Nephron Microanatomy: Structure, Histology & Segment-Specific Function
The nephron is the kidney’s functional unit, responsible for filtering blood, adjusting solute concentrations, regulating fluid balance, and producing urine. Although all nephrons share the same basic structure, each segment has distinct anatomical and histological features that support its specialised functions. Understanding the microanatomy of the nephron is essential for interpreting renal physiology, pharmacology, acid–base processes, and the selective vulnerability of different nephron segments to disease.
What You Need to Know
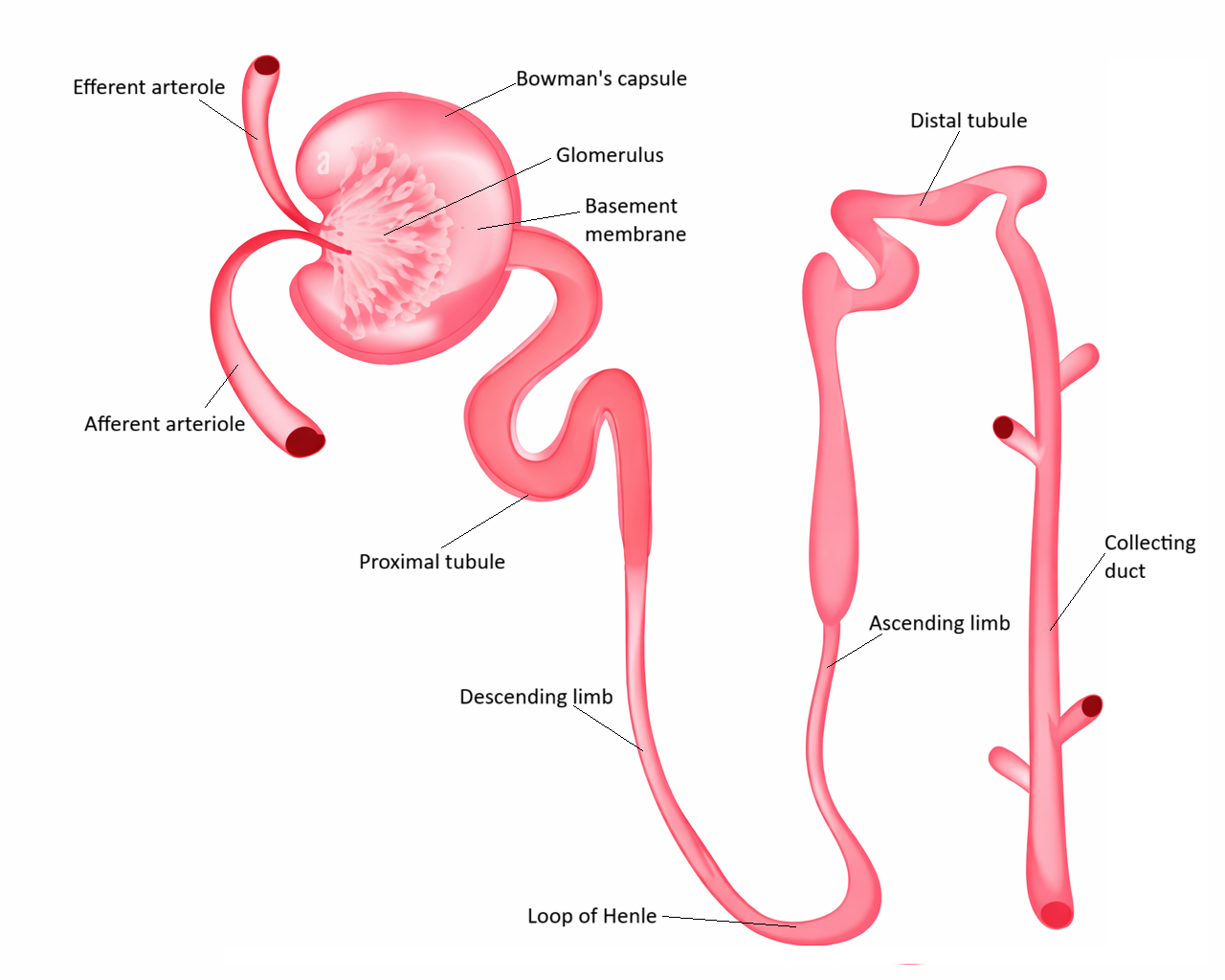
Each kidney contains around one million nephrons, which together form the microscopic filtration network responsible for maintaining the internal chemical environment of the body. Every nephron consists of a renal corpuscle, where blood is filtered, and a renal tubule, where that filtrate is progressively modified into urine. The renal corpuscle is made up of the glomerulus, a specialised capillary bed that allows water and small solutes to pass out of the bloodstream, and Bowman’s capsule, which captures this filtered fluid and directs it into the tubule while retaining cells and large proteins in the circulation.
The renal tubule is a continuous series of highly specialised segments, each with a distinct structural design that determines what it transports and how efficiently it does so. As filtrate moves through the tubule, it passes through:
the proximal convoluted tubule, where most water, electrolytes, glucose, and amino acids are reabsorbed
the loop of Henle, which extends into the medulla and creates the osmotic gradient needed to concentrate urine
the distal convoluted tubule, which fine-tunes electrolyte and acid–base balance
the collecting duct system, which determines the final volume and concentration of urine
The microscopic features of each segment are closely matched to these roles. Proximal tubule cells are packed with mitochondria and lined with microvilli to maximise surface area and energy supply for active transport, while the thin limbs of the loop of Henle allow passive movement of water and solutes down concentration gradients. Cells of the distal tubule and collecting ducts are organised to respond to hormones such as aldosterone and antidiuretic hormone, allowing the kidneys to rapidly adjust fluid balance, blood pressure, and plasma composition in response to the body’s needs.
Image: Nephron simplified for educational purposes.
Beyond the Basics
Renal Corpuscle
The renal corpuscle is located in the cortex and begins the process of urine formation. It consists of the glomerulus, a tuft of fenestrated capillaries, and Bowman’s capsule which encloses it. The filtration barrier is formed by the fenestrated endothelium, a negatively charged basement membrane and podocytes whose interdigitating foot processes create narrow filtration slits. This barrier allows water and small solutes to pass while preventing the movement of proteins and blood cells. Bowman’s capsule has a parietal layer of simple squamous epithelium and a visceral layer composed of podocytes. The space between the two layers collects the filtrate before it enters the proximal tubule. The arrangement of a high-pressure capillary bed enveloped by a collecting capsule allows the kidney to generate large volumes of ultrafiltrate very efficiently.
Proximal Convoluted Tubule
The proximal convoluted tubule is lined with cuboidal epithelial cells that possess a dense brush border of microvilli, greatly increasing the surface area for reabsorption. These cells contain abundant mitochondria to fuel active transport. Their intercellular junctions are relatively permeable, allowing both transcellular and paracellular movement of solutes and water.
The microstructure of the proximal tubule supports its role as the site of bulk reabsorption. Approximately two-thirds of filtered sodium and water, along with nearly all filtered glucose and amino acids, are reclaimed here. Bicarbonate, phosphate and many other solutes are also reabsorbed. Because of its high metabolic demands, this segment is particularly vulnerable to hypoxic injury and toxic damage.
Loop of Henle
The loop of Henle has distinct thin and thick segments, each with specialised histology. The thin descending limb is lined with simple squamous epithelium that is highly permeable to water but only moderately permeable to solutes. As filtrate descends deeper into the hyperosmotic medulla, water leaves the tubule, concentrating the tubular fluid. The thin ascending limb remains squamous but has markedly reduced water permeability; instead, it allows passive movement of sodium and chloride.
The thick ascending limb is lined with cuboidal epithelial cells lacking microvilli. These cells contain transport proteins such as the Na-K-2Cl transporter, which actively reabsorb solutes into the interstitium. The thick ascending limb is impermeable to water, meaning that filtrate becomes progressively more dilute as solutes are removed. Together, these structural differences create the countercurrent multiplier system that allows the kidney to generate a concentrated medullary gradient.
Distal Convoluted Tubule
The distal convoluted tubule contains low cuboidal epithelial cells with only a sparse border of microvilli. These cells have fewer mitochondria than the proximal tubule and form tighter junctions, allowing more precise control over solute movement. Calcium reabsorption in this segment is regulated by parathyroid hormone, while sodium handling is influenced by aldosterone. The microanatomy of the distal tubule supports fine-tuning of solute composition, in contrast with the bulk processing of earlier segments.
Juxtaglomerular Apparatus
Although it has its own dedicated functional post, a structural description is useful here. Where the distal tubule returns to contact the afferent and efferent arterioles of its own glomerulus, the epithelium becomes taller and more densely packed, forming the macula densa. These cells sense the sodium chloride concentration of tubular fluid. Adjacent to them are juxtaglomerular cells, modified smooth muscle cells in the afferent arteriole wall that store and release renin. Extraglomerular mesangial cells fill the space between these structures. This anatomical arrangement enables local regulation of filtration rate through tubuloglomerular feedback.
Collecting Duct System
The collecting ducts contain two main cell types. Principal cells are simple cuboidal cells that respond to aldosterone and antidiuretic hormone. They adjust sodium reabsorption and water permeability through regulated expression of membrane channels such as ENaC and aquaporins. Intercalated cells are fewer in number and contain abundant mitochondria. They are responsible for acid–base regulation through hydrogen ion secretion or bicarbonate generation. The collecting ducts therefore determine the final concentration and volume of urine.
Juxtamedullary and Cortical Nephrons
Nephrons differ in length and depth according to their location. Cortical nephrons sit high in the cortex and have short loops of Henle; they are primarily responsible for filtration and reabsorption. Juxtamedullary nephrons originate near the corticomedullary junction and possess long loops that extend deep into the medulla. Their structure allows the creation and maintenance of the osmotic gradient required for concentrated urine. The vasa recta surrounding these long loops participate in countercurrent exchange, preserving medullary osmolarity.
Clinical Connections
The unique microanatomy of each nephron segment determines both its physiological role and its vulnerability to disease. Because different segments rely on distinct transporters, energy demands, and permeability properties, injury or dysfunction tends to follow predictable anatomical patterns. The proximal tubule, for example, has extremely high metabolic activity due to its dense mitochondria and active transport processes. This makes it particularly susceptible to hypoxic and toxic injury, which is why acute tubular necrosis most often affects this segment, leading to impaired reabsorption and the appearance of solutes in the urine that would normally be reclaimed.
The pharmacology of diuretics is also tightly linked to nephron microanatomy. Each class targets a specific segment based on the transport proteins expressed there:
Loop diuretics inhibit the Na–K–2Cl transporter in the thick ascending limb, disrupting the medullary gradient and causing profound sodium and water loss
Thiazide diuretics act on the Na–Cl cotransporter in the distal convoluted tubule, producing a more moderate diuretic effect while increasing calcium reabsorption
Potassium-sparing diuretics act in the collecting duct, either by blocking aldosterone or by inhibiting sodium channels in principal cells
These site-specific actions explain why different diuretics produce different electrolyte disturbances and levels of diuresis.
Inherited tubular disorders can also be understood by linking disease to nephron structure. In Bartter syndrome, defects in transporters of the thick ascending limb mimic the effect of loop diuretics, leading to salt wasting, hypokalaemia, and metabolic alkalosis. Gitelman syndrome affects transporters in the distal convoluted tubule and produces a thiazide-like pattern of electrolyte loss. In contrast, Liddle syndrome involves overactive sodium channels in the collecting duct, causing excessive sodium reabsorption, volume expansion, and hypertension despite low aldosterone levels. These patterns make sense only when the microanatomy and transporter distribution of the nephron are understood.
Hormonal disorders are also related to nephron structure. Because antidiuretic hormone acts on aquaporins in the collecting duct, defects in this segment lead to disorders such as nephrogenic diabetes insipidus, where water cannot be conserved despite dehydration. Aldosterone resistance or excess alters sodium and potassium handling in principal cells, producing characteristic blood pressure and electrolyte changes.
Concept Check
Why does the proximal tubule have extensive microvilli while the distal tubule does not?
How does the structure of the thick ascending limb contribute to the formation of the medullary osmotic gradient?
What anatomical differences distinguish juxtamedullary nephrons from cortical nephrons?
Why are principal cells uniquely responsive to antidiuretic hormone?
How does the arrangement of the renal corpuscle ensure selective filtration?