TESTICULAR STRUCTURE & SPERMATOGENESIS
The testes are the primary male reproductive organs responsible for producing sperm and synthesising testosterone. Their structure is uniquely adapted to support continuous sperm production throughout adult life, a process that requires precise temperature regulation, hormonal control and a specialised microenvironment within the seminiferous tubules. Spermatogenesis is a complex, multi-stage developmental pathway that transforms diploid germ cells into highly specialised, motile sperm capable of fertilising an ovum.
What You Need to Know
The testes are paired reproductive and endocrine organs specialised for sperm production and testosterone secretion. Each testis is densely packed with seminiferous tubules, which form the functional site of spermatogenesis, and an interstitial compartment containing Leydig cells. Leydig cells respond to luteinising hormone by producing testosterone, which is essential for sperm development, male secondary sexual characteristics, and maintenance of reproductive function. Blood vessels, lymphatics, and connective tissue within the interstitium support this high metabolic activity.
Within the seminiferous tubules, developing germ cells are arranged in an organised sequence from the basement membrane toward the lumen, reflecting progressive stages of maturation. Sertoli cells span the full thickness of the tubule and provide structural support, metabolic nourishment, and regulatory signals to developing sperm. Tight junctions between adjacent Sertoli cells form the blood–testis barrier, a specialised boundary that protects developing germ cells from immune recognition while creating a controlled microenvironment for meiosis and differentiation.
Spermatogenesis is a continuous, highly regulated process that begins at puberty and continues throughout adult life. It proceeds through three broad phases that transform undifferentiated germ cells into motile spermatozoa capable of fertilisation:
Proliferation of spermatogonia, where diploid germ cells divide by mitosis to maintain the stem cell pool and generate cells committed to differentiation
Meiosis, producing haploid spermatids with half the genetic material required for fertilisation
Spermiogenesis, during which spermatids undergo structural remodelling to form mature spermatozoa with a head, midpiece, and tail
This entire process takes approximately 64 to 74 days in humans and depends on coordinated hormonal support. Follicle-stimulating hormone acts primarily on Sertoli cells, while testosterone produced by Leydig cells maintains high intratesticular androgen levels essential for normal sperm development. The slow, continuous nature of spermatogenesis explains why changes in fertility reflect events that occurred weeks earlier and highlights the sensitivity of sperm production to hormonal, thermal, and systemic influences.
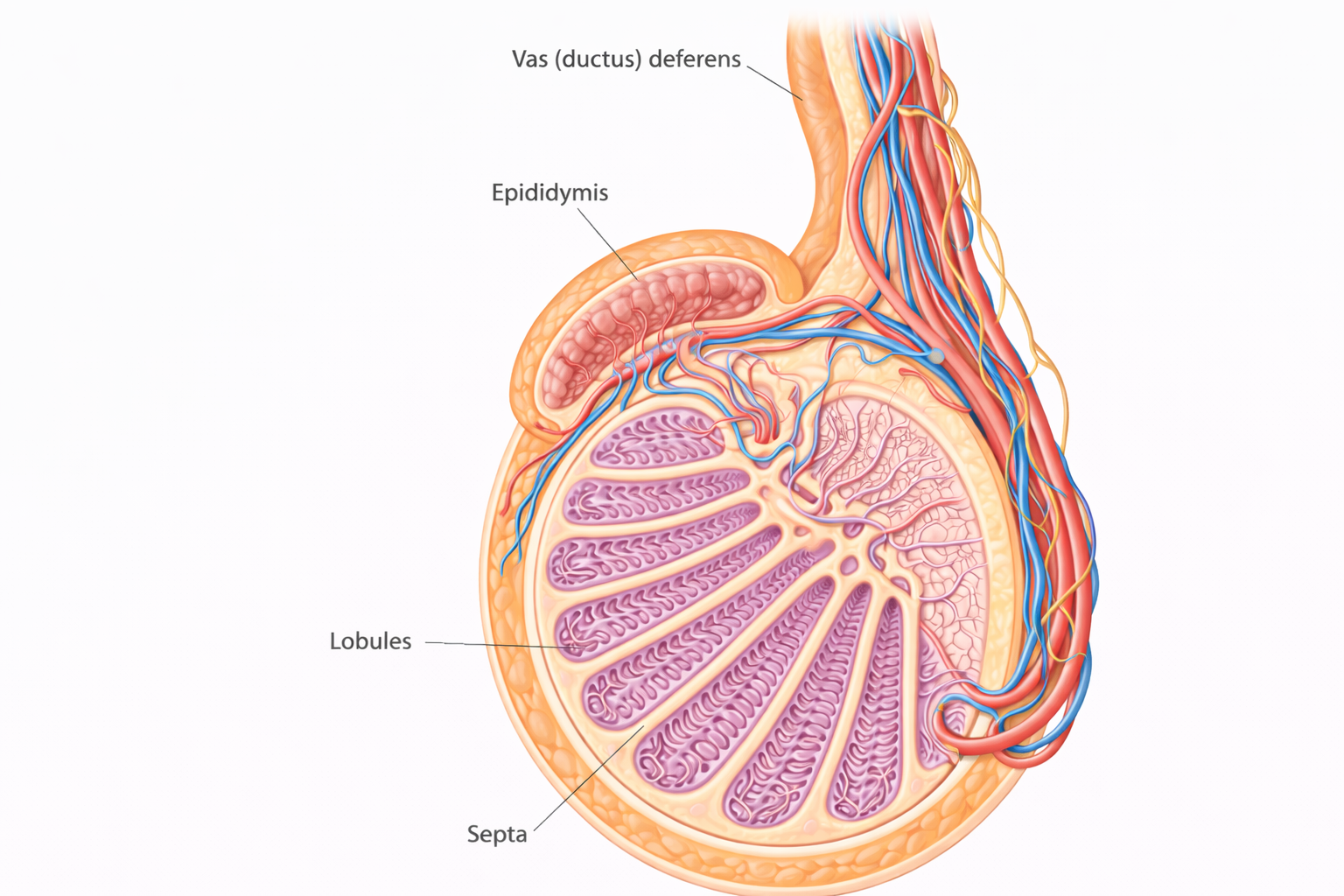
Image: The testis is organised into lobules separated by connective tissue septa, each containing coiled seminiferous tubules where sperm are produced. These tubules converge toward the central rete testis, with sperm then passing through efferent ductules into the epididymis for maturation, before continuing into the vas deferens.
Beyond the Basics
Gross Anatomy and Temperature Regulation
The testes are housed within the scrotum, an external structure that allows precise thermal regulation essential for sperm production. Optimal spermatogenesis requires temperatures approximately 2 to 4°C below core body temperature, and even small deviations can impair germ cell development. This temperature control is achieved through coordinated anatomical and physiological mechanisms rather than passive cooling alone.
The cremaster muscle elevates or lowers the testes in response to temperature and arousal, while the dartos muscle adjusts scrotal surface area by wrinkling or relaxing the skin to alter heat loss. Within the spermatic cord, the pampiniform plexus acts as a counter-current heat exchange system, cooling arterial blood before it reaches the testis by transferring heat to cooler venous blood returning from the scrotum. Disruption of these mechanisms, such as undescended testes, prolonged heat exposure, or external compression, compromises spermatogenesis and may have lasting effects on fertility.
Seminiferous Tubules and the Blood–Testis Barrier
The seminiferous tubules are the functional units of the testis and provide a highly organised environment for sperm development. Their walls are composed of Sertoli cells interspersed with germ cells at successive stages of maturation, arranged from the basement membrane toward the lumen. This spatial organisation reflects the progression of spermatogenesis and allows efficient coordination of cellular development.
Sertoli cells form the blood–testis barrier through tight junctions that divide the tubule into basal and adluminal compartments. This barrier isolates developing germ cells from the systemic circulation, protecting them from immune recognition and creating a specialised biochemical environment for meiosis and differentiation. Beyond structural support, Sertoli cells actively regulate spermatogenesis by responding to follicle-stimulating hormone and testosterone, secreting tubular fluid to facilitate sperm transport, and phagocytosing residual cytoplasm shed during sperm maturation.
Leydig Cells and Endocrine Control
Leydig cells reside within the interstitial tissue between seminiferous tubules and serve as the primary source of testosterone. In response to luteinising hormone, these cells synthesise and secrete testosterone, which diffuses locally into the seminiferous tubules and systemically into the circulation. Within the testis, testosterone concentrations are far higher than circulating levels, a gradient that is critical for maintaining normal spermatogenesis.
Endocrine regulation within the testis operates through tightly controlled feedback loops. Sertoli cells produce inhibin B, which acts on the anterior pituitary to modulate follicle-stimulating hormone secretion. This feedback system allows fine-tuning of sperm production according to functional demand and testicular activity rather than relying solely on systemic hormone levels.
Stages of Spermatogenesis
Spermatogenesis progresses through a sequence of developmental stages that transform undifferentiated germ cells into specialised male gametes. In the spermatogonial phase, spermatogonia located on the basal lamina divide by mitosis, maintaining the stem cell population while generating cells destined for differentiation. These early germ cells remain within the basal compartment of the tubule.
During the meiotic phase, primary spermatocytes cross the blood–testis barrier into the adluminal compartment and undergo meiosis I and II. This process reduces chromosome number and introduces genetic variation, producing haploid spermatids. The final phase, spermiogenesis, involves extensive cellular remodelling. Spermatids condense their nuclear material, form an acrosome essential for fertilisation, develop a flagellum for motility, reorganise mitochondria within the midpiece, and shed excess cytoplasm. Despite this transformation, sperm released into the tubule lumen are structurally complete but functionally immature.
Epididymal Maturation and Sperm Transport
Newly formed spermatozoa are transported from the seminiferous tubules into the epididymis, where they undergo further maturation over approximately 12 to 20 days. Within the epididymis, sperm acquire progressive motility, membrane stability, and the capacity to fertilise an oocyte. This maturation depends on luminal factors and a carefully regulated microenvironment rather than additional cell division.
Mature sperm are stored primarily in the tail of the epididymis until ejaculation. During ejaculation, sperm travel through the vas deferens and mix with secretions from the seminal vesicles, prostate, and bulbourethral glands to form semen. This final combination supports sperm viability, motility, and delivery, completing a process that began weeks earlier within the seminiferous tubules.
Clinical Connections
Disruption of spermatogenesis is one of the most common contributors to male infertility and often reflects impairment at multiple levels of testicular function rather than a single defect. Because sperm production depends on precise temperature regulation, intact tubular architecture, hormonal support, and epididymal maturation, disturbances in any of these processes can reduce sperm count, motility, or morphology. Importantly, abnormalities may develop gradually and only become apparent when fertility is assessed.
A range of clinical conditions interfere with normal sperm production and maturation. Varicocele alters testicular temperature and venous drainage, cryptorchidism exposes the testis to prolonged heat stress, and infections or inflammatory processes can damage seminiferous tubules. Toxic exposures, including alcohol, anabolic steroids, environmental toxins, chemotherapy, and radiation, preferentially injure rapidly dividing germ cells. Damage to the blood–testis barrier may expose sperm antigens to the immune system, resulting in anti-sperm antibody formation and impaired fertilisation. Common mechanisms leading to impaired spermatogenesis include:
Thermal disruption, as seen with varicocele, undescended testes, or prolonged heat exposure
Germ cell injury, from infection, inflammation, chemotherapy, radiation, or toxins
Hormonal imbalance, involving luteinising hormone, follicle-stimulating hormone, or testosterone deficiency
Immune-mediated effects, following breakdown of the blood–testis barrier
Endocrine disorders play a significant role in male reproductive dysfunction. Inadequate luteinising hormone reduces intratesticular testosterone, while follicle-stimulating hormone deficiency impairs Sertoli cell support of developing germ cells. Because intratesticular testosterone levels must remain far higher than circulating concentrations, even subtle endocrine disruption can significantly affect sperm production. Hormonal evaluation is therefore a key component of infertility assessment when spermatogenesis is impaired.
Semen analysis provides an integrated snapshot of male reproductive function. It reflects not only spermatogenesis within the seminiferous tubules, but also epididymal maturation, ductal patency, and contributions from accessory glands. Abnormal results should be interpreted in the context of sperm production timelines, as changes in semen parameters often represent events that occurred several weeks earlier.
Concept Check
What roles do Sertoli cells play in supporting spermatogenesis and maintaining the blood–testis barrier?
Why is intratesticular testosterone essential, and how is its production regulated?
What are the major stages of spermatogenesis, and what key events occur in each stage?
How does the epididymis contribute to sperm maturation after they leave the seminiferous tubules?
Why is temperature regulation critical for normal sperm production?