Glomerular Filtration Barrier: Microstructure, Selectivity and Filtration Dynamics
The glomerular filtration barrier is one of the most specialised and efficient filtration interfaces in the human body. Its structure is uniquely designed to allow rapid passage of water and small solutes while preventing the movement of proteins and cellular elements into the urine. The barrier’s integrity is essential for normal kidney function, and even subtle disruptions can lead to clinically significant proteinuria, oedema and progressive renal disease. Understanding its microanatomy provides the foundation for interpreting glomerulopathies, nephrotic and nephritic syndromes, and the physiological principles driving filtration.
What You Need to Know
The glomerular filtration barrier is a highly specialised interface that separates circulating blood from the filtrate that will become urine. It is formed by three tightly integrated layers: the fenestrated endothelium of the glomerular capillaries, the glomerular basement membrane, and the visceral epithelial cells known as podocytes. Together, these structures allow enormous volumes of plasma to be filtered while preserving the essential components of blood, including cells and most proteins.
Filtration across this barrier is driven by hydrostatic pressure within the glomerular capillaries and controlled by both physical and electrostatic selectivity. Each layer contributes a specific filtering role:
the fenestrated endothelium allows water and small solutes to pass while blocking blood cells
the basement membrane acts as a size- and charge-selective gel, repelling negatively charged proteins such as albumin
podocyte slit diaphragms provide a final molecular sieve that restricts large molecules from entering the filtrate
This multilayered structure ensures that useful substances such as glucose, electrolytes, and amino acids are filtered freely, while macromolecules remain in the circulation.
Although the three layers are anatomically distinct, they function as a single, integrated filtration unit. The basement membrane provides mechanical support and charge selectivity, while podocytes stabilise the capillary wall and fine-tune permeability through their interlocking foot processes. Even subtle structural or electrical changes to any layer can significantly alter what is filtered, making the integrity of this barrier essential for maintaining plasma composition and preventing protein loss in the urine.
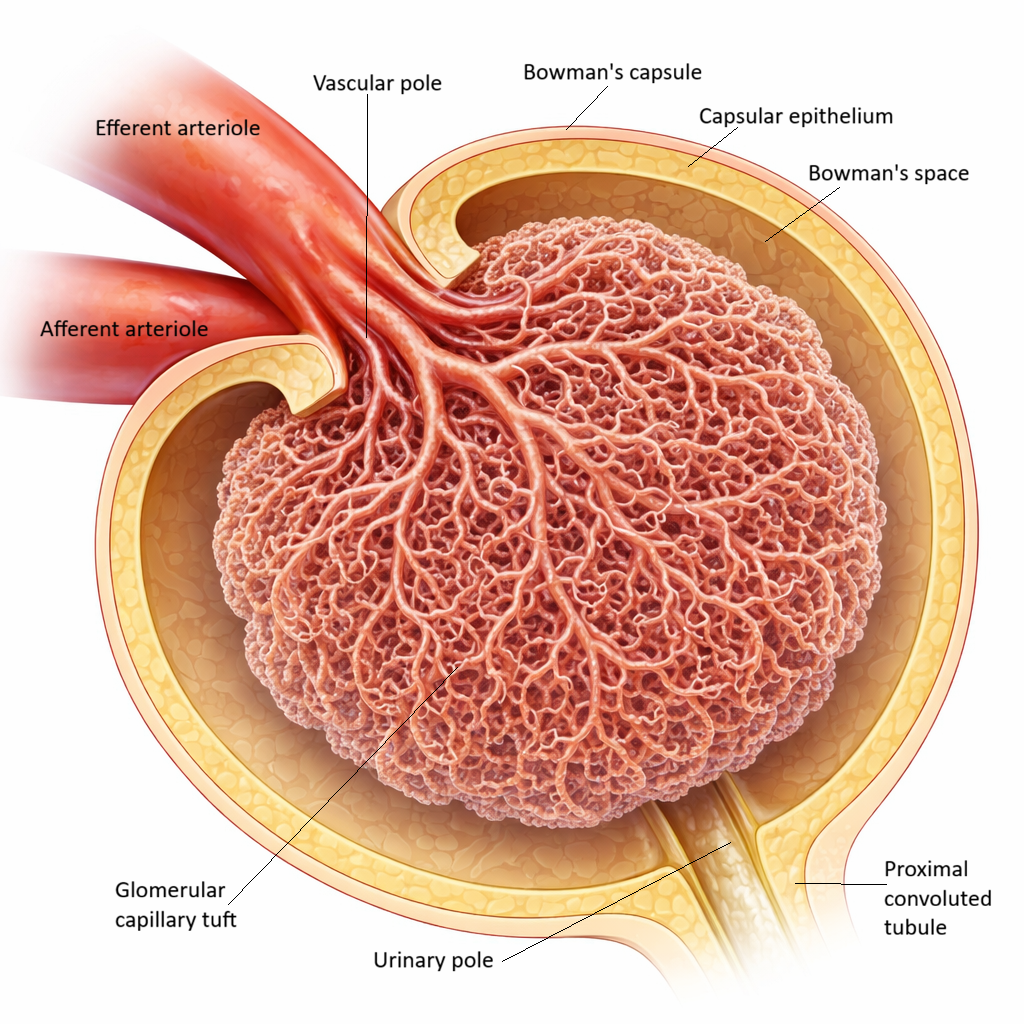
Image: The renal corpuscle contains a tightly coiled network of glomerular capillaries enclosed by Bowman’s capsule. As blood flows from the afferent to the efferent arteriole, hydrostatic pressure drives filtration into Bowman’s space, forming the initial filtrate that enters the proximal convoluted tubule at the urinary pole.
Beyond the Basics
Fenestrated Endothelium
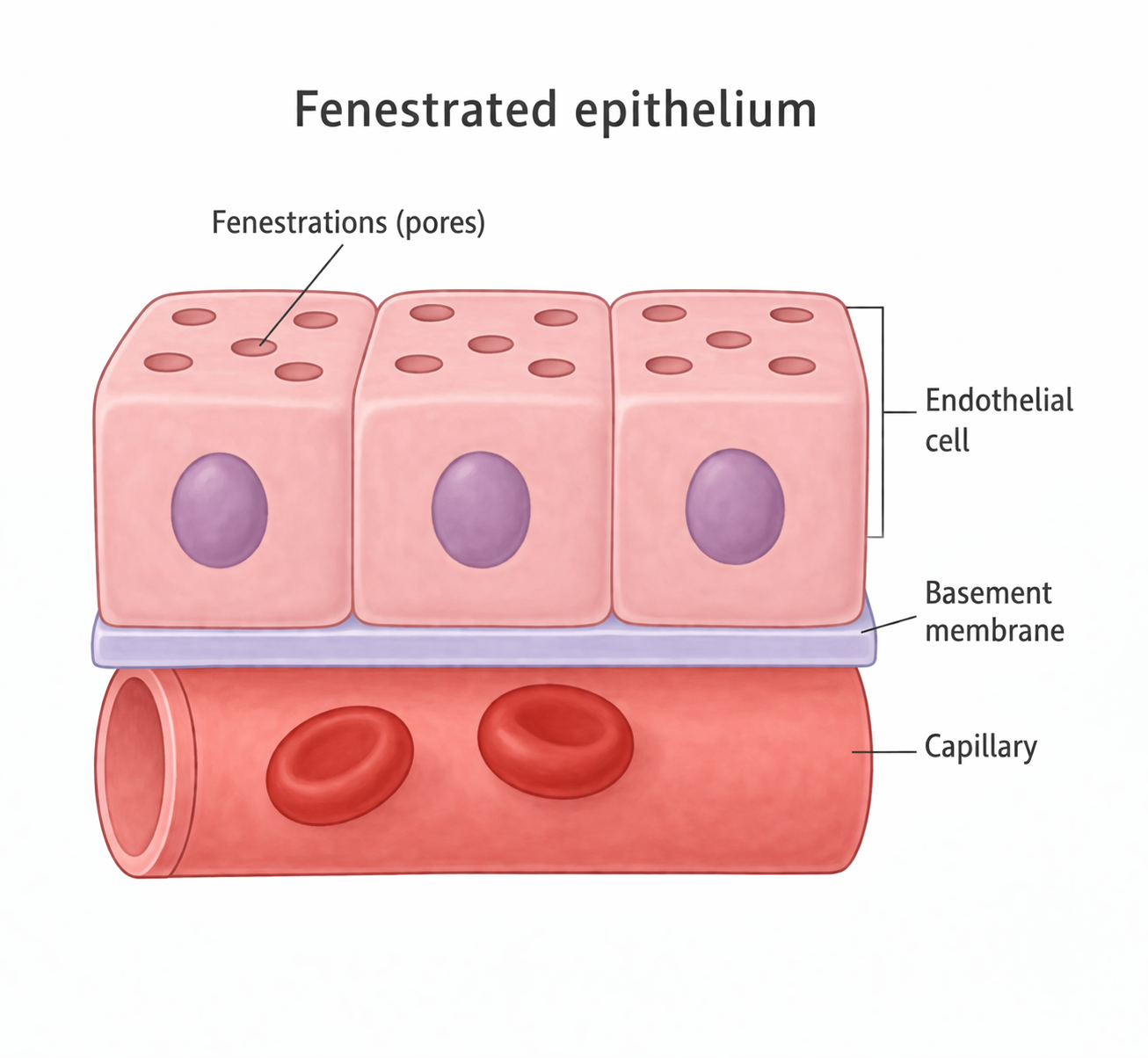
The innermost layer of the filtration barrier is the fenestrated endothelium that lines the glomerular capillaries. These endothelial cells contain large fenestrations (pores in the capillary wall) that allow unhindered passage of plasma water and dissolved solutes. Despite their openness, the fenestrations are small enough to prevent red and white blood cells from escaping into Bowman’s space. The endothelial surface is coated with a glycocalyx (a negatively charged surface layer of proteins and sugars), which adds an additional charge-selective barrier that repels plasma proteins. This surface layer plays an increasingly recognised role in maintaining filtration and is particularly vulnerable to inflammatory injury, sepsis, and diabetic damage.
Glomerular Basement Membrane
The glomerular basement membrane is a thick, trilaminar sheet composed of type IV collagen, laminin, nidogen, and negatively charged heparan sulphate proteoglycans (proteins with sugar chains that carry negative electrical charge). It is synthesised by both podocytes and endothelial cells. Its thickness and molecular structure provide mechanical filtration, while its negative charge prevents the passage of albumin and other anionic proteins. The membrane consists of three layers — the lamina rara interna adjacent to the endothelium, the lamina densa, and the lamina rara externa adjacent to the podocytes (a thin–dense–thin layered filter). Together, these layers form a physical and electrostatic barrier essential for selective filtration. Disorders such as Alport syndrome or diabetic nephropathy alter the structure or charge of this membrane, leading to protein leakage and progressive renal dysfunction.
Podocytes and the Slit Diaphragm
Podocytes are specialised epithelial cells with large cell bodies that extend numerous primary processes, which further branch into slender foot processes. These foot processes interdigitate with those of adjacent podocytes, forming narrow filtration slits bridged by slit diaphragms (molecular “zippers” between foot processes). The slit diaphragm is more than a simple gap; it contains a complex protein network including nephrin, podocin, CD2-associated protein, and other structural molecules that maintain selective permeability. Damage to any of these proteins can result in significant proteinuria, as seen in congenital nephrotic syndromes. Podocytes also regulate filtration through contractile elements within their cytoskeleton, allowing subtle adjustments in slit width. Because podocytes cannot readily proliferate (they cannot easily divide and replace themselves), injury to this layer often leads to permanent structural loss.
Integration of the Three Layers
Although each layer plays a distinct role, the filtration barrier functions as a unified system. The endothelium permits rapid fluid movement, the basement membrane provides physical and charge-based selectivity, and the podocytes refine filtration through the slit diaphragm. Filtration occurs only because the barrier simultaneously offers low resistance to water flow and high resistance to macromolecules. Disruption of any one layer leads to characteristic patterns of protein leakage, which is why understanding this microstructure is essential for interpreting renal pathology.
Clinical Connections
Damage to the glomerular filtration barrier produces predictable clinical patterns depending on which layer is affected. Injury to podocytes and the slit diaphragm primarily disrupts the barrier’s size selectivity, allowing large plasma proteins such as albumin to leak into the urine. This leads to the nephrotic syndrome pattern, characterised by:
heavy proteinuria
hypoalbuminaemia
peripheral and generalised oedema
hyperlipidaemia
Because podocytes cannot regenerate easily, these conditions often progress and can lead to permanent loss of filtration surface.
In contrast, inflammatory injury to the endothelium or basement membrane compromises both the structural and charge-selective components of the barrier. This allows red blood cells and inflammatory mediators to enter the filtrate, producing haematuria and reducing effective filtration. These changes underlie nephritic syndromes, which are typically associated with impaired kidney function, hypertension, and fluid retention rather than massive protein loss.
Systemic diseases frequently target specific components of the filtration barrier. In diabetes mellitus, non-enzymatic glycation thickens and alters the basement membrane, gradually increasing permeability to proteins. In autoimmune diseases such as lupus, immune complexes deposit within or along the membrane, triggering inflammation and capillary wall damage. Hypertension exposes the glomerulus to chronically elevated pressures, accelerating mechanical injury to the capillary wall and podocytes.
Renal biopsy findings reflect these structural disruptions. Effacement of podocyte foot processes, basement membrane thickening, or immune complex deposition can be directly correlated with the patient’s pattern of proteinuria, haematuria, and declining GFR. Understanding how each layer contributes to filtration allows clinicians to link microscopic changes to clinical presentation, disease progression, and treatment response.
Concept Check
How does the negative charge within the glomerular basement membrane contribute to selective filtration
Why is the podocyte slit diaphragm essential for preventing protein loss into Bowman’s space
How do structural changes within the fenestrated endothelium affect filtration of water and solutes
Why does damage to podocytes often lead to permanent structural impairment
How do the three layers of the filtration barrier work together to maintain filtration efficiency and selectivity