Polycystic Ovary Syndrome (PCOS)
Polycystic ovary syndrome is a complex endocrine condition characterised by chronic ovulatory dysfunction, hyperandrogenism, and metabolic dysregulation. Although commonly identified through menstrual irregularity or ovarian morphology, PCOS is a systemic disorder involving hormonal, metabolic, and inflammatory pathways. In PCOS, menstrual disturbance, infertility, acne, and metabolic disease frequently coexist, which is why management requires more than cycle regulation alone.
What You Need to Know
Polycystic ovary syndrome develops when the normal signalling between the hypothalamus, pituitary gland and ovaries becomes dysregulated. In a typical cycle, coordinated release of gonadotropins supports follicular maturation and ovulation. In PCOS, this balance is altered, favouring increased luteinising hormone activity and excessive ovarian androgen production. Elevated androgens interfere with normal follicle development, so follicles arrest in early stages rather than progressing to ovulation, leading to irregular or absent ovulatory cycles.
PCOS is not confined to the ovaries. Neuroendocrine disruption interacts closely with insulin resistance, which amplifies androgen production and reduces hepatic clearance of circulating androgens. Hyperinsulinaemia also alters ovarian responsiveness to gonadotropins, further impairing follicular maturation. These processes reinforce one another, creating a stable but abnormal hormonal environment that persists over time rather than fluctuating from cycle to cycle.
Several interconnected mechanisms explain the characteristic features of PCOS:
Increased androgen production disrupts follicle development and ovulation
Altered hypothalamic–pituitary signalling favours luteinising hormone dominance
Insulin resistance amplifies androgen excess and sustains hormonal imbalance
The result is a chronic endocrine state rather than a transient reproductive disturbance. Ovulatory dysfunction underlies infertility and menstrual irregularity, while prolonged androgen excess and insulin resistance contribute to long-term metabolic consequences. PCOS therefore represents a systemic disorder of hormonal regulation with reproductive, metabolic and cardiovascular implications that extend well beyond the ovaries themselves.
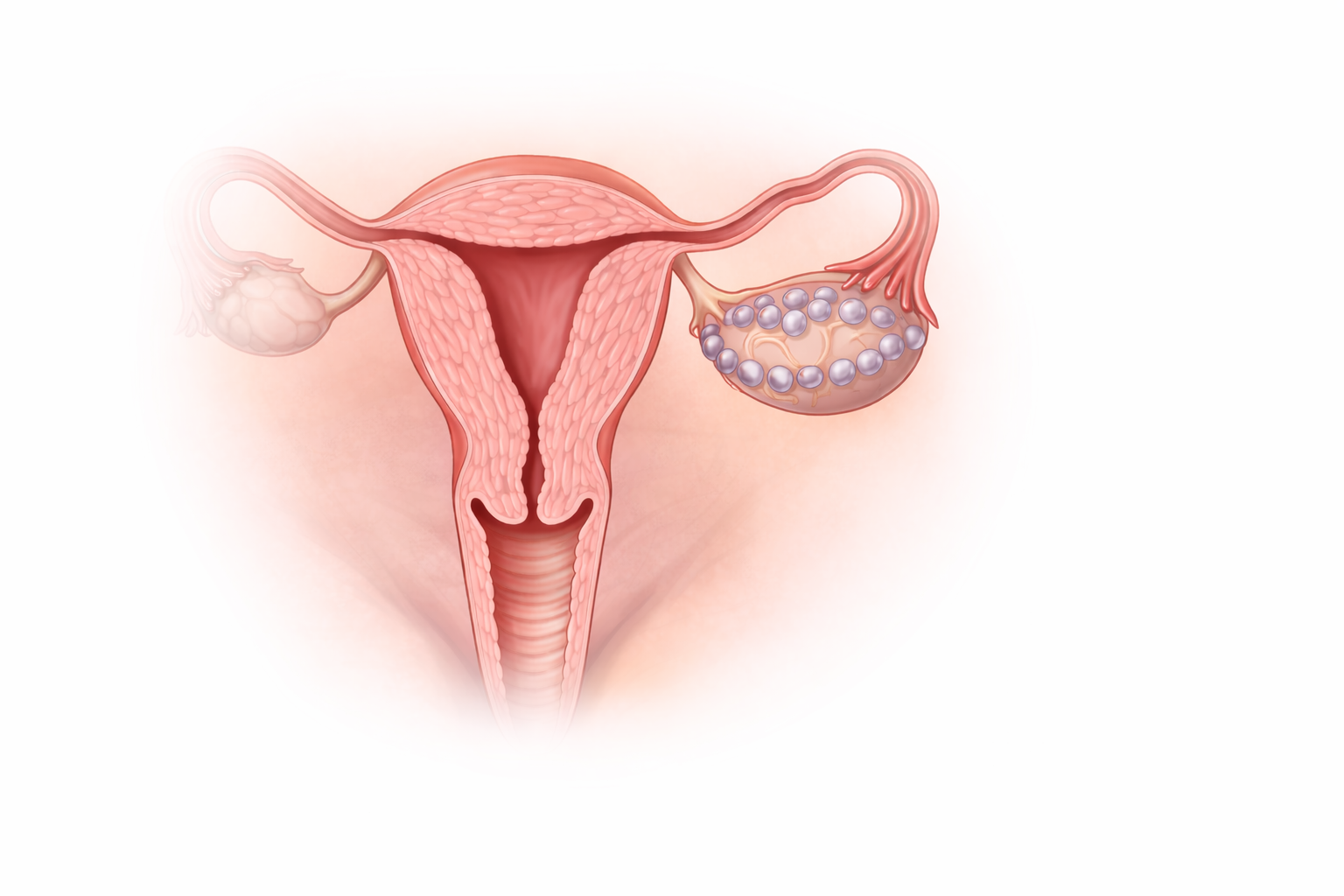
Image: Polycystic ovary syndrome is characterised by multiple small, immature follicles arranged peripherally within the ovary. These follicles represent arrested development rather than true cysts, reflecting disrupted ovulation and underlying hormonal imbalance.
Beyond the Basics
Hypothalamic–pituitary dysregulation
In PCOS, normal pulsatile communication between the hypothalamus and pituitary is altered, leading to a shift in gonadotropin secretion. Luteinising hormone release becomes relatively dominant, while follicle-stimulating hormone levels are insufficient to support coordinated follicular maturation. This imbalance disrupts the finely timed sequence required for selection of a dominant follicle.
Increased luteinising hormone preferentially stimulates ovarian theca cells to produce androgens, while reduced follicle-stimulating hormone activity limits granulosa cell function, including oestrogen production and follicle support. The result is arrested follicular development and failure of ovulation, establishing chronic anovulation as a core feature of the condition.
Hyperandrogenism and ovarian dysfunction
Excess androgen production is central to the pathophysiology of PCOS and drives both reproductive and clinical manifestations. Elevated androgens interfere with follicular maturation, preventing progression beyond early developmental stages. Instead of a single follicle reaching ovulation, multiple small follicles accumulate within the ovary. This produces the characteristic polycystic ovarian morphology seen on imaging, which reflects ongoing follicular arrest rather than cyst formation or primary ovarian disease. Systemically, androgen excess contributes to hirsutism, acne and scalp hair thinning, linking ovarian dysfunction directly to visible clinical features.
Insulin resistance and metabolic amplification
Insulin resistance plays a pivotal role in sustaining hormonal imbalance in PCOS and occurs across a wide range of body compositions. Compensatory hyperinsulinaemia acts directly on the ovary to enhance androgen production and simultaneously suppresses hepatic synthesis of sex hormone-binding globulin. Reduced binding capacity increases the proportion of free, biologically active androgens in circulation, intensifying their effects on target tissues. This metabolic–endocrine interaction explains why insulin resistance worsens reproductive dysfunction and why PCOS is strongly associated with type 2 diabetes, dyslipidaemia and increased cardiovascular risk over time.
Anovulation and endometrial instability
Chronic anovulation leads to prolonged exposure of the endometrium to oestrogen without the cyclical progesterone influence that normally follows ovulation. Without progesterone-mediated stabilisation and shedding, the endometrium undergoes continuous proliferation. This produces irregular, unpredictable bleeding patterns and increases the risk of endometrial hyperplasia. Endometrial instability therefore represents a downstream consequence of ovulatory failure rather than an isolated uterine disorder, highlighting the importance of hormonal regulation even when pregnancy is not desired.
Inflammation and adipose tissue contribution
Low-grade chronic inflammation is increasingly recognised as an integral component of PCOS. Adipose tissue, particularly visceral fat, releases inflammatory cytokines that worsen insulin resistance and interfere with endocrine signalling. This inflammatory environment amplifies metabolic dysfunction and further destabilises reproductive hormone regulation. Fatigue and reduced well-being commonly reported in PCOS reflect these systemic effects rather than ovarian pathology alone, reinforcing the condition’s multisystem nature.
Lifelong progression and variability
PCOS is heterogeneous in both presentation and progression. Menstrual irregularity and hyperandrogenic features often dominate earlier life, while metabolic complications such as insulin resistance, dyslipidaemia and cardiovascular risk become more prominent with age. This evolving pattern reflects cumulative exposure to hormonal imbalance, metabolic stress and inflammation rather than static ovarian abnormalities. PCOS is therefore best understood as a lifelong endocrine and metabolic disorder with variable expression, rather than a fixed reproductive condition confined to the ovaries.
Clinical Connections
PCOS most often presents with menstrual irregularity or amenorrhoea, infertility related to chronic anovulation, and features of androgen excess such as acne, hirsutism and scalp hair thinning. Weight gain and central adiposity are common but not universal and reflect underlying insulin resistance rather than lifestyle factors alone. Psychological distress, including anxiety, low mood and reduced self-esteem, is frequent and arises from a combination of hormonal effects, visible symptoms, reproductive uncertainty and the burden of managing a long-term condition that affects multiple aspects of health.
Several clinical features tend to cluster because they share the same endocrine and metabolic drivers:
Irregular or absent ovulation leading to infertility and unpredictable bleeding
Hyperandrogenic features affecting skin and hair due to excess circulating androgens
Metabolic disturbance, including insulin resistance and weight gain, contributing to fatigue and long-term cardiometabolic risk
Management targets these shared mechanisms rather than treating symptoms in isolation. Restoring more regular ovulation improves fertility and stabilises endometrial exposure, while reducing androgen activity alleviates cutaneous symptoms and improves quality of life. Improving insulin sensitivity is central, as hyperinsulinaemia amplifies ovarian androgen production and sustains hormonal imbalance. Protection of the endometrium is essential in those with ongoing anovulation to reduce the risk of hyperplasia and abnormal bleeding.
Effective care therefore integrates reproductive, metabolic and psychological priorities. Addressing insulin resistance reduces long-term risk of type 2 diabetes and cardiovascular disease while also improving ovulatory function and hormonal stability. Interventions are selected based on dominant clinical concerns, recognising that improvements in metabolic health often translate into broader reproductive and endocrine benefit rather than acting in isolation.
Concept Check
How does altered gonadotropin release disrupt ovulation in PCOS?
Why does insulin resistance worsen hyperandrogenism?
Why are polycystic ovaries a consequence rather than the cause of PCOS?
How does chronic anovulation increase endometrial risk?
Why is PCOS considered a lifelong metabolic as well as reproductive disorder?