Skeletal Muscle: Organisation, Specialised Proteins & Functional Design
Skeletal muscle is uniquely designed to convert electrical signals into powerful, precisely controlled mechanical force. While gross anatomy explains how muscles attach to bones and produce movement, it is the microscopic structure of skeletal muscle that explains how contraction actually occurs. Every voluntary movement, posture adjustment, and reflex relies on an intricate internal architecture of contractile proteins, connective tissues, ion channels, and specialised membrane systems.
What You Need to Know
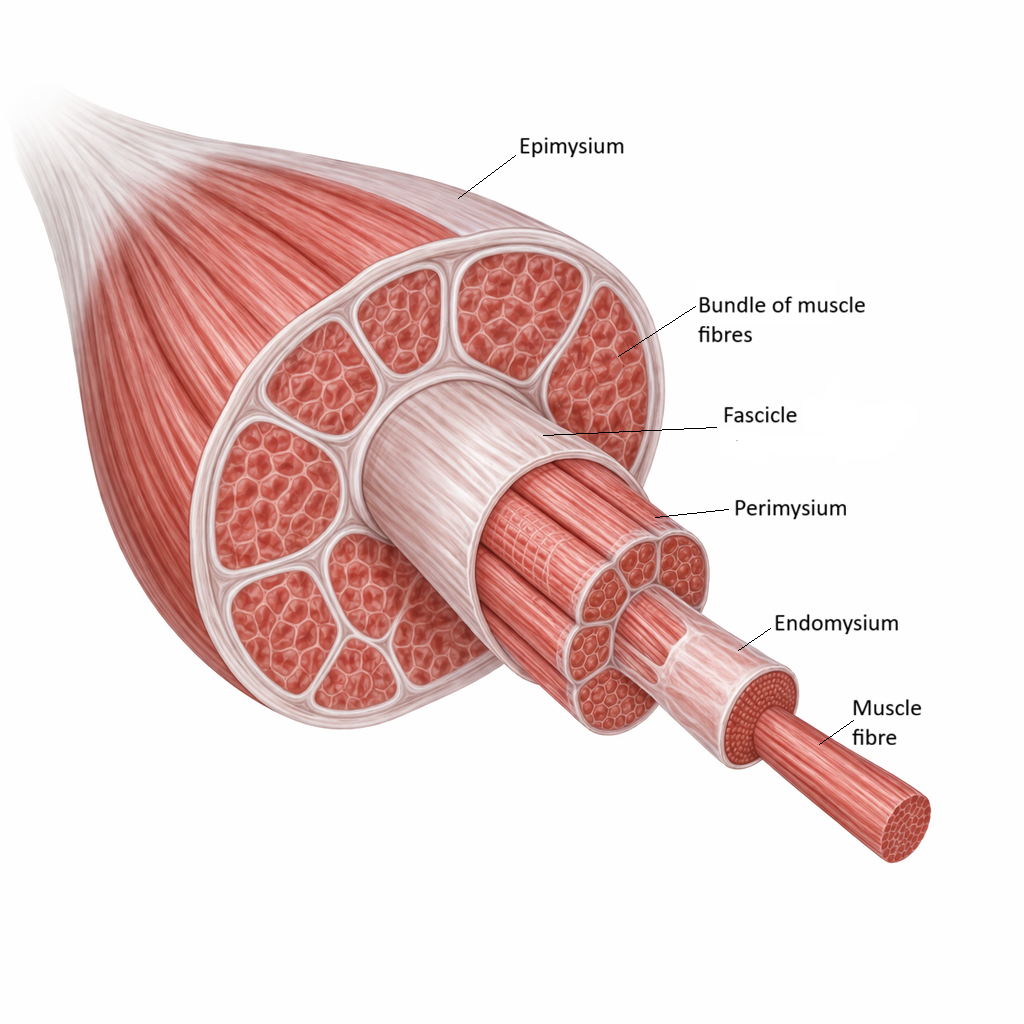
Skeletal muscle is organised in a hierarchical way so that microscopic contractile forces can be amplified and transmitted to bones as movement. This organisation also provides strength, protects delicate muscle fibres, and creates pathways for blood vessels and nerves. The connective tissue layers are not just passive wrappings; they form a continuous force-transmission network that merges into tendons.
The connective tissue organisation of skeletal muscle occurs at three structural levels:
Epimysium, which surrounds the entire muscle
Perimysium, which surrounds bundles of fibres called fascicles
Endomysium, which surrounds each individual muscle fibre
These layers bind the muscle together mechanically and converge into tendons, allowing forces generated inside individual fibres to be transferred efficiently to the skeleton.
A muscle fibre is a single, elongated, multinucleated cell that can span the length of a muscle. It is enclosed by the sarcolemma, a specialised membrane that conducts electrical signals along the fibre surface. Inside the fibre, the sarcoplasm contains mitochondria for ATP production, glycogen for energy storage, myoglobin for oxygen binding, and the contractile machinery that produces force.
Within each muscle fibre are thousands of myofibrils, which run lengthwise through the cell. Myofibrils are composed of repeating sarcomeres, the fundamental contractile units of skeletal muscle. When sarcomeres shorten, the myofibrils shorten, the muscle fibre shortens, and force is transmitted through the connective tissue framework to the tendon and then to bone, producing movement.
Image: Hierarchical organisation of skeletal muscle, showing muscle fibres bundled into fascicles and wrapped by endomysium, perimysium, and epimysium, which provide structural support and transmit force during contraction.
Beyond the Basics
The Sarcomere: The Functional Unit of Contraction
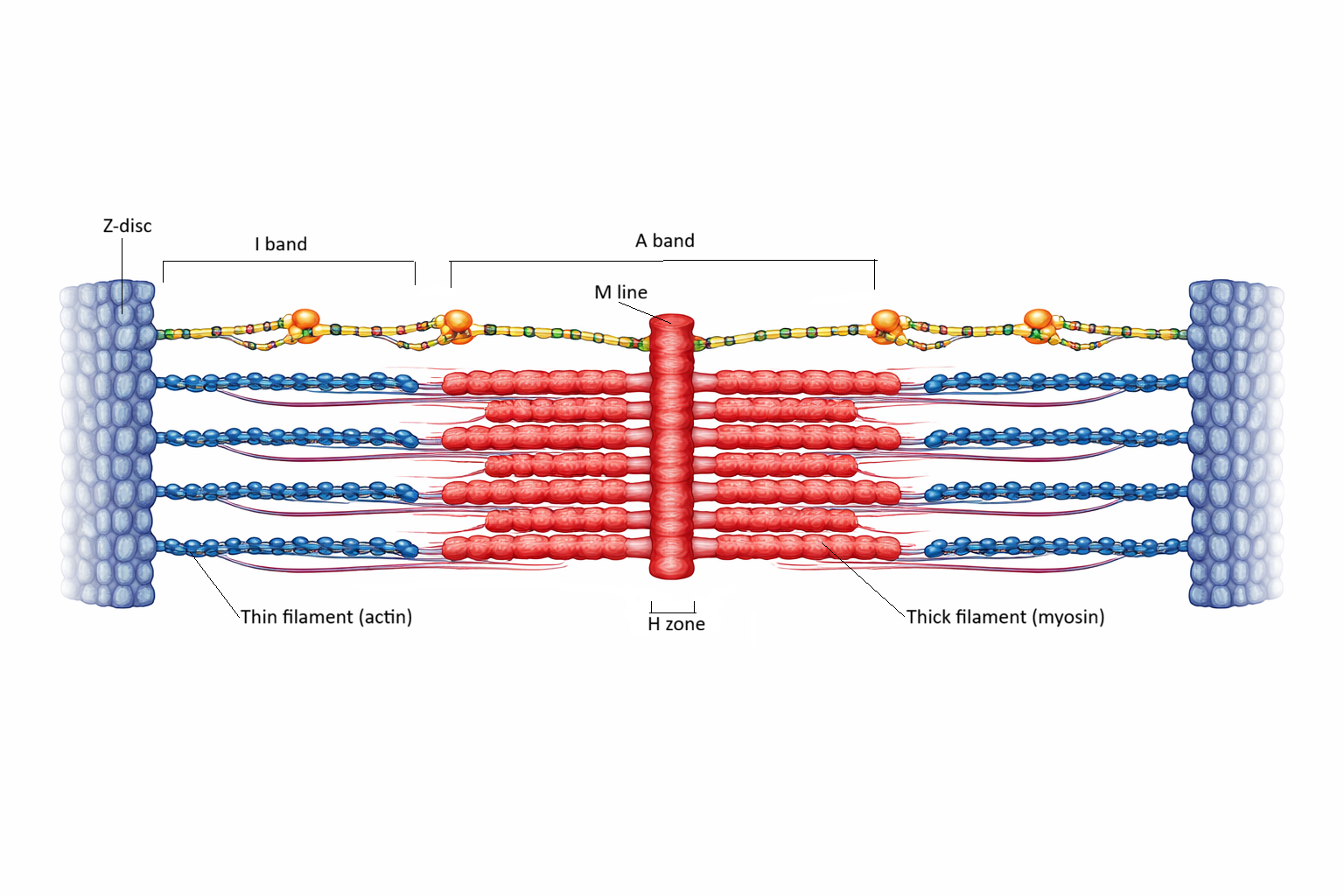
The sarcomere is the fundamental mechanical unit of skeletal muscle, extending from one Z-disc (the boundary between sarcomeres) to the next along each myofibril. Within this microscopic space lies a precisely ordered lattice of thick filaments composed primarily of myosin and thin filaments composed of actin, along with the regulatory proteins tropomyosin and troponin that control when contraction can occur. This arrangement allows muscle to generate force through controlled filament sliding rather than through filament shortening, preserving structural integrity even during repeated contraction.
The spatial organisation of these filaments produces the characteristic striated pattern seen under the microscope. The A band corresponds to the full length of the thick filament and remains constant regardless of muscle length, reflecting the fact that myosin itself does not change size during contraction. The I band contains only thin filaments and shortens as actin slides over myosin, while the H zone, the region where thick filaments are not overlapped by thin filaments, narrows and disappears as contraction progresses. These geometric changes form the physical basis of muscle shortening and explain why contraction increases filament overlap without compressing the filaments themselves.
Sarcomeres are arranged end-to-end in long chains within each myofibril. When thousands of sarcomeres shorten together, their individual movements sum to produce visible muscle contraction. The number of sarcomeres arranged in series determines how far a muscle fibre can shorten, while the number arranged in parallel determines how much force it can generate. This relationship explains why long, slender muscles are specialised for speed and range of motion, whereas thicker muscles are specialised for strength.
Image: The sarcomere shown in a partially contracted state, with actin and myosin filaments overlapping to generate force, while structural regions including the Z-discs, A band, I band, H zone, and M line remain clearly defined.
Membrane Systems: Electrical Signalling and Calcium Control
Skeletal muscle fibres are unusually large cells, and their size creates a challenge for rapid signal transmission. To overcome this, the sarcolemma (the muscle cell membrane) forms deep invaginations called transverse (T) tubules that carry electrical impulses from the surface of the cell directly into its interior. This ensures that an action potential reaches every myofibril almost simultaneously, allowing uniform contraction rather than a slow wave spreading inward.
Surrounding each myofibril is the sarcoplasmic reticulum (SR), a specialised form of smooth endoplasmic reticulum that acts as an intracellular calcium reservoir. When a T-tubule depolarises, the SR rapidly releases calcium into the cytoplasm, raising calcium levels from extremely low to very high within milliseconds. This sudden increase in calcium is what switches on the interaction between actin and myosin, allowing force to be generated.
The close anatomical relationship between a T-tubule and two adjacent terminal cisternae of the sarcoplasmic reticulum forms a triad. Triads ensure precise excitation–contraction coupling (conversion of an electrical signal into mechanical force), allowing muscle fibres to contract quickly, synchronously, and reliably.
Structural Proteins and Force Transmission
The contractile filaments of skeletal muscle are stabilised by a network of specialised structural proteins that keep everything aligned and able to withstand mechanical stress. Titin spans from the Z-disc to the thick filament and acts as both a molecular spring and a scaffold, helping the sarcomere return to its resting length after stretch and contributing to passive muscle elasticity.
Desmin forms an intermediate filament network that links neighbouring myofibrils together and anchors them to the sarcolemma. This ensures that when one sarcomere contracts, its force is transmitted evenly across the whole muscle fibre rather than being lost locally.
The internal contractile machinery is anchored to the extracellular matrix through a protein complex that includes dystrophin. This linkage prevents the sarcolemma from tearing during contraction and allows force generated inside the fibre to be transferred to tendons and bones. When this system fails, as in muscular dystrophy, muscle fibres become fragile and easily damaged.
Metabolic Infrastructure of Muscle Fibres
Skeletal muscle fibres are highly energy-dependent cells, and their microscopic organisation reflects this. Mitochondria are densely packed between myofibrils and beneath the sarcolemma, placing ATP production close to the sites where energy is needed for cross-bridge cycling, calcium pumping, and membrane excitability.
Muscle fibres also store large amounts of glycogen, which can be rapidly broken down to supply glucose for ATP generation, and myoglobin, an oxygen-binding protein that helps deliver oxygen inside the fibre. Together, these adaptations ensure that muscle can sustain contraction even during intense activity or when blood flow is temporarily limited.
Clinical Connections
Many muscle diseases arise from disruptions at the microscopic level rather than from gross changes in muscle shape or size. In muscular dystrophies, defects in structural proteins such as dystrophin weaken the links between the contractile machinery and the muscle cell membrane. As a result, fibres are easily damaged during normal contraction, leading to progressive fibre breakdown, inflammation, and replacement with fat and connective tissue. This explains why patients gradually lose strength even though their nerves and neuromuscular junctions may initially remain intact.
In myopathies, the contractile proteins themselves are affected. When actin, myosin, or regulatory proteins function abnormally, muscle fibres cannot generate normal force despite receiving appropriate neural stimulation. Patients therefore experience weakness and fatigue without the sensory loss or reflex changes seen in nerve disorders, which helps distinguish myopathic from neuropathic disease in clinical assessment.
These microscopic defects produce distinct patterns of dysfunction that are seen across muscle disease:
Structural protein defects cause fibre fragility and progressive degeneration
Contractile protein dysfunction leads to reduced force despite intact neural input
Calcium handling abnormalities produce sustained contraction and metabolic crisis
Loss of mitochondria and myofibrils results in weakness, fatigue, and poor endurance
Disorders of calcium handling illustrate how finely tuned muscle physiology must be. In malignant hyperthermia, abnormal calcium release from the sarcoplasmic reticulum causes continuous contraction, rapid ATP consumption, extreme heat production, and muscle breakdown when triggered by certain anaesthetic agents. This leads to hyperthermia, acidosis, and potentially fatal organ failure without rapid treatment.
Age-related muscle decline (sarcopenia) reflects cumulative microscopic deterioration. Loss of myofibrils reduces force production, fewer mitochondria limit energy supply, and impaired calcium regulation slows contraction and relaxation. Fatty and connective tissue infiltration further weakens muscle, explaining why older adults become slower, weaker, and more prone to falls even when joints and nerves remain intact.
After denervation or prolonged immobilisation, such as following stroke, spinal cord injury, or extended bed rest, muscle fibres rapidly lose their internal organisation. Myofibrils degrade, mitochondria decline, and connective tissue accumulates, producing weakness and stiffness. This is why early mobilisation, electrical stimulation, and restoration of neural input are essential to preserve muscle structure and function during recovery.
Concept Check
Why are skeletal muscle fibres multinucleated?
How does the organisation of sarcomeres explain the striated appearance of skeletal muscle?
Why are T-tubules and the sarcoplasmic reticulum essential for normal contraction?
What role does dystrophin play in protecting muscle fibres?
How do microscopic changes explain muscle weakness in ageing and disease?