Bladder Structure: Anatomy, Histology and Functional Organisation
The urinary bladder is a highly specialised hollow organ designed to store urine at low pressure and expel it efficiently during voiding. Its layered structure supports these dual functions by combining distensible epithelial tissue, robust smooth muscle and a sophisticated nerve supply. Although often clinically overshadowed by the kidneys, the bladder’s anatomy is essential for continence, voluntary control of micturition and protection of the upper urinary tract.
What You Need to Know
The urinary bladder is a highly specialised muscular reservoir located within the pelvic cavity, designed to store urine at low pressure and expel it in a controlled manner when appropriate. Its wall is organised into distinct layers that work together to allow repeated filling and emptying without tissue damage. The innermost mucosa is lined with transitional epithelium, which can stretch and recoil as bladder volume changes, while the underlying lamina propria provides structural support and elasticity.
Surrounding these layers is the detrusor muscle, a thick coat of interlacing smooth muscle fibres responsible for generating the force required for voiding. The outermost layer is either adventitia or serosa, depending on the bladder’s relationship to the peritoneum, anchoring it within the pelvis while allowing mobility. At the bladder neck, smooth muscle fibres form the internal urethral sphincter, which contributes to involuntary continence and works in coordination with the external urethral sphincter to regulate urine flow.
Several specialised anatomical features are essential for normal bladder function:
the transitional epithelium, which accommodates large volume changes without leakage or injury
the detrusor muscle, which contracts uniformly to empty the bladder efficiently
the trigone, a smooth, triangular region between the ureteric orifices and urethral opening that helps coordinate urine flow and bladder emptying
the sphincteric structures, which maintain continence during filling and relax appropriately during voiding
These structural adaptations give the bladder its characteristic compliance, allowing it to store increasing volumes of urine with minimal rise in internal pressure. This anatomy, combined with precise neural control, protects the upper urinary tract from pressure-related injury and enables effective, voluntary control of micturition.
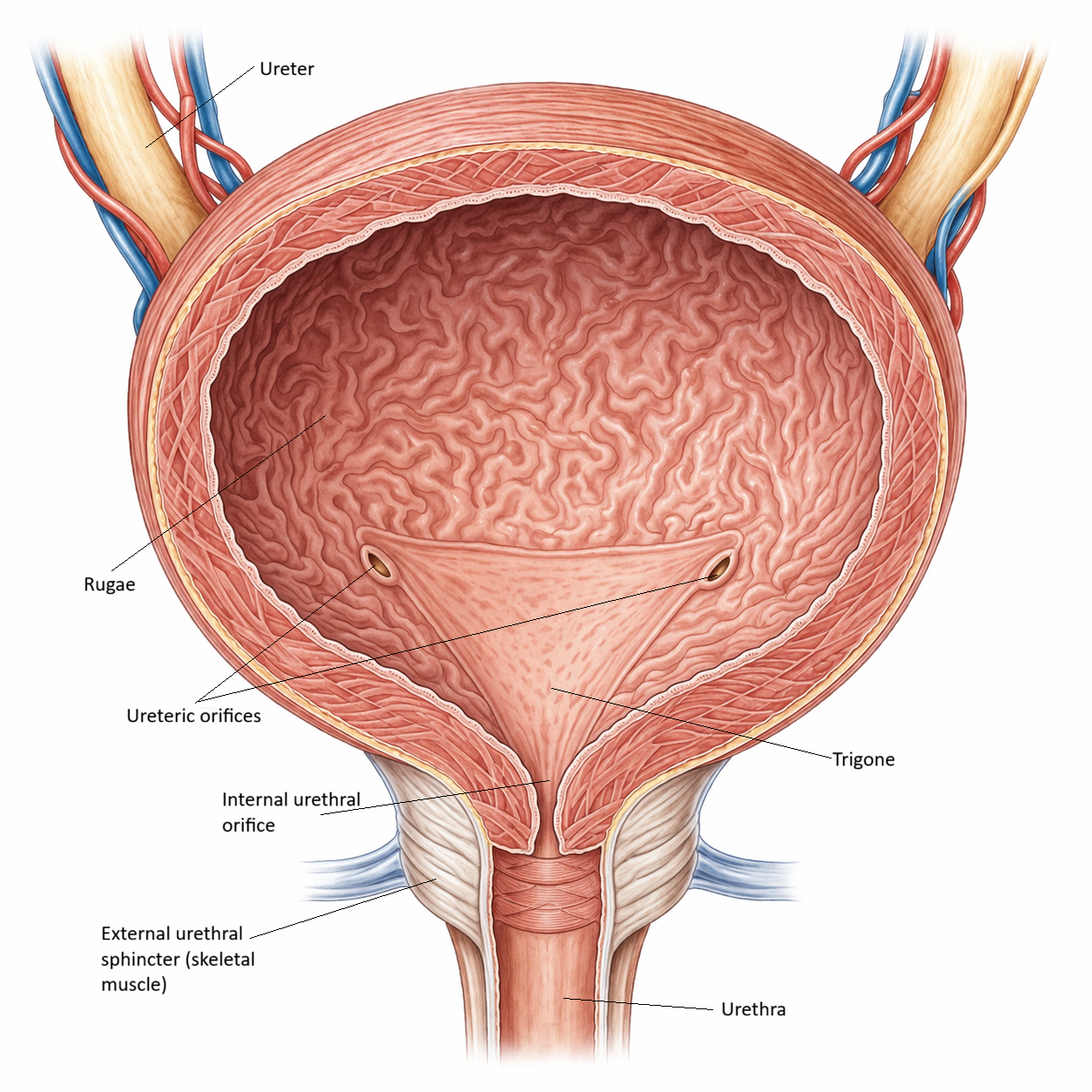
Image: Urinary bladder (internal view). A distensible reservoir with mucosal rugae for expansion and a smooth trigone that directs urine toward the urethra.
Beyond the Basics
Mucosa and transitional epithelium
The innermost layer of the bladder is lined by transitional epithelium, also known as urothelium, a highly specialised tissue adapted to repeated cycles of filling and emptying. When the bladder is empty, the epithelial cells appear cuboidal and the mucosa forms folds known as rugae. As the bladder fills, the urothelium stretches and the cells flatten into a squamous-like configuration, allowing marked expansion without epithelial damage.
Superficial umbrella cells form tight junctions that create an effective barrier against urine, protecting deeper tissues from potentially toxic solutes. These cells contain specialised membrane plaques that unfold during filling and refold during emptying, preserving epithelial integrity while maintaining impermeability.
Beneath the urothelium lies the lamina propria, a vascular connective tissue layer containing blood vessels, nerves, and immune cells. In addition to providing mechanical support, this layer plays an important role in sensory signalling during bladder filling, contributing to perception of bladder volume and urgency.
Detrusor muscle
The detrusor muscle forms the thick middle layer of the bladder wall and consists of smooth muscle fibres arranged in interwoven longitudinal, circular, and oblique orientations. This complex three-dimensional organisation allows the bladder to contract evenly, ensuring efficient expulsion of urine during voiding.
A key functional property of the detrusor is its ability to accommodate increasing volumes with minimal rise in intravesical pressure, a phenomenon known as stress relaxation. This compliance is essential for safe urine storage, as it prevents transmission of high pressures to the ureters and kidneys, reducing the risk of reflux and upper urinary tract damage.
During micturition, coordinated detrusor contraction generates the pressure required to overcome urethral resistance and empty the bladder effectively.
Bladder neck and internal urethral sphincter
At the base of the bladder, the detrusor muscle transitions into the bladder neck, where smooth muscle fibres form the internal urethral sphincter. This sphincter is not under voluntary control and plays a critical role in maintaining continence during bladder filling by remaining tonically contracted.
In males, the internal urethral sphincter also prevents retrograde ejaculation by closing the bladder neck during emission. Its structural design allows firm closure at rest and coordinated relaxation during voiding, ensuring unidirectional urine flow.
Trigone: a structurally distinct region
The trigone is a smooth, triangular area defined by the two ureteric orifices and the internal urethral opening. Unlike the remainder of the bladder mucosa, the trigone lacks rugae and remains relatively fixed in position. This rigidity ensures that the ureteric openings remain patent during bladder filling and emptying, helping prevent vesicoureteral reflux.
The trigone is richly innervated and contains a dense sensory network that contributes to detection of bladder filling and initiation of the urge to void, linking structural specialisation with sensory control.
Adventitia and serosa
The outer covering of the bladder varies with anatomical location. Most of the bladder is surrounded by adventitia, a connective tissue layer that anchors the organ within the pelvic cavity. The superior surface, which lies adjacent to the peritoneum, is covered by serosa.
These outer layers provide stability while still allowing the bladder to expand upward into the abdominal cavity as it fills, accommodating increasing urine volumes without compromising surrounding structures.
Blood supply and innervation
The bladder receives arterial blood primarily from the superior and inferior vesical arteries, branches of the internal iliac artery, with venous drainage following a similar pattern. This rich vascular supply supports the metabolic demands of the detrusor and urothelium during repeated cycles of filling and voiding.
Innervation of the bladder reflects its dual storage and emptying functions. Parasympathetic fibres from the pelvic nerves stimulate detrusor contraction during micturition. Sympathetic fibres promote storage by relaxing the detrusor and tightening the internal urethral sphincter. Somatic fibres innervate the external urethral sphincter, providing voluntary control. The layered organisation of neural pathways within the bladder wall underpins the precise coordination required for normal continence and effective voiding.
Clinical Connections
Loss of urothelial integrity compromises the bladder’s protective barrier, increasing susceptibility to urinary tract infection and irritation from urinary solutes. Conditions that damage or inflame the urothelium reduce its impermeability, exposing deeper tissues to chemical injury and triggering urgency, dysuria, and recurrent infection.
Alterations in detrusor structure and function have major clinical consequences. Detrusor overactivity, commonly associated with ageing, neurological disease, or altered sensory signalling, leads to urgency, frequency, and urge incontinence. In contrast, chronic outlet obstruction at the bladder neck or urethra forces the detrusor to generate higher pressures to empty, resulting in muscle hypertrophy, reduced compliance, and progressively elevated intravesical pressure. Over time, this pressure can be transmitted to the upper urinary tract, threatening renal function.
Common causes of bladder dysfunction are:
urothelial damage, increasing infection risk and irritative symptoms
detrusor overactivity, producing urgency and urge incontinence
outflow obstruction, leading to detrusor hypertrophy, poor compliance, and urinary retention
elevated bladder pressure, increasing risk of vesicoureteral reflux and renal impairment
Damage to neural pathways produces equally characteristic effects. Spinal cord injury, diabetic neuropathy, and central neurological disease disrupt coordination between detrusor contraction and sphincter relaxation, resulting in retention, incontinence, or dyssynergia depending on the level and extent of injury. These patterns directly reflect the distinct structural and functional roles of smooth and skeletal muscle within the bladder wall and outlet.
Concept Check
How does transitional epithelium allow the bladder to expand without compromising its barrier function
Why does the detrusor muscle require fibres arranged in multiple orientations
What structural features make the trigone functionally different from the rest of the bladder mucosa
How does the structure of the internal urethral sphincter contribute to continence
Why is bladder compliance important for protecting the upper urinary tract