Kidney Gross Anatomy: Structure, Organisation & Vascular Architecture
The kidneys are highly specialised organs whose structure is precisely organised to support filtration, reabsorption, endocrine activity, and fluid–electrolyte regulation. Their anatomical design reflects the complexity of these functions, from the layered arrangement of the cortex and medulla to the intricate vascular network that maintains high-pressure filtration and low-pressure reabsorption simultaneously. Understanding the gross anatomy of the kidneys provides the foundation for studying nephron physiology, interpreting renal pathology, and recognising how systemic disease affects renal function.
What You Need to Know
Each kidney is a bean-shaped, retroperitoneal organ located on either side of the vertebral column, extending roughly from T12 to L3. The right kidney typically sits slightly lower than the left due to the position of the liver. A tough fibrous capsule, a cushioning layer of perirenal fat, and the renal fascia anchor the kidneys securely within the posterior abdominal cavity while protecting them from mechanical stress and movement during respiration and posture changes. Their position outside the peritoneal cavity also helps isolate them from fluctuations in intra-abdominal pressure, supporting stable blood flow and filtration.
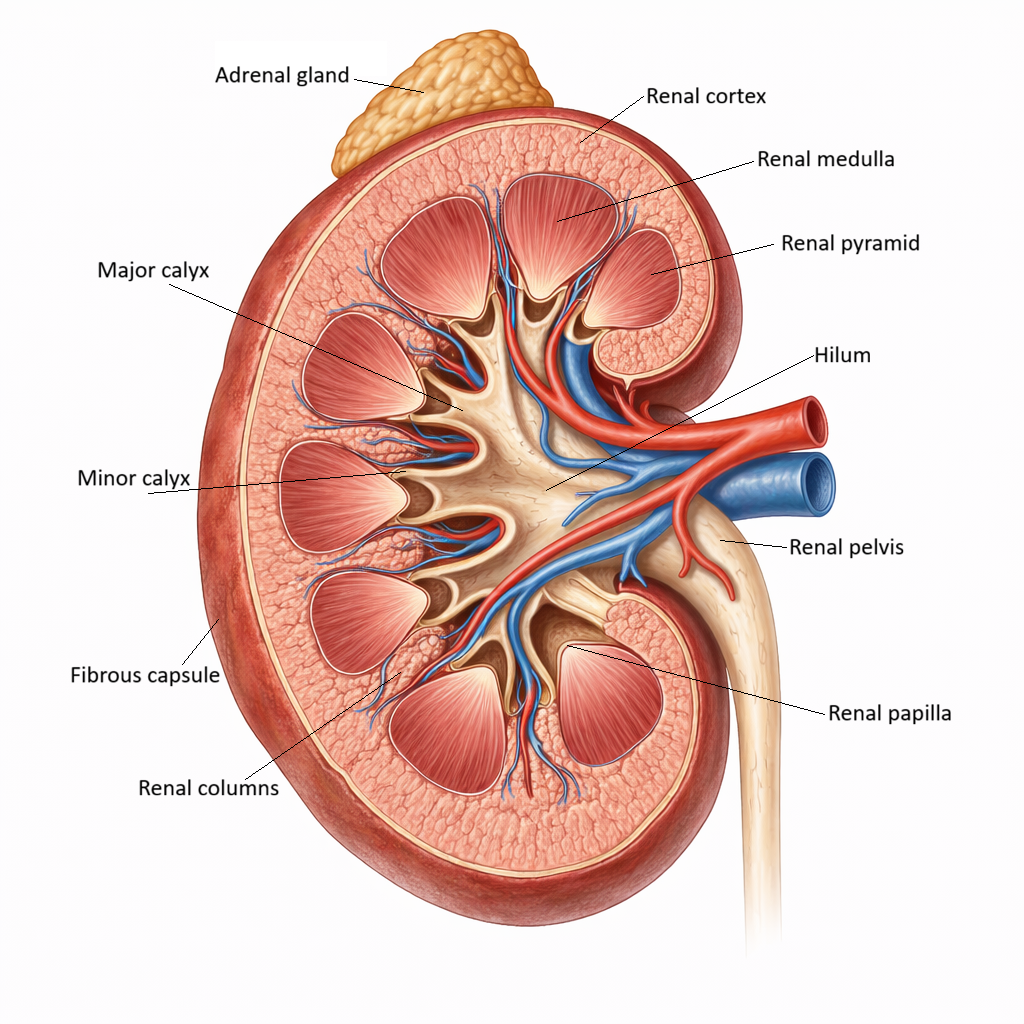
Internally, the kidney is organised into an outer cortex and an inner medulla. The cortex contains the majority of the glomeruli and convoluted tubules, where blood is filtered and the initial processing of filtrate occurs. The medulla is arranged into pyramid-shaped structures that contain loops of Henle and collecting ducts, which concentrate urine and direct it toward the renal pelvis. The tips of these pyramids, called papillae, empty into minor calyces that merge into major calyces before forming the renal pelvis, creating a continuous drainage system from nephron to ureter.
The renal hilum on the medial surface acts as the gateway of the kidney, allowing the renal artery to deliver a large and constant blood supply, while the renal vein returns filtered blood to the circulation. The ureter carries urine away from the kidney to the bladder, and lymphatic vessels and nerves enter and exit through the same region. This highly ordered structural arrangement ensures that filtration, reabsorption, secretion, and urine drainage occur efficiently and without interruption, allowing the kidneys to continuously regulate fluid balance, electrolytes, and waste removal.
Key structures at a glance
Fibrous capsule, perirenal fat, and renal fascia for protection and stability
Cortex for filtration and early tubular processing
Medulla for urine concentration and flow toward the pelvis
Renal pelvis and calyces for urine collection and drainage
Renal hilum as the entry and exit point for vessels, nerves, and ureter
Image: Key anatomical structures of the kidney.
Beyond the Basics
External Structure & Supporting Tissues
The outermost covering of the kidney, the renal capsule, is a tough yet flexible connective tissue sheath that tightly adheres to the kidney’s surface. This capsule forms a physical barrier against trauma and limits the spread of infection, helping preserve the integrity of the delicate internal structures. Beneath the capsule lies the perirenal (perinephric) fat, a thick, cushioning adipose layer that absorbs mechanical shock and stabilises the kidney during body movement, breathing, and postural changes.
Both the kidney and its surrounding fat are enclosed by the renal fascia, a dense fibrous connective tissue layer that anchors the organ to surrounding structures such as the posterior abdominal wall and diaphragm. This anchoring prevents excessive movement or twisting of the renal vessels and ureter, which could compromise blood flow or urine drainage. Outside the renal fascia sits the pararenal fat, which provides an additional layer of padding and further insulation. Together, these layers create a protective compartment that preserves kidney position, maintains internal pressure relationships, and supports uninterrupted filtration.
Internal Structure: Cortex, Medulla, Pyramids & Columns
The internal anatomy of the kidney is organised into two distinct regions: the renal cortex and the renal medulla, each specialised for different stages of urine formation. The cortex forms the outer layer and contains the renal corpuscles (glomeruli and Bowman’s capsules) as well as the proximal and distal convoluted tubules. This dense concentration of microscopic filtration and processing units gives the cortex its granular appearance and reflects its role as the primary site of blood filtration and early tubular modification of filtrate.
The medulla lies deep to the cortex and is arranged into 8–18 cone-shaped renal pyramids, which are made up of loops of Henle and collecting ducts running in parallel. These structures generate the osmotic gradients required to concentrate urine and regulate water and electrolyte balance. The apex of each pyramid forms a renal papilla, where multiple collecting ducts converge and deliver urine into a minor calyx. Between the pyramids, extensions of cortical tissue called renal columns provide structural support and house blood vessels that supply the medulla. This highly ordered architecture ensures that filtrate produced in the cortex can move efficiently through the medulla while being progressively modified into urine.
Calyces, Renal Pelvis & Ureter
Urine leaving the collecting ducts enters the papilla and drains into a funnel-shaped minor calyx. Several minor calyces join to form a major calyx, and multiple major calyces merge into the renal pelvis, a broad collecting chamber that marks the beginning of the urinary outflow tract. This branching system allows urine from thousands of nephrons to be gathered in an organised and low-pressure manner.
The renal pelvis narrows to form the ureter, a muscular tube that propels urine toward the bladder using rhythmic peristaltic contractions. These contractions occur independently of gravity, ensuring continuous drainage regardless of body position. The smooth, tapered design of the pelvis and ureter minimises resistance to flow and reduces the risk of urine stagnation, helping maintain sterility and preventing backflow.
Renal Hilum & Vascular Organisation
The renal hilum is the recessed medial entry point into the kidney and serves as the gateway for all structures entering or leaving the organ. From anterior to posterior, it contains the renal vein, renal artery, and renal pelvis, an important spatial relationship used in both imaging and surgery. This orderly arrangement ensures that blood, urine, lymphatics, and nerves can pass through the hilum without compression or obstruction.
Once inside the hilum, these structures branch into the renal sinus, a cavity that houses the major calyces, renal pelvis, and the main renal vessels before they distribute throughout the kidney. This centralised access point allows the kidney to maintain its compact shape while accommodating a large volume of blood flow and continuous urine output.
Renal Blood Supply: A High-Flow, High-Pressure System
Despite their small size, the kidneys receive approximately 20–25% of cardiac output, reflecting their critical role in filtering plasma and regulating internal homeostasis. This high-flow system allows enormous volumes of blood to pass through the kidneys each day, enabling rapid removal of waste products and fine-tuned control of fluid, electrolytes, and acid–base balance.
Blood enters through the renal artery and undergoes a highly ordered branching pattern: segmental arteries divide into interlobar arteries that run between renal pyramids, then into arcuate arteries that arch over the pyramid bases, and finally into interlobular arteries that penetrate the cortex. From these vessels, afferent arterioles deliver blood to the glomeruli, where filtration occurs. Efferent arterioles then exit the glomeruli and form either the peritubular capillaries, which support reabsorption and secretion in the cortex, or the vasa recta, which maintain the medullary osmotic gradient.
This arrangement of two capillary beds in series is unique to the kidneys and allows them to perform two contrasting tasks simultaneously: filtering large volumes of plasma while selectively reclaiming what the body needs. The first capillary bed supports filtration under high pressure, while the second supports reabsorption under low pressure, making precise chemical control possible.
Innervation: Sympathetic Regulation of Renal Blood Flow
The kidneys are richly supplied by sympathetic nerves that provide powerful regulatory control over renal function. These fibres innervate the renal blood vessels, juxtaglomerular cells, and renal tubules, allowing the nervous system to adjust kidney activity in response to physiological stress. By altering the diameter of afferent and efferent arterioles, sympathetic input can rapidly change glomerular filtration rate and renal blood flow.
Sympathetic stimulation also promotes the release of renin, activating the renin–angiotensin–aldosterone system, and increases sodium reabsorption in the proximal tubule. During situations such as haemorrhage, dehydration, or acute stress, this neural control helps preserve circulating blood volume and blood pressure by reducing urine output, although prolonged activation can compromise renal perfusion.
Functional Integration with Nephron Architecture
The kidney’s gross anatomical design is tightly integrated with nephron organisation to support efficient filtration and urine concentration. Cortical nephrons, which make up the majority of nephrons, are optimised for filtration and solute reabsorption and remain mostly within the cortex. Juxtamedullary nephrons, however, extend deep into the medulla and play a critical role in establishing the steep osmotic gradient required to concentrate urine.
The parallel arrangement of loops of Henle, collecting ducts, and vasa recta within the renal pyramids enables countercurrent exchange, allowing water and solutes to be precisely regulated. Meanwhile, the dense vascular network of the cortex ensures rapid plasma delivery to glomeruli and efficient transport of reabsorbed substances back into the circulation. Together, this structural and functional integration allows the kidneys to continuously maintain internal chemical stability despite wide fluctuations in fluid intake, blood pressure, and metabolic demand.
Clinical Connections
Obstruction anywhere along the collecting system causes pressure to build upstream. In hydronephrosis, urine accumulates in the renal pelvis and calyces, progressively dilating these structures and compressing the renal medulla and cortex. As this pressure rises, it reduces glomerular filtration by opposing blood flow through the glomerular capillaries and can eventually lead to nephron loss if prolonged. Imaging often reveals ballooning of the calyces and pelvis, reflecting this anatomical back-pressure effect.
Vascular anatomy is equally important in understanding renal disease. Renal artery stenosis reduces perfusion pressure to the glomeruli, triggering the juxtaglomerular apparatus to release renin and activate the renin–angiotensin–aldosterone system (RAAS). This leads to:
Systemic vasoconstriction
Sodium and water retention
Progressive hypertension and further renal injury
Over time, reduced blood supply also causes ischaemic damage to nephrons, contributing to chronic kidney disease.
The renal capsule plays a critical role during inflammation, trauma, or obstruction. Because it is tough and non-elastic, any swelling within the kidney rapidly increases internal pressure. This can:
Compress intrarenal blood vessels
Reduce glomerular filtration
Worsen ischaemia and tissue injury
This is why acute infections, haemorrhage, or obstruction can lead to rapid deterioration in kidney function even when the underlying lesion is relatively small.
Finally, the organisation of the cortex, medulla, and papillae explains characteristic patterns seen in disease. Infections such as pyelonephritis often track along the pyramids toward the papillae, while papillary necrosis tends to occur where blood supply is most vulnerable. Recognising these anatomical relationships helps clinicians link imaging, laboratory findings, and clinical presentation into a coherent picture of renal pathology.
Concept Check
How does the organisation of the cortex and medulla support nephron function?
Why does the kidney receive such a large percentage of cardiac output?
How does the structure of the renal hilum influence the arrangement of vessels?
Why are juxtamedullary nephrons essential for concentrating urine?
How does renal sympathetic innervation affect renal blood flow during stress?