Glomerulonephritis: Immune-Mediated Glomerular Inflammation and Filtration Failure
Glomerulonephritis refers to a group of conditions characterised by inflammation and injury of the glomeruli, the kidney’s primary filtration units. Unlike tubular causes of kidney injury, glomerulonephritis primarily disrupts filtration at its source, leading to abnormal loss of blood and protein into the urine and reduced clearance of waste. Understanding the pathophysiology of glomerulonephritis explains why urine abnormalities are often prominent, why blood pressure and fluid balance are affected early, and why immune activity plays a central role in kidney damage.
What You Need to Know
Glomerulonephritis refers to inflammation and injury of the glomeruli, the microscopic filtration units of the kidney. The glomerulus acts as a highly selective barrier that allows water and small solutes to pass into the urine while retaining blood cells and large proteins within the circulation. When this structure becomes inflamed or damaged, that selectivity is lost, and substances that should remain in the blood begin to leak into the urine.
As the glomerular capillary walls swell and become more permeable, filtration becomes both less efficient and less precise. Protein and red blood cells may pass through the damaged barrier, while the overall filtration rate often falls because inflammation narrows capillary lumens and reduces blood flow through the glomerulus. This combination explains why glomerulonephritis produces both abnormal urine findings and impaired kidney function.
Key consequences of glomerular injury include:
protein leaking into the urine, reducing plasma oncotic pressure and contributing to oedema
red blood cells entering the urine, producing haematuria
reduced filtration leading to fluid, electrolyte, and waste retention
Glomerulonephritis is most often triggered by immune-mediated processes, such as infection-related immune complex deposition or autoimmune disease. Although the initial insult occurs at the microscopic level, the resulting disruption to filtration affects the entire body, making glomerular disease a major cause of fluid overload, hypertension, and acute or chronic kidney injury.
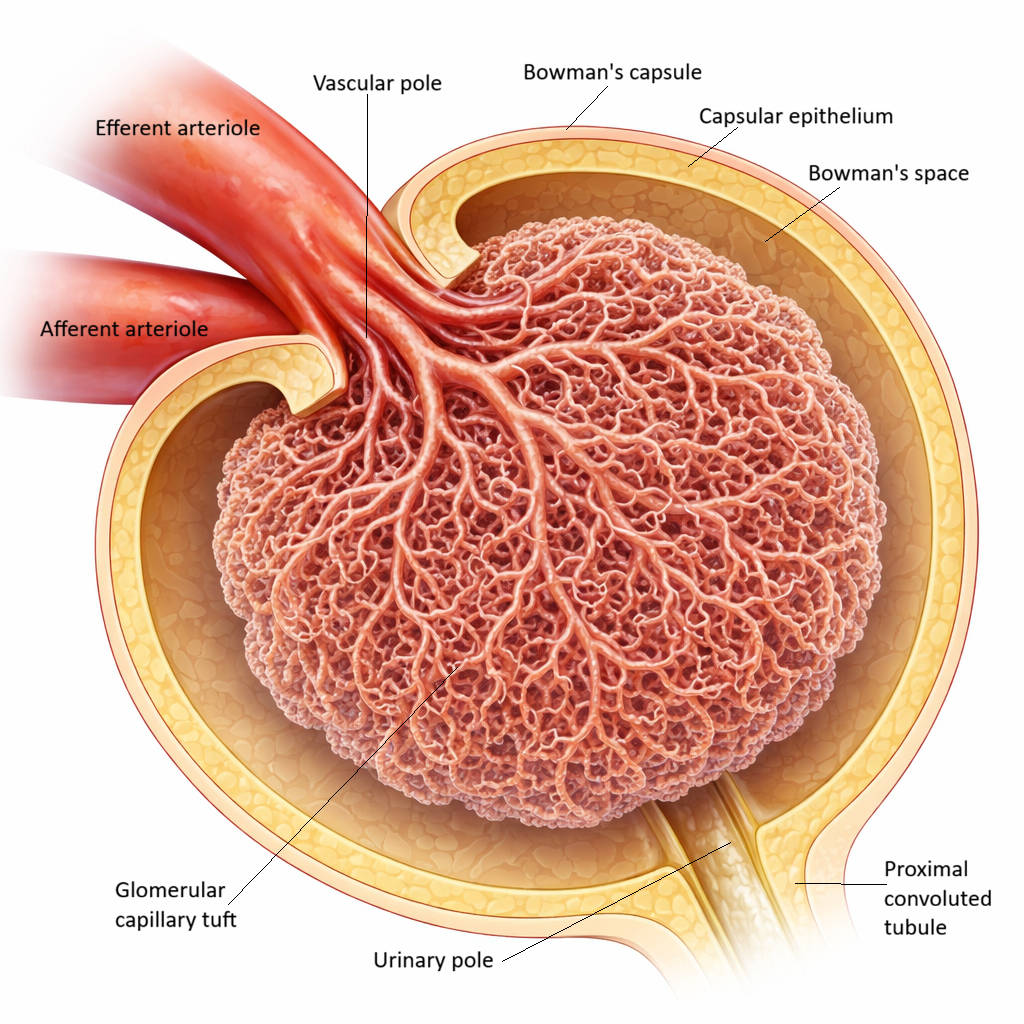
Image: The glomerulus is a network of capillaries within the renal corpuscle where blood filtration begins. In glomerulonephritis, inflammation of these structures disrupts filtration, leading to leakage of proteins and blood into the urine and reduced kidney function.
Beyond the Basics
Immune activation and glomerular injury
Glomerulonephritis is an immune-mediated disease in which inflammatory processes target the glomerular capillaries. Immune complexes, clusters of antibodies bound to antigens, may become trapped within the glomerular basement membrane, or antibodies may bind directly to glomerular structures. These events activate the complement system, a cascade of immune proteins that amplifies inflammation and recruits white blood cells. Activated immune cells release cytokines, proteases and reactive oxygen species, which injure endothelial cells, podocytes and the basement membrane itself. Because many immune triggers follow infection or occur as part of autoimmune disease, glomerular inflammation is often delayed rather than a direct result of kidney exposure to pathogens.
Increased permeability and protein loss
As inflammation alters the glomerular filtration barrier, its normally selective permeability is lost. The filtration slits between podocytes widen and the basement membrane becomes more porous, allowing plasma proteins, particularly albumin, to pass into the urine. Loss of albumin reduces plasma oncotic pressure, the force that normally retains fluid within the vascular space. Fluid then shifts into the interstitial tissues, producing oedema and contributing to intravascular volume depletion even when total body fluid is increased. Although heavy protein loss is more typical of nephrotic syndromes, even moderate proteinuria in glomerulonephritis is sufficient to disturb fluid balance and promote swelling.
Haematuria and reduced filtration
Inflammation also disrupts the integrity of glomerular capillary walls, allowing red blood cells to enter the filtrate. As these cells pass through the damaged filtration barrier, they become distorted, producing dysmorphic red blood cells characteristic of glomerular haematuria. At the same time, inflammatory swelling and cellular infiltration narrow capillary lumens, reducing the surface area available for filtration. Glomerular filtration rate falls, leading to accumulation of urea, creatinine and other metabolic wastes in the bloodstream and contributing to acute or progressive renal dysfunction.
Sodium retention, fluid overload, and hypertension
As filtration declines, the kidneys interpret reduced flow as a threat to circulating volume. This triggers sodium and water retention through increased tubular reabsorption and activation of the renin–angiotensin–aldosterone system, which promotes vasoconstriction and further sodium retention. Inflammatory changes within the glomerulus also raise intraglomerular pressure, increasing mechanical stress on already damaged capillaries. The combination of fluid retention and elevated pressure drives oedema and hypertension while accelerating glomerular injury, creating a self-perpetuating cycle of inflammation, pressure and nephron loss.
Acute and chronic patterns of disease
In acute glomerulonephritis, inflammation develops rapidly but may resolve if immune activity is controlled and structural damage is limited. During recovery, glomerular cells can restore architecture and filtration may return toward baseline. Chronic glomerulonephritis reflects persistent immune activation that leads to progressive scarring of the glomeruli and surrounding interstitium. As functioning nephrons are lost, remaining glomeruli are exposed to increased workload and pressure, promoting further sclerosis and irreversible progression toward chronic kidney disease.
Systemic effects of glomerular inflammation
Glomerulonephritis is often part of a broader immune disorder rather than an isolated renal process. Patients may experience systemic features such as fever, malaise, rashes or joint symptoms depending on the underlying cause. As renal function declines, fluid overload, hypertension, uraemia and electrolyte disturbance disrupt whole-body homeostasis. These effects demonstrate how injury at the level of the glomerulus can produce widespread physiological consequences well beyond the kidney itself.
Clinical Connections
Glomerulonephritis typically presents with urine and fluid changes that reflect damage to the glomerular filtration barrier. Patients may notice dark, cola-coloured, or blood-tinged urine from glomerular bleeding, and foamy urine from protein loss. Urine microscopy often shows dysmorphic red blood cells or red cell casts, which point to a glomerular source of haematuria.
Common clinical features include:
proteinuria leading to periorbital and peripheral oedema
rising blood pressure due to sodium and water retention
reduced urine output and increasing creatinine as filtration falls
As plasma albumin is lost and fluid shifts into tissues, patients may develop rapid weight gain, ankle swelling, or shortness of breath from pulmonary congestion. Hypertension is both a consequence of glomerular injury and a driver of further nephron damage, making blood pressure control a key part of management.
In practice, treatment focuses on:
controlling blood pressure to reduce intraglomerular stress
managing fluid overload with diuretics
suppressing immune-mediated inflammation when indicated
Early detection through urine testing and monitoring of renal function is critical, as ongoing inflammation and pressure within the glomeruli increase the risk of irreversible scarring and progression to chronic kidney disease.
Concept Check
Why does glomerular inflammation lead to haematuria and proteinuria?
How does immune activation damage the filtration barrier?
Why is hypertension common in glomerulonephritis?
How does persistent inflammation lead to chronic kidney disease?
Why are urine findings often more prominent than tubular abnormalities?