Juxtaglomerular Apparatus: Structure, Cellular Components and Filtration Regulation
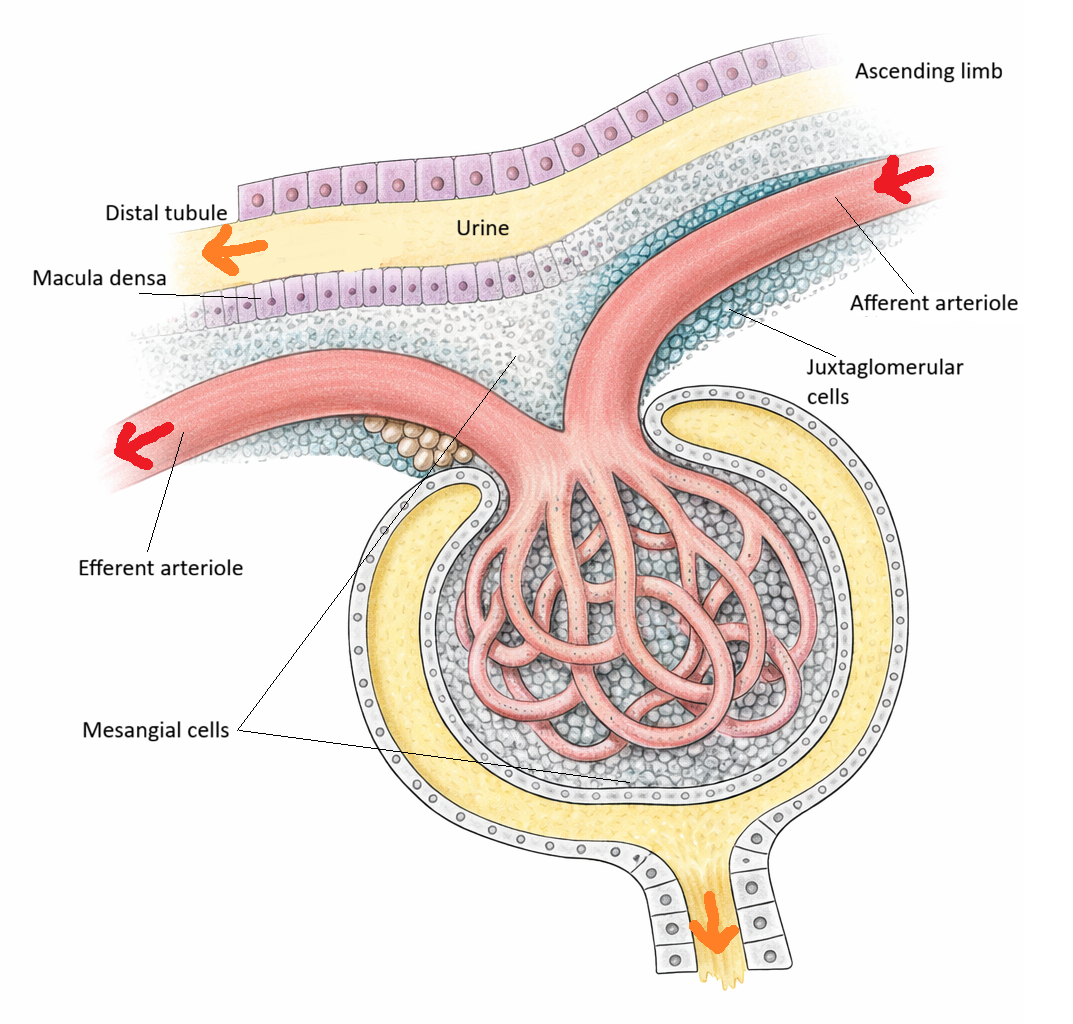
The juxtaglomerular apparatus (JGA) is a specialised anatomical region where the distal convoluted tubule comes into intimate contact with the afferent and efferent arterioles of the same nephron. Despite its small size, the JGA is central to regulating glomerular filtration rate and systemic blood pressure. Its structure enables precise communication between tubular fluid composition and arteriolar tone, ensuring that filtration remains stable under varying physiological conditions.
What You Need to Know
The juxtaglomerular apparatus (JGA) is a specialised regulatory structure located where the distal convoluted tubule comes into close contact with the glomerulus and its afferent arteriole. Its primary role is to match glomerular filtration to both tubular sodium delivery and systemic circulatory demands. Rather than functioning as a passive anatomical junction, the JGA actively senses changes in tubular fluid composition and adjusts renal haemodynamics and hormone release in response.
This regulatory role is achieved through the coordinated activity of three closely associated cellular components, each with a distinct but interdependent function:
the macula densa, which detects changes in sodium chloride concentration in the distal tubule
the juxtaglomerular (granular) cells, which modify afferent arteriole tone and secrete renin
the extraglomerular mesangial cells, which provide structural support and facilitate signalling between tubular and vascular elements
The JGA links tubular function directly to vascular control, allowing rapid adjustment of afferent arteriole diameter and renin release. This ensures that glomerular filtration rate remains stable despite fluctuations in blood pressure, fluid status, or sodium intake.
The juxtaglomerular apparatus is also a key interface between local renal autoregulation and systemic blood pressure control. By integrating tubuloglomerular feedback with activation of the renin–angiotensin–aldosterone system, the JGA plays a central role in maintaining extracellular fluid volume, electrolyte balance, and long-term cardiovascular homeostasis.
Image: The juxtaglomerular apparatus is a specialised region at the vascular pole of the renal corpuscle where the distal convoluted tubule contacts the afferent arteriole. It integrates tubular sodium chloride sensing with regulation of renin release and glomerular filtration.
Beyond the Basics
Macula Densa: Specialised Tubular Epithelium
The macula densa is a segment of the distal convoluted tubule where the epithelial cells become taller, more densely packed and more columnar in appearance. These cells lack the brush border characteristic of the proximal tubule, but they contain specialised transporters that sense sodium chloride concentration.
When NaCl levels in the tubular fluid rise, macula densa cells detect this through Na-K-2Cl cotransporters. In response, they release paracrine mediators that constrict the afferent arteriole, reducing glomerular filtration rate and limiting further NaCl delivery.
Conversely, when NaCl levels fall, the macula densa signals to dilate the afferent arteriole and stimulate renin release, helping restore pressure and filtration. Structurally, the macula densa forms the sensory limb of this feedback loop.
Juxtaglomerular (Granular) Cells: Renin-Producing Smooth Muscle
Juxtaglomerular cells are modified smooth muscle cells located primarily in the wall of the afferent arteriole, though some extend into the efferent arteriole. These cells contain prominent secretory granules that store renin, the enzyme that initiates the renin–angiotensin–aldosterone cascade.
Their dual identity as muscle and endocrine cells allows them to integrate mechanical stretch signals with chemical cues from the macula densa. A reduction in arteriolar pressure reduces the stretch on JG cells, prompting renin release. Sympathetic activation via beta-1 receptors also stimulates these cells. The structural adaptation of smooth muscle into a renin-secreting endocrine cell type highlights the complexity of renal vascular regulation.
Extraglomerular Mesangial Cells: Support and Signal Transmission
These cells occupy the triangular region between the macula densa and the arterioles. They form part of the “Lacis cell” region and are continuous with intraglomerular mesangial cells. Although their exact role remains less defined than the other components, structural and physiological studies suggest that extraglomerular mesangial cells facilitate communication between the macula densa and juxtaglomerular cells. Their cytoplasmic processes form a relay network, transmitting signals that coordinate vascular and endocrine responses. They also provide structural stability and modulate extracellular matrix composition, helping maintain the architecture of the JGA.
Physical Arrangement and Functional Integration
The JGA exists precisely where the distal tubule loops back toward its parent glomerulus. This physical arrangement allows the nephron to sense its own performance. The close proximity of tubular epithelium, vascular smooth muscle and mesangial cells enables efficient paracrine signalling.
Tubuloglomerular feedback arises directly from this architecture. When macula densa cells detect changes in tubular NaCl, their structural positioning allows rapid modification of afferent arteriole tone. Similarly, the JG cells’ placement directly within the arteriole wall allows instant release of renin into the circulation when required. The structural organisation of the JGA is therefore essential for stabilising GFR and regulating systemic blood pressure.
Clinical Connections
Disruption of juxtaglomerular apparatus (JGA) function has direct implications for blood pressure control, fluid balance, and kidney perfusion. When renal blood flow decreases, such as with renal artery stenosis, hypovolaemia, or heart failure, juxtaglomerular cells increase renin release in response to decreased perfusion pressure and sympathetic activation. This drives activation of the renin–angiotensin–aldosterone system (RAAS), promoting vasoconstriction and sodium retention. When persistent or inappropriate, this contributes to sustained hypertension and ongoing renal injury.
Altered sodium delivery to the macula densa also has important clinical consequences. Reduced NaCl delivery (e.g. with volume depletion or aggressive diuresis) signals a perceived drop in filtration, triggering renin release and afferent arteriolar dilation to increase glomerular filtration. In contrast, increased NaCl delivery suppresses renin and promotes afferent constriction. Disruption of this feedback—seen in chronic kidney disease (CKD)—leads to impaired autoregulation, with greater variability in glomerular filtration and progressive nephron damage.
Many commonly used medications exert their effects by modifying JGA signalling:
ACE inhibitors / ARBs - reduce angiotensin II–mediated vasoconstriction and aldosterone release, lowering blood pressure and intraglomerular pressure
Beta-blockers - decrease renin secretion by inhibiting sympathetic stimulation of juxtaglomerular cells
Diuretics - increase NaCl delivery to the macula densa, altering tubuloglomerular feedback and indirectly influencing renin release
These effects are clinically relevant when initiating or titrating therapy. For example, ACE inhibitors and ARBs can cause an initial rise in creatinine due to reduced efferent arteriolar constriction, particularly in patients with renal artery stenosis or pre-existing CKD. Similarly, diuretic-induced changes in sodium delivery can activate RAAS, contributing to compensatory sodium retention if not carefully managed.
Concept Check
How does the structure of the macula densa enable it to detect changes in tubular sodium chloride concentration
Why are juxtaglomerular cells uniquely adapted to act as both smooth muscle and endocrine cells
How do extraglomerular mesangial cells contribute to communication within the JGA
Why is the physical proximity of the distal tubule and afferent arteriole essential for tubuloglomerular feedback
How can structural changes to the JGA contribute to hypertension and impaired renal autoregulation