Bone Tissue Structure: Composition, Organisation & Mechanical Strength
Bone is a living, dynamic connective tissue that provides the structural framework of the body, protects vital organs, enables movement through muscle attachment, stores minerals, and houses blood cell production. Far from being inert, bone tissue is metabolically active and continuously remodels throughout life in response to mechanical forces, hormonal signals, nutritional status, and ageing. The unique strength and resilience of bone arise from its highly specialised microscopic structure, which combines rigid mineral components with flexible organic matrix.
What You Need to Know
Bone tissue consists of a specialised extracellular matrix embedded with living cells. The matrix is composed of two major components: an organic portion, primarily type I collagen fibres, and an inorganic mineral portion, mainly calcium phosphate in the form of hydroxyapatite crystals. Collagen provides tensile strength and flexibility, while the mineral component provides hardness and resistance to compression. The precise balance between these components allows bone to be both strong and slightly flexible, preventing easy fracture under stress.
This balance gives bone its key mechanical properties:
Collagen resists stretching and bending forces
Mineral content resists compression and weight-bearing
Combined structure allows load distribution without structural failure
Bone tissue exists in two distinct architectural forms: compact bone and spongy (trabecular) bone. Compact bone forms the dense outer shell of bones and provides protection and mechanical strength. Spongy bone is located internally, particularly at the ends of long bones and within flat, irregular, and vertebral bones. Its lattice-like structure distributes forces efficiently while reducing overall skeletal weight.
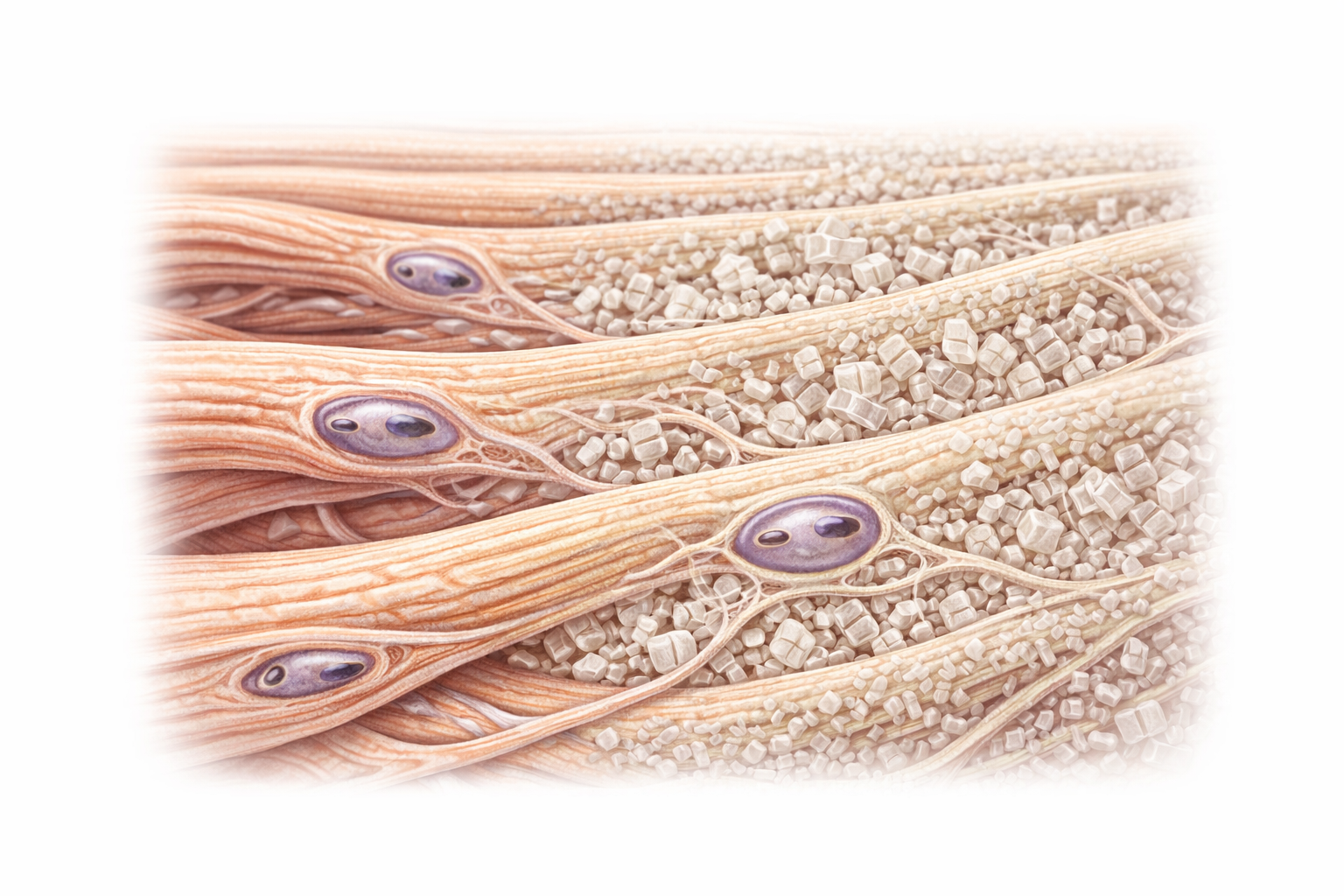
The functional unit of compact bone is the osteon, also known as the Haversian system. Each osteon consists of concentric rings of mineralised matrix called lamellae surrounding a central Haversian canal that contains blood vessels and nerves. This arrangement allows nutrients to reach bone cells efficiently and enables bone to withstand compressive forces along its long axis.
Image: Bone matrix showing organised collagen fibres reinforced by mineral deposition, with osteocytes embedded in lacunae and interconnected through canaliculi, forming a structure that provides both strength and flexibility.
Beyond the Basics
Cellular Organisation of Bone
Bone is a dynamic, living tissue maintained by the coordinated activity of specialised cells embedded within a mineralised matrix. Osteoblasts are responsible for producing the organic framework of bone, known as osteoid, which consists primarily of collagen fibres that provide tensile strength and flexibility. These cells also regulate the deposition of calcium and phosphate salts, allowing the matrix to become rigid and load-bearing. As osteoblasts become surrounded by the material they secrete, they differentiate into osteocytes, which remain permanently embedded within the bone and act as its primary sensory and regulatory cells.
Osteocytes reside within small cavities called lacunae and extend long cytoplasmic processes through microscopic channels known as canaliculi. These processes connect neighbouring osteocytes and link them to blood vessels within the bone, forming an extensive communication network. Through this system, osteocytes detect mechanical strain, changes in nutrient supply, and microdamage within the matrix, allowing them to signal when bone should be strengthened, remodelled, or repaired. This sensory role explains how bone adapts to increased load during exercise and weakens during prolonged immobility.
Bone Resorption and Remodelling
Opposing the actions of osteoblasts are osteoclasts, large multinucleated cells specialised for bone resorption. Osteoclasts attach tightly to the bone surface and create a sealed microenvironment in which they secrete acids and proteolytic enzymes that dissolve mineral salts and degrade collagen. This process releases calcium, phosphate, and other minerals into the bloodstream, contributing to the regulation of blood chemistry as well as structural renewal of bone.
The continual balance between osteoblast-mediated formation and osteoclast-mediated resorption forms the basis of bone remodelling, a process that occurs throughout life. Remodelling allows bone to repair microscopic damage, adapt to changing mechanical stresses, and maintain mineral homeostasis. In healthy bone, formation and resorption remain closely matched, preserving both strength and metabolic function.
Compact and Spongy Bone Architecture
Bone is organised into two structurally distinct but complementary forms: compact bone and spongy bone. Compact bone forms the dense outer shell of most bones and is arranged into cylindrical units called osteons, which provide exceptional resistance to bending and torsion. These tightly packed structures allow bones to withstand the high forces generated by movement and weight-bearing.
Spongy bone, in contrast, consists of a lattice of thin bony plates called trabeculae that are arranged along lines of mechanical stress. This architecture provides remarkable strength while minimising weight, making it ideal for areas subjected to complex loading such as the ends of long bones and the interior of vertebrae. The spaces between trabeculae often contain red bone marrow, where blood cells are produced, linking the skeletal system directly to haematopoietic function.
Vascular and Neural Support
Bone is a highly vascular tissue, with an extensive network of blood vessels supplying oxygen, nutrients, and minerals while removing metabolic waste. These vessels also allow bone to function as a major mineral reservoir, enabling rapid exchange of calcium and phosphate with the bloodstream as physiological demands change.
Bone is also richly innervated, particularly in the periosteum, the tough connective tissue membrane that covers the outer surface of most bones. The periosteum contains pain receptors, blood vessels, and osteogenic cells, making it essential for growth, repair, and the attachment of tendons and ligaments. Its sensitivity explains why fractures and bone injuries are intensely painful, even when the bone itself is not directly damaged.
Mechanical Strength & Adaptation
Bone structure is highly responsive to mechanical load. According to Wolff’s law, bone remodels in response to the stresses placed upon it. Increased mechanical loading stimulates osteoblastic activity and increases bone density, while reduced loading leads to bone resorption and weakening. This principle explains why weight-bearing exercise strengthens bones, while prolonged immobilisation and bed rest result in rapid bone loss.
The alignment of collagen fibres and mineral crystals within lamellae follows specific orientations that resist torsional, tensile, and compressive forces. This microscopic organisation is what enables long bones to withstand bending, twisting, and loading during movement. The cortical thickness of compact bone increases in response to habitual stress, while trabecular thickness and orientation adapt to shifting load patterns.
Clinical Connections
Disorders of bone tissue reflect disruptions in its microscopic structure and cellular activity. In osteoporosis, bone resorption outpaces bone formation, leading to reduced bone density, thinning of trabeculae, and increased fracture risk, particularly in the vertebrae, hips, and wrists. These structural changes may be invisible externally but dramatically affect bone strength.
These conditions alter bone integrity in distinct ways:
Reduced bone mass decreases the ability to withstand load and increases fracture risk
Impaired mineralisation reduces stiffness, allowing bone to deform under pressure
Abnormal remodelling disrupts normal architecture, producing structurally weak bone
In rickets and osteomalacia, inadequate mineralisation of bone matrix due to vitamin D deficiency leads to soft, deformable bones that bend under load. In contrast, osteopetrosis involves impaired osteoclast function, resulting in abnormally dense but brittle bones that are prone to fracture.
Bone tissue structure also explains patterns of fracture. Compact bone fractures tend to propagate along osteonal pathways, while trabecular collapse in spongy bone contributes to vertebral compression fractures.
Following fracture, bone healing recapitulates developmental processes through inflammation, callus formation, mineralisation, and remodelling, all of which depend on the coordinated activity of osteoblasts, osteoclasts, and osteocytes.
Concept Check
Why does the combination of collagen and hydroxyapatite make bone strong yet flexible?
How does the osteon structure support nutrient delivery and mechanical strength?
Why does spongy bone not contain osteons?
How does Wolff’s law explain bone loss during prolonged immobility?
Why does osteoporosis primarily affect trabecular bone?