Tubular Reabsorption & Secretion: How the Kidney Fine-Tunes the Filtrate
Once blood has been filtered at the glomerulus, the resulting filtrate still contains enormous quantities of water, electrolytes, nutrients, and metabolic by-products. If this filtrate were excreted without modification, the body would rapidly become dehydrated and critically imbalanced. The survival function of the kidneys therefore depends not only on filtration, but on the precise and highly selective processes of tubular reabsorption and secretion. These mechanisms allow the kidneys to conserve essential substances, remove waste and toxins, regulate acid–base balance, control electrolyte concentrations, and maintain blood volume and pressure within narrow limits.
What You Need to Know
Tubular reabsorption and secretion are the processes that allow the kidneys to precisely adjust the composition of the filtrate after it leaves the glomerulus. Reabsorption returns useful substances such as water, electrolytes, glucose, and amino acids back into the bloodstream via the peritubular capillaries and vasa recta, while secretion moves selected substances from the blood into the tubular fluid for elimination. These mechanisms transform a large volume of relatively non-selective filtrate into a small volume of highly regulated urine.
As filtrate flows through the nephron, different segments contribute to reabsorption and secretion in highly specialised ways:
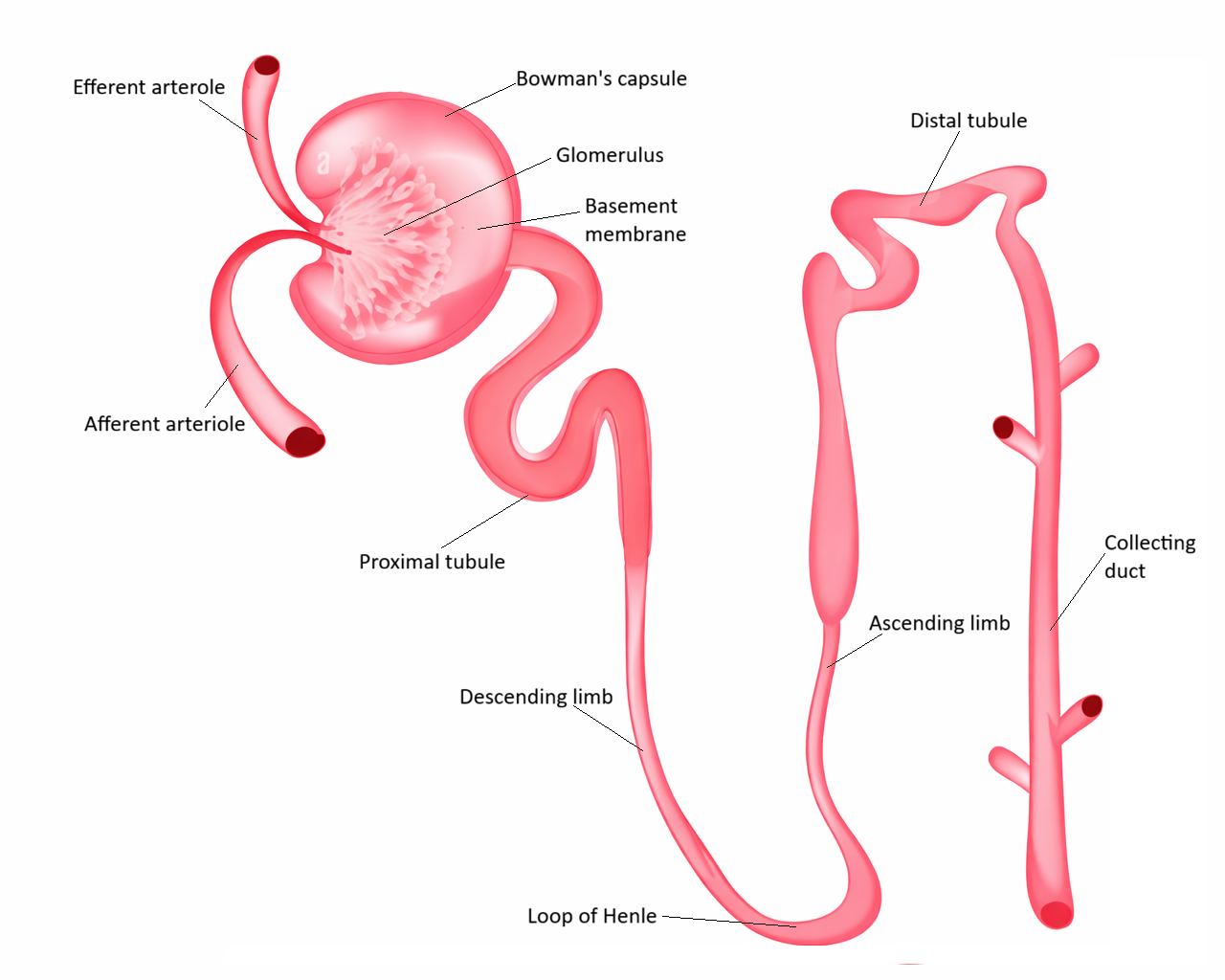
the proximal convoluted tubule reabsorbs the majority of filtered water, sodium, and nutrients and secretes many metabolic wastes and drugs
the loop of Henle modifies water and solute movement to establish the medullary osmotic gradient
the distal convoluted tubule fine-tunes electrolyte and acid–base balance
the collecting ducts determine the final volume and concentration of urine under hormonal control
This segmental organisation allows the kidneys to respond dynamically to changing physiological needs.
Transport across the tubular epithelium occurs through multiple mechanisms depending on the substance being moved. Small non-polar molecules and some ions may diffuse passively down concentration gradients, while others require carrier proteins to facilitate their movement. Many critical solutes, such as sodium and glucose, rely on active transport systems that use cellular energy either directly or indirectly. Through this coordinated network of transport processes, the kidneys can conserve essential substances, eliminate toxins, and maintain the stability of the body’s internal environment.
Beyond the Basics
Proximal Convoluted Tubule: Bulk Recovery of Essential Substances
The proximal convoluted tubule (PCT) is responsible for the majority of reabsorption in the nephron and acts as the kidney’s primary recovery site. Approximately 65–70% of filtered water and sodium, nearly all filtered glucose and amino acids, and large proportions of bicarbonate, phosphate, potassium, and calcium are reclaimed here. This massive transport capacity is made possible by the dense brush border of microvilli (tiny membrane folds that increase surface area) lining the epithelial cells and the large number of mitochondria that provide the energy required for active transport.
Sodium reabsorption is the driving force behind most solute and water movement in the PCT. As sodium enters tubular cells via secondary active transport, substances such as glucose and amino acids are carried along with it. Water then follows passively by osmosis, allowing large volumes of fluid to be reabsorbed while keeping the tubular fluid close to plasma concentration. Bicarbonate reabsorption occurs through a carbonic acid buffering system that is central to maintaining normal blood pH and preventing metabolic acidosis.
The PCT is also a major site of tubular secretion. Hydrogen ions, creatinine, uric acid, organic acids, organic bases, and many drugs are actively transported from the blood into the tubular fluid. This allows the kidney to eliminate substances that are either poorly filtered at the glomerulus or must be tightly regulated for chemical balance.
Loop of Henle: Separation of Water and Solute Handling
Although the loop of Henle does not perform large-scale reabsorption of nutrients, it is essential for separating how water and solutes are handled. In the descending limb, the tubule is highly permeable to water but relatively impermeable to salts. As filtrate moves deeper into the increasingly concentrated medulla, water leaves the tubule by osmosis, making the tubular fluid progressively more concentrated.
In contrast, the ascending limb is impermeable to water but actively reabsorbs sodium, potassium, and chloride into the surrounding tissue. This removes solute without removing water, diluting the tubular fluid while strengthening the corticomedullary osmotic gradient. This separation of water and solute movement is what allows the kidney to later produce either concentrated or dilute urine depending on hydration status.
Distal Convoluted Tubule: Precision Electrolyte Control
The distal convoluted tubule (DCT) is where fine-tuning of electrolyte balance occurs. Sodium reabsorption continues here, but unlike in the PCT, it is regulated by aldosterone, allowing the body to adjust sodium retention in response to blood pressure and volume. Potassium secretion becomes more prominent in this segment, making the DCT a key regulator of plasma potassium concentration.
The DCT is also a major site of calcium regulation. Parathyroid hormone increases calcium reabsorption here, linking kidney function directly to bone metabolism and neuromuscular stability. Hydrogen ion secretion also increases in this segment, allowing more precise control of acid–base balance.
Collecting Ducts: Final Control of Water, Sodium & Acid–Base Balance
The collecting ducts determine the final composition of urine. Sodium reabsorption continues under aldosterone control and is tightly linked to potassium secretion, allowing the kidneys to regulate both extracellular fluid volume and potassium levels with great precision.
Water handling in this segment is almost entirely controlled by antidiuretic hormone (ADH). When ADH is present, aquaporin-2 water channels (membrane pores that allow water to move) are inserted into the tubule wall, permitting water to leave the filtrate and be reabsorbed into the hyperosmotic medulla. When ADH is absent, these channels are removed, and the collecting ducts remain relatively impermeable to water, resulting in the excretion of dilute urine.
Acid–base balance is finalised here through specialised intercalated cells, which either secrete hydrogen ions or bicarbonate depending on the body’s needs. This allows rapid correction of even small disturbances in blood pH.
Forces Driving Reabsorption Into the Blood
Reabsorption from the tubules into the peritubular capillaries is driven by a unique combination of Starling forces. Low hydrostatic pressure in these capillaries, combined with high oncotic pressure created by concentrated plasma proteins, strongly pulls water and solutes out of the interstitium and back into the bloodstream. This ensures efficient recovery of filtered substances without interfering with filtration at the glomerulus.
Tubular Secretion as a Protective Mechanism
Tubular secretion provides an additional layer of chemical protection that filtration alone cannot achieve. Potassium secretion prevents dangerous hyperkalaemia, hydrogen ion secretion prevents acidosis, and drug and toxin secretion ensures removal of substances that could accumulate to harmful levels. Without these active secretion processes, the kidneys would be unable to maintain the precise internal environment required for normal cellular function.
Clinical Connections
Disruption of tubular reabsorption and secretion produces rapid and often severe physiological consequences because these processes are responsible for maintaining the body’s chemical stability. When proximal tubular function is impaired, substances that should be conserved are lost into the urine. Glucose, bicarbonate, phosphate, and amino acids spill into the filtrate, leading to metabolic acidosis, electrolyte imbalance, and progressive nutritional depletion. This pattern is seen in conditions such as proximal renal tubular acidosis and Fanconi syndrome, where the kidney’s ability to reclaim filtered solutes is fundamentally compromised.
Defects in the distal nephron have a different clinical signature. Because this region controls fine electrolyte and acid–base regulation, dysfunction leads to:
abnormal potassium levels, increasing the risk of arrhythmias and muscle weakness
impaired calcium handling, contributing to bone demineralisation
reduced hydrogen ion secretion, leading to acidosis
These abnormalities may appear even when overall filtration is relatively preserved.
Diuretics exploit the segmental organisation of tubular transport. Loop diuretics block sodium reabsorption in the thick ascending limb, disrupting the medullary gradient and producing powerful natriuresis and diuresis. Thiazide diuretics act in the distal convoluted tubule, producing more moderate sodium loss while increasing calcium retention. Potassium-sparing diuretics act in the collecting ducts, either by blocking aldosterone or sodium channels, limiting potassium loss while promoting sodium excretion. These predictable effects arise directly from the transport proteins present in each nephron segment.
In acute kidney injury, failure of tubular transport rapidly leads to life-threatening imbalances. Potassium accumulates, acids are not excreted, fluid cannot be eliminated, and metabolic wastes build up in the blood. Even when some filtration persists, the loss of reabsorptive and secretory function can produce severe hyperkalaemia, acidosis, pulmonary oedema, and uraemia, highlighting how critical tubular processing is to survival.
Concept Check
Why does sodium reabsorption drive most other forms of tubular transport?
Why is water reabsorption hormonally regulated only in the collecting ducts?
How does tubular secretion protect against both hyperkalaemia and metabolic acidosis?
Why do different classes of diuretics act on different nephron segments?
Why does proximal tubular damage cause both metabolic and nutritional disturbances?