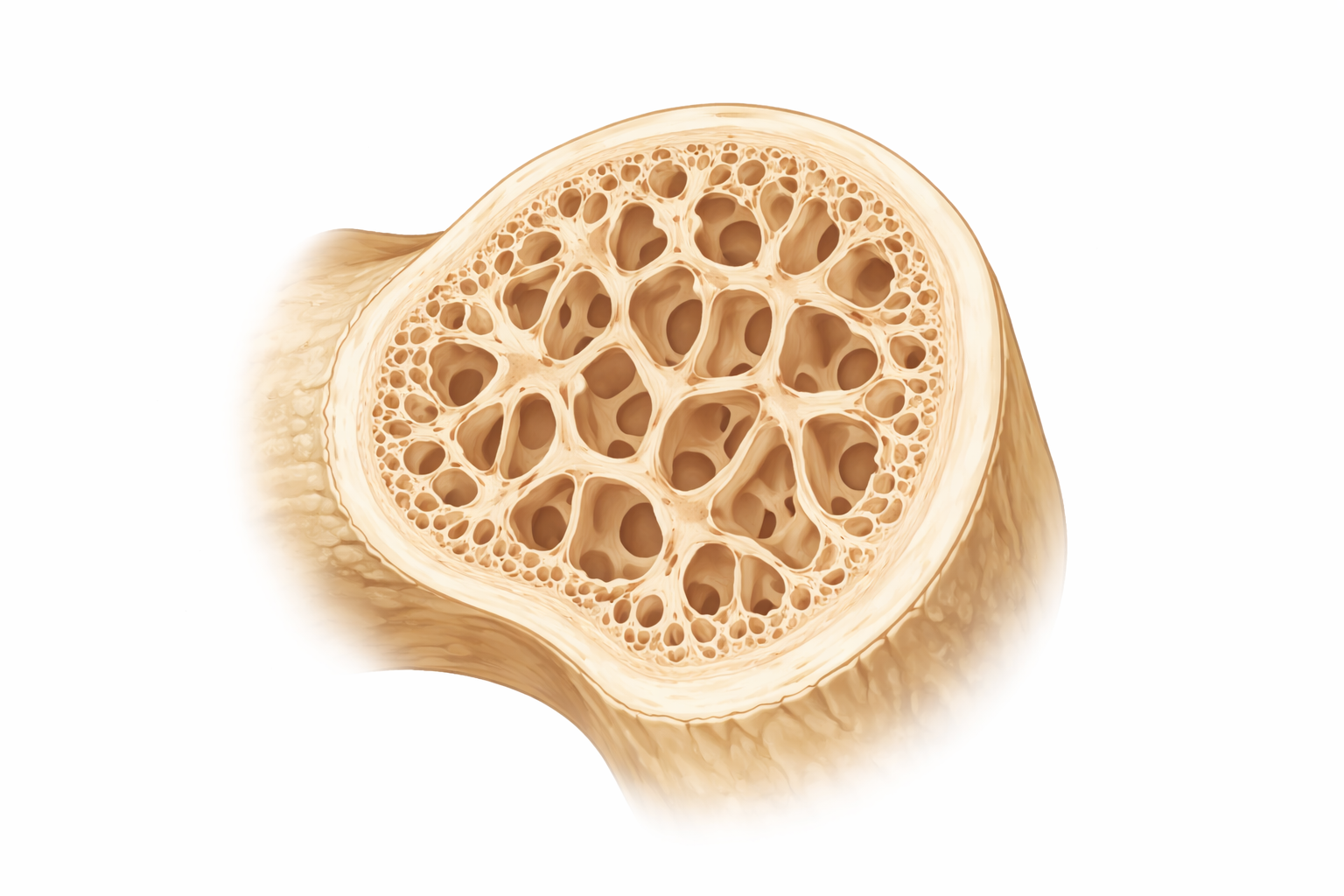
Image: Osteoporotic bone shows thinning and loss of trabeculae (the internal supporting structure), resulting in larger spaces and reduced overall bone density. This weakened architecture makes bone more fragile and increases the risk of fracture, even with minimal trauma.
Beyond the Basics
Failure of remodelling balance
Bone remodelling occurs within small, localised units that continuously remove microdamaged bone and replace it with new tissue adapted to mechanical demand. In osteoporosis, this tightly coordinated process becomes uncoupled. Osteoclast activity is preserved or increased, while osteoblast recruitment, survival and bone-forming capacity decline. As a result, resorption cavities are incompletely refilled, allowing microdamage to persist and accumulate across successive remodelling cycles.
Loss of trabecular connectivity is a key consequence of this imbalance. Trabecular bone acts as an internal scaffold that distributes load and absorbs force, particularly within the vertebral bodies and proximal femur. As trabeculae thin and disconnect, stress is concentrated across fewer structural elements. Bone strength falls rapidly, even when overall bone size changes little, increasing susceptibility to vertebral compression fractures and hip fractures.
Hormonal influence and accelerated bone loss
Sex hormones play a central role in regulating bone turnover. Oestrogen suppresses osteoclast formation and activity and promotes osteoclast apoptosis, limiting the duration of bone resorption. When oestrogen levels fall, such as after menopause, this restraint is removed. Osteoclast numbers increase, resorption deepens, and bone loss accelerates, particularly within trabecular-rich regions.
Excess cortisol further shifts remodelling toward bone loss. Glucocorticoids suppress osteoblast differentiation, reduce collagen synthesis and prolong osteoclast survival. These effects explain the increased fracture risk associated with ageing, menopause and long-term glucocorticoid exposure, even in the absence of other metabolic disease.
Mechanical loading and disuse
Bone formation is strongly influenced by mechanical loading generated through weight-bearing and muscle contraction. Mechanical strain stimulates osteoblast activity and directs bone deposition along lines of stress, strengthening the skeleton where it is most needed. When physical activity is reduced, this stimulus is lost, and resorption predominates.
Prolonged immobility, hospitalisation or neuromuscular disease therefore leads to rapid bone loss. Loss of muscle mass compounds this effect by reducing mechanical load and impairing balance and protective responses. Fracture risk rises through a combination of weaker bone and increased likelihood of falls.
Calcium economy and secondary adaptation
Calcium balance is tightly regulated to maintain normal neuromuscular and cellular function. When dietary calcium intake or vitamin D availability is insufficient, parathyroid hormone secretion increases. This response mobilises calcium from bone to stabilise serum levels, prioritising short-term physiological needs over skeletal preservation.
Chronic elevation of parathyroid hormone leads to sustained bone resorption, gradually depleting skeletal mineral stores. Importantly, serum calcium levels often remain within reference ranges despite substantial bone loss. This masking effect allows osteoporosis to progress silently until structural failure occurs, emphasising that bone disease may advance long before biochemical abnormalities become evident.
Clinical Connections
Osteoporosis commonly progresses without symptoms until structural failure occurs. Fragility fractures develop when everyday loads exceed weakened bone capacity rather than from high-energy trauma. Vertebral fractures may present with acute or chronic back pain, loss of height, or progressive kyphosis, and are frequently under-recognised because pain may be attributed to ageing or degenerative disease. Hip fractures represent a more overt manifestation and are associated with rapid functional decline, loss of independence, and increased mortality, particularly in older adults with limited physiological reserve.
Several clinical features raise concern for osteoporotic fracture and underlying remodelling failure:
Fracture following minimal trauma, such as a fall from standing height
Vertebral height loss, spinal curvature, or unexplained back pain
Recurrent fractures involving the hip, wrist, vertebrae, or proximal humerus
Assessment focuses on both skeletal injury and contributing biological risk. Diagnosis of osteoporosis is supported by bone density testing, but clinical decision-making also incorporates fracture history, age, medication exposure, endocrine status, renal function, nutritional intake, and mobility. These factors influence bone turnover, repair capacity, and future fracture risk more strongly than bone density alone.
Prevention and management require correction of the processes driving remodelling imbalance. Optimising hormonal status limits excessive resorption, adequate nutrition supports matrix production and mineralisation, mechanical loading through safe weight-bearing activity stimulates bone formation, and fall risk reduction limits exposure to injurious forces. Pharmacological therapies act by suppressing resorption or promoting formation, but their effectiveness depends on addressing these physiological contributors in parallel rather than relying on calcium supplementation in isolation.
Concept Check
Why does osteoporosis involve deterioration of bone microarchitecture rather than bone size alone?
How does uncoupling of osteoclast and osteoblast activity weaken bone over time?
Why does oestrogen deficiency accelerate bone resorption?
How does reduced mechanical loading contribute to osteoporosis?
Why can osteoporosis progress despite normal serum calcium levels?